mrvi-2023123100018232392023FYFALSEhttp://fasb.org/us-gaap/2023#AccountingStandardsUpdate201602MemberP2Yhttp://fasb.org/us-gaap/2023#OtherAssetsNoncurrenthttp://fasb.org/us-gaap/2023#PropertyPlantAndEquipmentAndFinanceLeaseRightOfUseAssetAfterAccumulatedDepreciationAndAmortizationhttp://fasb.org/us-gaap/2023#PropertyPlantAndEquipmentAndFinanceLeaseRightOfUseAssetAfterAccumulatedDepreciationAndAmortizationhttp://fasb.org/us-gaap/2023#PropertyPlantAndEquipmentAndFinanceLeaseRightOfUseAssetAfterAccumulatedDepreciationAndAmortizationhttp://fasb.org/us-gaap/2023#OtherAssetsNoncurrenthttp://fasb.org/us-gaap/2023#OtherAssetsNoncurrenthttp://fasb.org/us-gaap/2023#AccruedLiabilitiesCurrenthttp://fasb.org/us-gaap/2023#AccruedLiabilitiesCurrenthttp://fasb.org/us-gaap/2023#OtherLiabilitiesNoncurrenthttp://fasb.org/us-gaap/2023#OtherLiabilitiesNoncurrentP4Yzerozerozero00018232392023-01-012023-12-3100018232392023-06-30iso4217:USD0001823239us-gaap:CommonClassAMember2024-02-21xbrli:shares0001823239us-gaap:CommonClassBMember2024-02-2100018232392023-12-3100018232392022-12-310001823239us-gaap:NonrelatedPartyMember2023-12-310001823239us-gaap:NonrelatedPartyMember2022-12-310001823239us-gaap:RelatedPartyMember2023-12-310001823239us-gaap:RelatedPartyMember2022-12-310001823239us-gaap:CommonClassAMember2023-12-31iso4217:USDxbrli:shares0001823239us-gaap:CommonClassAMember2022-12-310001823239us-gaap:CommonClassBMember2023-12-310001823239us-gaap:CommonClassBMember2022-12-3100018232392022-01-012022-12-3100018232392021-01-012021-12-310001823239us-gaap:CommonClassAMemberus-gaap:CommonStockMember2020-12-310001823239us-gaap:CommonClassBMemberus-gaap:CommonStockMember2020-12-310001823239us-gaap:AdditionalPaidInCapitalMember2020-12-310001823239us-gaap:RetainedEarningsMember2020-12-310001823239us-gaap:AccumulatedOtherComprehensiveIncomeMember2020-12-310001823239us-gaap:NoncontrollingInterestMember2020-12-3100018232392020-12-3100018232392020-01-012020-12-310001823239us-gaap:RetainedEarningsMembersrt:CumulativeEffectPeriodOfAdoptionAdjustmentMember2020-12-310001823239us-gaap:NoncontrollingInterestMembersrt:CumulativeEffectPeriodOfAdoptionAdjustmentMember2020-12-310001823239srt:CumulativeEffectPeriodOfAdoptionAdjustmentMember2020-12-310001823239us-gaap:CommonClassAMemberus-gaap:CommonStockMember2021-01-012021-12-310001823239us-gaap:CommonClassBMemberus-gaap:CommonStockMember2021-01-012021-12-310001823239us-gaap:AdditionalPaidInCapitalMember2021-01-012021-12-310001823239us-gaap:NoncontrollingInterestMember2021-01-012021-12-310001823239us-gaap:RetainedEarningsMember2021-01-012021-12-310001823239us-gaap:AccumulatedOtherComprehensiveIncomeMember2021-01-012021-12-310001823239us-gaap:CommonClassAMemberus-gaap:CommonStockMember2021-12-310001823239us-gaap:CommonClassBMemberus-gaap:CommonStockMember2021-12-310001823239us-gaap:AdditionalPaidInCapitalMember2021-12-310001823239us-gaap:RetainedEarningsMember2021-12-310001823239us-gaap:AccumulatedOtherComprehensiveIncomeMember2021-12-310001823239us-gaap:NoncontrollingInterestMember2021-12-3100018232392021-12-310001823239us-gaap:CommonClassAMemberus-gaap:CommonStockMember2022-01-012022-12-310001823239us-gaap:AdditionalPaidInCapitalMember2022-01-012022-12-310001823239us-gaap:NoncontrollingInterestMember2022-01-012022-12-310001823239us-gaap:RetainedEarningsMember2022-01-012022-12-310001823239us-gaap:CommonClassAMemberus-gaap:CommonStockMember2022-12-310001823239us-gaap:CommonClassBMemberus-gaap:CommonStockMember2022-12-310001823239us-gaap:AdditionalPaidInCapitalMember2022-12-310001823239us-gaap:RetainedEarningsMember2022-12-310001823239us-gaap:AccumulatedOtherComprehensiveIncomeMember2022-12-310001823239us-gaap:NoncontrollingInterestMember2022-12-310001823239us-gaap:CommonClassBMemberus-gaap:CommonStockMember2023-01-012023-12-310001823239us-gaap:AdditionalPaidInCapitalMember2023-01-012023-12-310001823239us-gaap:NoncontrollingInterestMember2023-01-012023-12-310001823239us-gaap:CommonClassAMemberus-gaap:CommonStockMember2023-01-012023-12-310001823239us-gaap:RetainedEarningsMember2023-01-012023-12-310001823239us-gaap:CommonClassAMemberus-gaap:CommonStockMember2023-12-310001823239us-gaap:CommonClassBMemberus-gaap:CommonStockMember2023-12-310001823239us-gaap:AdditionalPaidInCapitalMember2023-12-310001823239us-gaap:RetainedEarningsMember2023-12-310001823239us-gaap:AccumulatedOtherComprehensiveIncomeMember2023-12-310001823239us-gaap:NoncontrollingInterestMember2023-12-310001823239mrvi:MaravaiLifeSciencesHoldingsLLCMember2023-01-012023-12-310001823239mrvi:MaravaiLifeSciencesHoldingsLLCMember2022-01-012022-12-310001823239mrvi:MaravaiLifeSciencesHoldingsLLCMember2021-01-012021-12-310001823239mrvi:MaravaiLifeSciencesHoldings2LLCMember2023-01-012023-12-310001823239mrvi:MaravaiLifeSciencesHoldings2LLCMember2022-01-012022-12-310001823239mrvi:MaravaiLifeSciencesHoldings2LLCMember2021-01-012021-12-31mrvi:segment0001823239mrvi:NucleicAcidProductionSegmentMembersrt:NorthAmericaMember2023-01-012023-12-310001823239mrvi:BiologicsSafetyTestingSegmentMembersrt:NorthAmericaMember2023-01-012023-12-310001823239srt:NorthAmericaMember2023-01-012023-12-310001823239mrvi:NucleicAcidProductionSegmentMemberus-gaap:EMEAMember2023-01-012023-12-310001823239mrvi:BiologicsSafetyTestingSegmentMemberus-gaap:EMEAMember2023-01-012023-12-310001823239us-gaap:EMEAMember2023-01-012023-12-310001823239srt:AsiaPacificMembermrvi:NucleicAcidProductionSegmentMember2023-01-012023-12-310001823239srt:AsiaPacificMembermrvi:BiologicsSafetyTestingSegmentMember2023-01-012023-12-310001823239srt:AsiaPacificMember2023-01-012023-12-310001823239mrvi:NucleicAcidProductionSegmentMembermrvi:LatinAndCentralAmericaMember2023-01-012023-12-310001823239mrvi:BiologicsSafetyTestingSegmentMembermrvi:LatinAndCentralAmericaMember2023-01-012023-12-310001823239mrvi:LatinAndCentralAmericaMember2023-01-012023-12-310001823239mrvi:NucleicAcidProductionSegmentMember2023-01-012023-12-310001823239mrvi:BiologicsSafetyTestingSegmentMember2023-01-012023-12-310001823239mrvi:NucleicAcidProductionSegmentMembersrt:NorthAmericaMember2022-01-012022-12-310001823239mrvi:BiologicsSafetyTestingSegmentMembersrt:NorthAmericaMember2022-01-012022-12-310001823239srt:NorthAmericaMember2022-01-012022-12-310001823239mrvi:NucleicAcidProductionSegmentMemberus-gaap:EMEAMember2022-01-012022-12-310001823239mrvi:BiologicsSafetyTestingSegmentMemberus-gaap:EMEAMember2022-01-012022-12-310001823239us-gaap:EMEAMember2022-01-012022-12-310001823239srt:AsiaPacificMembermrvi:NucleicAcidProductionSegmentMember2022-01-012022-12-310001823239srt:AsiaPacificMembermrvi:BiologicsSafetyTestingSegmentMember2022-01-012022-12-310001823239srt:AsiaPacificMember2022-01-012022-12-310001823239mrvi:NucleicAcidProductionSegmentMembermrvi:LatinAndCentralAmericaMember2022-01-012022-12-310001823239mrvi:BiologicsSafetyTestingSegmentMembermrvi:LatinAndCentralAmericaMember2022-01-012022-12-310001823239mrvi:LatinAndCentralAmericaMember2022-01-012022-12-310001823239mrvi:NucleicAcidProductionSegmentMember2022-01-012022-12-310001823239mrvi:BiologicsSafetyTestingSegmentMember2022-01-012022-12-310001823239mrvi:NucleicAcidProductionSegmentMembersrt:NorthAmericaMember2021-01-012021-12-310001823239mrvi:BiologicsSafetyTestingSegmentMembersrt:NorthAmericaMember2021-01-012021-12-310001823239mrvi:ProteinDetectionSegmentMembersrt:NorthAmericaMember2021-01-012021-12-310001823239srt:NorthAmericaMember2021-01-012021-12-310001823239mrvi:NucleicAcidProductionSegmentMemberus-gaap:EMEAMember2021-01-012021-12-310001823239mrvi:BiologicsSafetyTestingSegmentMemberus-gaap:EMEAMember2021-01-012021-12-310001823239mrvi:ProteinDetectionSegmentMemberus-gaap:EMEAMember2021-01-012021-12-310001823239us-gaap:EMEAMember2021-01-012021-12-310001823239srt:AsiaPacificMembermrvi:NucleicAcidProductionSegmentMember2021-01-012021-12-310001823239srt:AsiaPacificMembermrvi:BiologicsSafetyTestingSegmentMember2021-01-012021-12-310001823239srt:AsiaPacificMembermrvi:ProteinDetectionSegmentMember2021-01-012021-12-310001823239srt:AsiaPacificMember2021-01-012021-12-310001823239mrvi:NucleicAcidProductionSegmentMembermrvi:LatinAndCentralAmericaMember2021-01-012021-12-310001823239mrvi:BiologicsSafetyTestingSegmentMembermrvi:LatinAndCentralAmericaMember2021-01-012021-12-310001823239mrvi:ProteinDetectionSegmentMembermrvi:LatinAndCentralAmericaMember2021-01-012021-12-310001823239mrvi:LatinAndCentralAmericaMember2021-01-012021-12-310001823239mrvi:NucleicAcidProductionSegmentMember2021-01-012021-12-310001823239mrvi:BiologicsSafetyTestingSegmentMember2021-01-012021-12-310001823239mrvi:ProteinDetectionSegmentMember2021-01-012021-12-310001823239us-gaap:ShippingAndHandlingMember2023-01-012023-12-310001823239us-gaap:ShippingAndHandlingMember2022-01-012022-12-310001823239us-gaap:ShippingAndHandlingMember2021-01-012021-12-310001823239srt:MinimumMember2023-01-012023-12-310001823239srt:MaximumMember2023-01-012023-12-31xbrli:pure0001823239mrvi:MaravaiTopcoHoldingsLLCMember2023-12-310001823239mrvi:MaravaiTopcoHoldingsLLCMembermrvi:MaravaiLifeSciencesHoldingsLLCMember2023-12-310001823239mrvi:MaravaiTopcoHoldingsLLCMembermrvi:MaravaiLifeSciencesHoldingsLLCMembermrvi:TaxDistributionMember2023-01-012023-12-310001823239mrvi:MaravaiTopcoHoldingsLLCMembermrvi:MaravaiLifeSciencesHoldingsLLCMembermrvi:TaxDistributionMember2022-01-012022-12-310001823239mrvi:MaravaiTopcoHoldingsLLCMembermrvi:MaravaiLifeSciencesHoldingsLLCMembermrvi:TaxDistributionMember2021-01-012021-12-310001823239us-gaap:LeaseholdImprovementsMember2023-12-310001823239srt:MinimumMemberus-gaap:FurnitureAndFixturesMember2023-12-310001823239us-gaap:FurnitureAndFixturesMembersrt:MaximumMember2023-12-310001823239us-gaap:RevenueFromContractWithCustomerMembermrvi:NacalaiUSAIncMemberus-gaap:CustomerConcentrationRiskMember2023-01-012023-12-310001823239mrvi:NacalaiUSAIncMembermrvi:AccountsReceivableBenchmarkMemberus-gaap:CustomerConcentrationRiskMember2023-01-012023-12-310001823239mrvi:NacalaiUSAIncMembermrvi:AccountsReceivableBenchmarkMemberus-gaap:CustomerConcentrationRiskMember2022-01-012022-12-310001823239mrvi:CureVacMemberus-gaap:RevenueFromContractWithCustomerMemberus-gaap:CustomerConcentrationRiskMember2021-01-012021-12-310001823239mrvi:CureVacMembermrvi:AccountsReceivableBenchmarkMemberus-gaap:CustomerConcentrationRiskMember2023-01-012023-12-310001823239mrvi:CureVacMembermrvi:AccountsReceivableBenchmarkMemberus-gaap:CustomerConcentrationRiskMember2022-01-012022-12-310001823239mrvi:BioNTechSEMemberus-gaap:RevenueFromContractWithCustomerMemberus-gaap:CustomerConcentrationRiskMember2022-01-012022-12-310001823239mrvi:BioNTechSEMemberus-gaap:RevenueFromContractWithCustomerMemberus-gaap:CustomerConcentrationRiskMember2021-01-012021-12-310001823239mrvi:BioNTechSEMembermrvi:AccountsReceivableBenchmarkMemberus-gaap:CustomerConcentrationRiskMember2022-01-012022-12-310001823239us-gaap:RevenueFromContractWithCustomerMembermrvi:PfizerIncMemberus-gaap:CustomerConcentrationRiskMember2022-01-012022-12-310001823239us-gaap:RevenueFromContractWithCustomerMembermrvi:PfizerIncMemberus-gaap:CustomerConcentrationRiskMember2021-01-012021-12-310001823239mrvi:PfizerIncMembermrvi:AccountsReceivableBenchmarkMemberus-gaap:CustomerConcentrationRiskMember2022-01-012022-12-310001823239mrvi:AlphazymeLLCMember2023-01-182023-01-180001823239mrvi:AlphazymeLLCMember2023-01-012023-12-310001823239mrvi:AlphazymeLLCMember2023-01-182023-06-300001823239mrvi:AlphazymeLLCMember2023-06-012023-06-300001823239mrvi:AlphazymeLLCMembermrvi:SecuritiesPurchaseAgreementMaximumPerformancePaymentMember2023-01-180001823239mrvi:SecuritiesPurchaseAgreementRetentionPaymentMember2023-01-180001823239mrvi:MyChemLegacyOwnersMembermrvi:AlphazymeLLCMember2023-01-182023-01-180001823239mrvi:MyChemLegacyOwnersMembermrvi:SecuritiesPurchaseAgreementRetentionPaymentMembermrvi:AlphazymeLLCMemberus-gaap:ResearchAndDevelopmentExpenseMember2023-01-012023-12-310001823239mrvi:AlphazymeLLCMember2023-01-180001823239mrvi:AlphazymeLLCMember2023-07-012023-09-300001823239mrvi:PotentialWorkingCapitalAdjustmentsMembermrvi:AlphazymeLLCMember2023-01-180001823239mrvi:AlphazymeLLCMembermrvi:SecureRepresentationsAndWarrantiesMember2023-01-180001823239mrvi:PotentialWorkingCapitalAdjustmentsMembermrvi:AlphazymeLLCMember2023-04-012023-06-300001823239mrvi:AlphazymeLLCMember2023-04-012023-06-300001823239us-gaap:TradeNamesMembermrvi:AlphazymeLLCMember2023-01-180001823239us-gaap:TradeNamesMembermrvi:AlphazymeLLCMember2023-01-182023-01-180001823239mrvi:AlphazymeLLCMemberus-gaap:DevelopedTechnologyRightsMember2023-01-180001823239mrvi:AlphazymeLLCMemberus-gaap:DevelopedTechnologyRightsMember2023-01-182023-01-180001823239mrvi:AlphazymeLLCMemberus-gaap:CustomerRelationshipsMember2023-01-180001823239mrvi:AlphazymeLLCMemberus-gaap:CustomerRelationshipsMember2023-01-182023-01-180001823239srt:MinimumMembermrvi:AlphazymeLLCMemberus-gaap:ValuationTechniqueDiscountedCashFlowMembermrvi:MeasurementInputRevenueGrowthRateMember2023-01-180001823239mrvi:AlphazymeLLCMemberus-gaap:ValuationTechniqueDiscountedCashFlowMembermrvi:MeasurementInputRevenueGrowthRateMembersrt:MaximumMember2023-01-180001823239us-gaap:MeasurementInputDiscountRateMembermrvi:AlphazymeLLCMemberus-gaap:ValuationTechniqueDiscountedCashFlowMember2023-01-180001823239mrvi:AlphazymeLLCMemberus-gaap:ValuationTechniqueDiscountedCashFlowMembermrvi:MeasurementInputObsolescentCurveMember2023-01-180001823239mrvi:MyChemLLCMember2022-01-272022-01-270001823239mrvi:MyChemLLCMember2022-01-012022-12-310001823239mrvi:MyChemLLCMember2022-01-272022-11-300001823239mrvi:MyChemLLCMember2022-11-012022-11-300001823239mrvi:MyChemLLCMembermrvi:SecuritiesPurchaseAgreementMaximumPerformancePaymentMember2022-01-270001823239mrvi:MyChemLLCMembermrvi:SecuritiesPurchaseAgreementRetentionPaymentMember2022-01-27mrvi:employee0001823239mrvi:MyChemLLCMembermrvi:MyChemLegacyOwnersMember2022-01-272022-01-270001823239mrvi:MyChemLLCMembermrvi:MyChemLegacyOwnersMembermrvi:SecuritiesPurchaseAgreementRetentionPaymentMemberus-gaap:CostOfSalesMember2023-01-012023-12-310001823239mrvi:MyChemLLCMembermrvi:MyChemLegacyOwnersMembermrvi:SecuritiesPurchaseAgreementRetentionPaymentMemberus-gaap:ResearchAndDevelopmentExpenseMember2023-01-012023-12-310001823239mrvi:MyChemLLCMembermrvi:MyChemLegacyOwnersMembermrvi:SecuritiesPurchaseAgreementRetentionPaymentMemberus-gaap:ResearchAndDevelopmentExpenseMember2022-01-012022-12-310001823239mrvi:MyChemLLCMembermrvi:SecuritiesPurchaseAgreementCompletionOfAcquiredInventoryMember2023-12-310001823239mrvi:MyChemLLCMembermrvi:SecuritiesPurchaseAgreementCompletionOfAcquiredInventoryMember2023-01-282023-01-280001823239mrvi:MyChemLLCMembermrvi:SecuritiesPurchaseAgreementCompletionOfAcquiredInventoryMember2023-01-292023-12-310001823239mrvi:MyChemLLCMember2022-01-270001823239mrvi:MyChemLLCMember2022-10-012022-12-310001823239mrvi:MyChemLLCMembermrvi:PotentialWorkingCapitalAdjustmentsMember2022-01-270001823239mrvi:MyChemLLCMembermrvi:SecureRepresentationsAndWarrantiesMember2022-01-270001823239mrvi:MyChemLLCMembermrvi:PotentialWorkingCapitalAdjustmentsMember2022-10-012022-12-310001823239mrvi:MyChemLLCMembermrvi:SecureRepresentationsAndWarrantiesMember2023-01-282023-03-310001823239mrvi:MyChemLLCMembermrvi:IndemnificationOfPreClosingLiabilitiesMember2023-01-292023-12-310001823239mrvi:MyChemLLCMemberus-gaap:TradeNamesMember2022-01-270001823239mrvi:MyChemLLCMemberus-gaap:TradeNamesMember2022-01-272022-01-270001823239mrvi:MyChemLLCMemberus-gaap:DevelopedTechnologyRightsMember2022-01-270001823239mrvi:MyChemLLCMemberus-gaap:DevelopedTechnologyRightsMember2022-01-272022-01-270001823239mrvi:MyChemLLCMemberus-gaap:CustomerRelationshipsMember2022-01-270001823239mrvi:MyChemLLCMemberus-gaap:CustomerRelationshipsMember2022-01-272022-01-270001823239mrvi:MyChemLLCMembersrt:MinimumMemberus-gaap:ValuationTechniqueDiscountedCashFlowMembermrvi:MeasurementInputRevenueGrowthRateMember2022-01-270001823239mrvi:MyChemLLCMemberus-gaap:ValuationTechniqueDiscountedCashFlowMembermrvi:MeasurementInputRevenueGrowthRateMembersrt:MaximumMember2022-01-270001823239mrvi:MyChemLLCMemberus-gaap:MeasurementInputDiscountRateMemberus-gaap:ValuationTechniqueDiscountedCashFlowMember2022-01-270001823239mrvi:MyChemLLCMembersrt:MinimumMemberus-gaap:ValuationTechniqueDiscountedCashFlowMembermrvi:MeasurementInputObsolescentCurveMember2022-01-270001823239mrvi:MyChemLLCMemberus-gaap:ValuationTechniqueDiscountedCashFlowMembermrvi:MeasurementInputObsolescentCurveMembersrt:MaximumMember2022-01-270001823239us-gaap:DisposalGroupDisposedOfBySaleNotDiscontinuedOperationsMembermrvi:VectorLaboratoriesIncMember2021-08-012021-08-310001823239us-gaap:DisposalGroupDisposedOfBySaleNotDiscontinuedOperationsMembermrvi:VectorLaboratoriesIncMember2021-09-012021-09-300001823239us-gaap:DisposalGroupDisposedOfBySaleNotDiscontinuedOperationsMembermrvi:VectorLaboratoriesIncMember2021-01-012021-12-310001823239us-gaap:DisposalGroupDisposedOfBySaleNotDiscontinuedOperationsMembermrvi:VectorLaboratoriesIncMember2021-12-310001823239srt:MinimumMemberus-gaap:DisposalGroupDisposedOfBySaleNotDiscontinuedOperationsMembermrvi:VectorLaboratoriesIncMember2023-01-012023-12-310001823239us-gaap:DisposalGroupDisposedOfBySaleNotDiscontinuedOperationsMembermrvi:VectorLaboratoriesIncMembersrt:MaximumMember2023-01-012023-12-310001823239mrvi:MLSH1IncentiveUnitMember2021-01-012021-12-310001823239mrvi:CostRealignmentPlanMember2023-11-012023-11-300001823239mrvi:NucleicAcidProductionSegmentMembermrvi:CostRealignmentPlanMember2023-01-012023-12-310001823239us-gaap:CorporateMembermrvi:CostRealignmentPlanMember2023-01-012023-12-310001823239mrvi:CostRealignmentPlanMember2023-01-012023-12-310001823239mrvi:CostRealignmentPlanMemberus-gaap:EmployeeSeveranceMember2022-12-310001823239mrvi:StockBasedCompensationExpenseBenefitMembermrvi:CostRealignmentPlanMember2022-12-310001823239us-gaap:FacilityClosingMembermrvi:CostRealignmentPlanMember2022-12-310001823239us-gaap:OtherRestructuringMembermrvi:CostRealignmentPlanMember2022-12-310001823239mrvi:CostRealignmentPlanMember2022-12-310001823239mrvi:CostRealignmentPlanMemberus-gaap:EmployeeSeveranceMember2023-01-012023-12-310001823239mrvi:StockBasedCompensationExpenseBenefitMembermrvi:CostRealignmentPlanMember2023-01-012023-12-310001823239us-gaap:FacilityClosingMembermrvi:CostRealignmentPlanMember2023-01-012023-12-310001823239us-gaap:OtherRestructuringMembermrvi:CostRealignmentPlanMember2023-01-012023-12-310001823239mrvi:CostRealignmentPlanMemberus-gaap:EmployeeSeveranceMember2023-12-310001823239mrvi:StockBasedCompensationExpenseBenefitMembermrvi:CostRealignmentPlanMember2023-12-310001823239us-gaap:FacilityClosingMembermrvi:CostRealignmentPlanMember2023-12-310001823239us-gaap:OtherRestructuringMembermrvi:CostRealignmentPlanMember2023-12-310001823239mrvi:CostRealignmentPlanMember2023-12-310001823239mrvi:CostRealignmentPlanMemberus-gaap:SubsequentEventMember2024-01-310001823239mrvi:NucleicAcidProductionSegmentMembermrvi:CostRealignmentPlanMemberus-gaap:SubsequentEventMember2024-01-310001823239us-gaap:CorporateMembermrvi:CostRealignmentPlanMemberus-gaap:SubsequentEventMember2024-01-31mrvi:reporting_unit0001823239mrvi:ProteinDetectionSegmentMember2023-01-012023-12-3100018232392023-10-012023-12-310001823239mrvi:NucleicAcidProductionSegmentMember2022-12-310001823239mrvi:BiologicsSafetyTestingSegmentMember2022-12-310001823239mrvi:NucleicAcidProductionSegmentMember2023-12-310001823239mrvi:BiologicsSafetyTestingSegmentMember2023-12-310001823239srt:MinimumMember2023-12-310001823239srt:MaximumMember2023-12-310001823239us-gaap:TradeNamesMember2023-12-310001823239us-gaap:TradeNamesMembersrt:MinimumMember2023-12-310001823239us-gaap:TradeNamesMembersrt:MaximumMember2023-12-310001823239us-gaap:TechnologyBasedIntangibleAssetsMember2023-12-310001823239srt:MinimumMemberus-gaap:TechnologyBasedIntangibleAssetsMember2023-12-310001823239us-gaap:TechnologyBasedIntangibleAssetsMembersrt:MaximumMember2023-12-310001823239us-gaap:CustomerRelationshipsMember2023-12-310001823239srt:MinimumMemberus-gaap:CustomerRelationshipsMember2023-12-310001823239us-gaap:CustomerRelationshipsMembersrt:MaximumMember2023-12-310001823239us-gaap:TradeNamesMember2022-12-310001823239us-gaap:TradeNamesMembersrt:MinimumMember2022-12-310001823239us-gaap:TradeNamesMembersrt:MaximumMember2022-12-310001823239us-gaap:TechnologyBasedIntangibleAssetsMember2022-12-310001823239srt:MinimumMemberus-gaap:TechnologyBasedIntangibleAssetsMember2022-12-310001823239us-gaap:TechnologyBasedIntangibleAssetsMembersrt:MaximumMember2022-12-310001823239us-gaap:CustomerRelationshipsMember2022-12-310001823239srt:MinimumMemberus-gaap:CustomerRelationshipsMember2022-12-310001823239us-gaap:CustomerRelationshipsMembersrt:MaximumMember2022-12-310001823239mrvi:AlphazymeLLCMember2023-03-310001823239us-gaap:CostOfSalesMember2023-01-012023-12-310001823239us-gaap:CostOfSalesMember2022-01-012022-12-310001823239us-gaap:CostOfSalesMember2021-01-012021-12-310001823239us-gaap:SellingGeneralAndAdministrativeExpensesMember2023-01-012023-12-310001823239us-gaap:SellingGeneralAndAdministrativeExpensesMember2022-01-012022-12-310001823239us-gaap:SellingGeneralAndAdministrativeExpensesMember2021-01-012021-12-310001823239us-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel1Memberus-gaap:MoneyMarketFundsMember2023-12-310001823239us-gaap:FairValueInputsLevel2Memberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:MoneyMarketFundsMember2023-12-310001823239us-gaap:FairValueMeasurementsRecurringMemberus-gaap:MoneyMarketFundsMemberus-gaap:FairValueInputsLevel3Member2023-12-310001823239us-gaap:FairValueMeasurementsRecurringMemberus-gaap:MoneyMarketFundsMember2023-12-310001823239us-gaap:InterestRateCapMemberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel1Member2023-12-310001823239us-gaap:FairValueInputsLevel2Memberus-gaap:InterestRateCapMemberus-gaap:FairValueMeasurementsRecurringMember2023-12-310001823239us-gaap:InterestRateCapMemberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel3Member2023-12-310001823239us-gaap:InterestRateCapMemberus-gaap:FairValueMeasurementsRecurringMember2023-12-310001823239us-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel1Member2023-12-310001823239us-gaap:FairValueInputsLevel2Memberus-gaap:FairValueMeasurementsRecurringMember2023-12-310001823239us-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel3Member2023-12-310001823239us-gaap:FairValueMeasurementsRecurringMember2023-12-310001823239us-gaap:InterestRateCapMemberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel1Member2022-12-310001823239us-gaap:FairValueInputsLevel2Memberus-gaap:InterestRateCapMemberus-gaap:FairValueMeasurementsRecurringMember2022-12-310001823239us-gaap:InterestRateCapMemberus-gaap:FairValueMeasurementsRecurringMemberus-gaap:FairValueInputsLevel3Member2022-12-310001823239us-gaap:InterestRateCapMemberus-gaap:FairValueMeasurementsRecurringMember2022-12-310001823239us-gaap:MeasurementInputDiscountRateMembermrvi:AlphazymeLLCMember2023-01-18mrvi:payment0001823239mrvi:MyChemLLCMemberus-gaap:MeasurementInputDiscountRateMember2022-01-270001823239mrvi:MyChemLLCMember2022-04-012022-06-300001823239us-gaap:LeaseholdImprovementsMember2022-12-310001823239us-gaap:FurnitureAndFixturesMember2023-12-310001823239us-gaap:FurnitureAndFixturesMember2022-12-310001823239us-gaap:SoftwareDevelopmentMember2023-12-310001823239us-gaap:SoftwareDevelopmentMember2022-12-310001823239mrvi:DepreciablePropertyPlantAndEquipmentMember2023-12-310001823239mrvi:DepreciablePropertyPlantAndEquipmentMember2022-12-310001823239us-gaap:ConstructionInProgressMember2023-12-310001823239us-gaap:ConstructionInProgressMember2022-12-310001823239mrvi:MyChemLLCMember2023-12-310001823239mrvi:MyChemLLCMember2022-12-310001823239mrvi:AlphazymeLLCMember2023-12-310001823239mrvi:AlphazymeLLCMember2022-12-310001823239mrvi:SanDiegoCaliforniaMember2022-05-31mrvi:building0001823239mrvi:CooperativeAgreementMember2022-05-012022-05-310001823239mrvi:CooperativeAgreementMember2022-05-310001823239mrvi:CooperativeAgreementMember2023-01-012023-12-310001823239mrvi:CooperativeAgreementMember2023-12-310001823239mrvi:CooperativeAgreementMember2022-01-012022-12-310001823239us-gaap:OtherAssetsMembermrvi:CooperativeAgreementMember2022-12-310001823239us-gaap:PropertyPlantAndEquipmentMembermrvi:CooperativeAgreementMember2022-12-310001823239mrvi:CooperativeAgreementMember2022-12-310001823239mrvi:JupiterFloridaMember2023-01-31mrvi:extension_option0001823239mrvi:SanDiegoCaliforniaFlandersIMember2023-03-310001823239mrvi:SanDiegoCaliforniaFlandersIIMember2023-06-300001823239mrvi:NewCreditAgreementMemberus-gaap:LetterOfCreditMemberus-gaap:LineOfCreditMember2023-12-310001823239mrvi:NewCreditAgreementMemberus-gaap:SecuredDebtMemberus-gaap:LineOfCreditMember2020-10-310001823239mrvi:NewCreditAgreementMemberus-gaap:RevolvingCreditFacilityMemberus-gaap:LineOfCreditMember2020-10-310001823239mrvi:NewCreditAgreementMemberus-gaap:SecuredDebtMembermrvi:SecuredOvernightFinancingRateSOFRMemberus-gaap:LineOfCreditMember2022-01-012022-01-310001823239mrvi:NewCreditAgreementMemberus-gaap:RevolvingCreditFacilityMemberus-gaap:BaseRateMemberus-gaap:LineOfCreditMember2022-01-012022-01-310001823239mrvi:NewCreditAgreementInitialTermLoansMemberus-gaap:BaseRateMemberus-gaap:LineOfCreditMember2022-01-012022-01-310001823239mrvi:NewCreditAgreementInitialTermLoansMembermrvi:SecuredOvernightFinancingRateSOFRMemberus-gaap:LineOfCreditMember2022-01-012022-01-310001823239mrvi:SecuredOvernightFinancingRateSOFRMembermrvi:NewCreditAgreementNonInitialTermLoansMemberus-gaap:LineOfCreditMember2022-01-012022-01-310001823239mrvi:NewCreditAgreementMemberus-gaap:SecuredDebtMemberus-gaap:LineOfCreditMember2023-12-310001823239mrvi:NewCreditAgreementMemberus-gaap:LetterOfCreditMemberus-gaap:LineOfCreditMember2022-01-310001823239mrvi:NewCreditAgreementMemberus-gaap:SecuredDebtMemberus-gaap:LineOfCreditMember2022-01-012022-01-310001823239mrvi:NewCreditAgreementMember2022-01-012022-01-310001823239mrvi:NewCreditAgreementMember2020-01-012020-12-310001823239mrvi:NewCreditAgreementMember2023-01-012023-12-31mrvi:loan0001823239mrvi:NewCreditAgreementMemberus-gaap:SecuredDebtMemberus-gaap:LineOfCreditMember2022-01-310001823239mrvi:NewCreditAgreementMemberus-gaap:RevolvingCreditFacilityMemberus-gaap:LineOfCreditMember2022-01-310001823239mrvi:NewCreditAgreementMemberus-gaap:RevolvingCreditFacilityMemberus-gaap:LineOfCreditMember2023-12-310001823239mrvi:NewCreditAgreementMemberus-gaap:SecuredDebtMembersrt:MaximumMemberus-gaap:LineOfCreditMember2023-12-310001823239srt:MinimumMembermrvi:NewCreditAgreementMemberus-gaap:SecuredDebtMemberus-gaap:LineOfCreditMember2023-12-310001823239mrvi:NewCreditAgreementMemberus-gaap:SecuredDebtMemberus-gaap:LineOfCreditMember2022-03-012022-03-310001823239mrvi:NewCreditAgreementMemberus-gaap:RevolvingCreditFacilityMembersrt:MaximumMemberus-gaap:LineOfCreditMember2022-01-012022-01-310001823239srt:MinimumMembermrvi:NewCreditAgreementMemberus-gaap:RevolvingCreditFacilityMemberus-gaap:LineOfCreditMember2022-01-012022-01-310001823239us-gaap:InterestRateCapMember2021-03-310001823239us-gaap:InterestRateCapMember2022-05-310001823239us-gaap:InterestRateCapMember2023-12-310001823239mrvi:NewCreditAgreementMemberus-gaap:SecuredDebtMemberus-gaap:LineOfCreditMember2022-12-310001823239mrvi:NewCreditAgreementMemberus-gaap:RevolvingCreditFacilityMemberus-gaap:LineOfCreditMember2022-12-310001823239us-gaap:CommonClassAMember2020-11-300001823239us-gaap:CommonClassBMember2020-11-3000018232392020-11-30mrvi:vote0001823239us-gaap:CommonClassAMember2023-01-012023-12-310001823239us-gaap:CommonClassBMember2023-01-012023-12-310001823239mrvi:SecondaryOfferingMember2021-04-012021-04-300001823239mrvi:SecondaryOfferingByMLSH2Member2021-04-012021-04-300001823239us-gaap:OverAllotmentOptionMember2021-04-012021-04-300001823239us-gaap:OverAllotmentOptionMember2021-04-300001823239mrvi:SecondaryOfferingMembermrvi:MaravaiLifeSciencesHoldingsLLCMember2021-04-012021-04-300001823239mrvi:SecondaryOfferingMember2021-09-012021-09-300001823239mrvi:SecondaryOfferingByMLSH2Member2021-09-012021-09-300001823239us-gaap:OverAllotmentOptionMember2021-09-300001823239mrvi:SecondaryOfferingMembermrvi:MaravaiLifeSciencesHoldingsLLCMember2021-09-012021-09-300001823239mrvi:MaravaiTopcoHoldingsLLCMember2021-12-012021-12-3100018232392021-12-012021-12-3100018232392021-12-312021-12-310001823239us-gaap:CommonClassBMember2021-12-310001823239mrvi:AlphazymeHoldingsIncMember2023-01-222023-01-2200018232392023-01-222023-01-220001823239mrvi:MaravaiLifeSciencesHoldingsIncAndAlphazymeHoldingsIncMember2023-01-222023-01-220001823239mrvi:MaravaiLifeSciencesHoldingsLLCMember2023-01-222023-01-220001823239us-gaap:CommonClassBMember2023-01-220001823239us-gaap:RestrictedStockUnitsRSUMember2023-01-012023-12-310001823239us-gaap:RestrictedStockUnitsRSUMember2022-01-012022-12-310001823239us-gaap:RestrictedStockUnitsRSUMember2021-01-012021-12-310001823239us-gaap:EmployeeStockOptionMember2023-01-012023-12-310001823239us-gaap:EmployeeStockOptionMember2022-01-012022-12-310001823239us-gaap:EmployeeStockOptionMember2021-01-012021-12-310001823239us-gaap:EmployeeStockMember2023-01-012023-12-310001823239us-gaap:EmployeeStockMember2022-01-012022-12-310001823239us-gaap:EmployeeStockMember2021-01-012021-12-310001823239us-gaap:CommonClassBMember2023-01-012023-12-310001823239us-gaap:CommonClassBMember2022-01-012022-12-310001823239us-gaap:CommonClassBMember2021-01-012021-12-310001823239mrvi:A2020OmnibusIncentivePlanMember2020-11-012020-11-300001823239us-gaap:EmployeeStockMember2020-11-012020-11-300001823239mrvi:PerformanceStockUnitsMember2021-01-012021-12-310001823239us-gaap:EmployeeStockOptionMember2023-01-012023-12-310001823239us-gaap:EmployeeStockOptionMember2022-01-012022-12-310001823239us-gaap:EmployeeStockOptionMember2021-01-012021-12-310001823239us-gaap:RestrictedStockUnitsRSUMember2022-12-310001823239us-gaap:RestrictedStockUnitsRSUMember2023-01-012023-12-310001823239us-gaap:RestrictedStockUnitsRSUMember2023-12-310001823239us-gaap:RestrictedStockUnitsRSUMember2022-01-012022-12-310001823239us-gaap:RestrictedStockUnitsRSUMember2021-01-012021-12-310001823239srt:MinimumMembermrvi:MLSH1IncentiveUnitMember2023-01-012023-12-310001823239mrvi:MLSH1IncentiveUnitMembersrt:MaximumMember2023-01-012023-12-310001823239mrvi:MLSH1IncentiveUnitMember2023-01-012023-12-310001823239mrvi:MLSH1IncentiveUnitMember2022-01-012022-12-310001823239mrvi:MLSH1IncentiveUnitMember2022-12-310001823239mrvi:MLSH1IncentiveUnitMember2023-12-310001823239us-gaap:ResearchAndDevelopmentExpenseMember2023-01-012023-12-310001823239us-gaap:ResearchAndDevelopmentExpenseMember2022-01-012022-12-310001823239us-gaap:ResearchAndDevelopmentExpenseMember2021-01-012021-12-310001823239us-gaap:RestructuringChargesMember2023-01-012023-12-310001823239us-gaap:RestructuringChargesMember2022-01-012022-12-310001823239us-gaap:RestructuringChargesMember2021-01-012021-12-310001823239mrvi:PerformanceStockUnitsMember2023-01-012023-12-310001823239mrvi:MaravaiTopcoHoldingsLLCMember2023-12-310001823239us-gaap:DomesticCountryMember2023-12-310001823239us-gaap:StateAndLocalJurisdictionMember2023-12-310001823239us-gaap:RelatedPartyMembermrvi:MLSH1AndMLSH2Membermrvi:TaxReceivableAgreementPaymentsMember2023-12-310001823239us-gaap:RelatedPartyMembermrvi:TaxReceivableAgreementNonCurrentLiabilityDerecognizedMember2023-01-012023-12-310001823239us-gaap:RelatedPartyMembermrvi:MLSH1AndMLSH2Membermrvi:TaxReceivableAgreementPaymentsMember2023-01-012023-12-310001823239mrvi:TaxReceivableAgreementInterestPaymentsMemberus-gaap:RelatedPartyMembermrvi:MLSH1AndMLSH2Member2023-01-012023-12-310001823239us-gaap:RelatedPartyMembermrvi:MLSH1AndMLSH2Membermrvi:TaxReceivableAgreementPaymentsMember2022-01-012022-12-310001823239mrvi:TaxReceivableAgreementInterestPaymentsMemberus-gaap:RelatedPartyMembermrvi:MLSH1AndMLSH2Member2022-01-012022-12-310001823239us-gaap:RelatedPartyMembermrvi:MLSH1AndMLSH2Membermrvi:TaxReceivableAgreementPaymentsMember2022-12-310001823239mrvi:MaravaiTopcoHoldingsLLCMembermrvi:TaxDistributionMember2023-01-012023-12-310001823239mrvi:MaravaiLifeSciencesHoldingsIncMembermrvi:MaravaiTopcoHoldingsLLCMembermrvi:TaxDistributionMember2023-01-012023-12-310001823239mrvi:MaravaiTopcoHoldingsLLCMembermrvi:TaxDistributionMember2022-01-012022-12-310001823239mrvi:MaravaiLifeSciencesHoldingsIncMembermrvi:MaravaiTopcoHoldingsLLCMembermrvi:TaxDistributionMember2022-01-012022-12-310001823239mrvi:MaravaiTopcoHoldingsLLCMembermrvi:TaxDistributionMember2021-01-012021-12-310001823239mrvi:MaravaiLifeSciencesHoldingsIncMembermrvi:MaravaiTopcoHoldingsLLCMembermrvi:TaxDistributionMember2021-01-012021-12-310001823239mrvi:CuriaGlobalMemberus-gaap:RelatedPartyMembermrvi:ConsultingServicesMember2021-01-012021-12-310001823239us-gaap:RelatedPartyMembermrvi:MaravaiLifeSciencesFoundationMembermrvi:CharitableFoundationContributionMember2021-01-012021-12-310001823239mrvi:CuriaGlobalMemberus-gaap:RelatedPartyMembermrvi:ConsultingServicesMember2022-01-012022-12-310001823239mrvi:CuriaGlobalMemberus-gaap:RelatedPartyMembermrvi:ConsultingServicesMember2023-01-012023-12-310001823239us-gaap:OperatingSegmentsMembermrvi:NucleicAcidProductionSegmentMember2023-01-012023-12-310001823239us-gaap:OperatingSegmentsMembermrvi:NucleicAcidProductionSegmentMember2022-01-012022-12-310001823239us-gaap:OperatingSegmentsMembermrvi:NucleicAcidProductionSegmentMember2021-01-012021-12-310001823239us-gaap:OperatingSegmentsMembermrvi:BiologicsSafetyTestingSegmentMember2023-01-012023-12-310001823239us-gaap:OperatingSegmentsMembermrvi:BiologicsSafetyTestingSegmentMember2022-01-012022-12-310001823239us-gaap:OperatingSegmentsMembermrvi:BiologicsSafetyTestingSegmentMember2021-01-012021-12-310001823239us-gaap:OperatingSegmentsMembermrvi:ProteinDetectionSegmentMember2023-01-012023-12-310001823239us-gaap:OperatingSegmentsMembermrvi:ProteinDetectionSegmentMember2022-01-012022-12-310001823239us-gaap:OperatingSegmentsMembermrvi:ProteinDetectionSegmentMember2021-01-012021-12-310001823239us-gaap:OperatingSegmentsMember2023-01-012023-12-310001823239us-gaap:OperatingSegmentsMember2022-01-012022-12-310001823239us-gaap:OperatingSegmentsMember2021-01-012021-12-310001823239us-gaap:IntersegmentEliminationMember2023-01-012023-12-310001823239us-gaap:IntersegmentEliminationMember2022-01-012022-12-310001823239us-gaap:IntersegmentEliminationMember2021-01-012021-12-31
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
| | | | | |
| ý | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2023
OR
| | | | | |
| o | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
Commission file number 001-39725
Maravai LifeSciences Holdings, Inc.
(Exact name of registrant as specified in its charter)
| | | | | | | | | | | | | | |
| Delaware | | | | 85-2786970 |
| (State or other jurisdiction of incorporation or organization) | | | | (I.R.S. Employer Identification No.) |
| | | | |
10770 Wateridge Circle Suite 200 San Diego, California | | | | 92121 |
(Address of principal executive offices) | | | | (Zip code) |
______________________________
Registrant’s telephone number, including area code: (858) 546-0004
______________________________
Securities registered pursuant to Section 12(b) of the Act:
| | | | | | | | | | | | | | |
| Title of each class | | Trading Symbol(s) | | Name of each exchange on which registered |
| Class A common stock, $0.01 par value | | MRVI | | The Nasdaq Stock Market LLC |
Securities registered pursuant to section 12(g) of the Act: None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes x No o
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes o No x
Indicate by check mark whether the registrant: (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports); and (2) has been subject to such filing requirements for the past 90 days. Yes x No o
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes x No o
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act. (Check one):
| | | | | | | | | | | |
| Large accelerated filer | ý | Accelerated filer | o |
| Non-accelerated filer | o | Smaller reporting company | o |
| | Emerging growth company | o |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. o
Indicate by check mark whether the registrant has filed a report on and attestation to its management's assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report. x
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements. ▢
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b). ▢
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act). Yes ☐ No x
The aggregate market value of the registrant’s voting common equity held by non-affiliates as of June 30, 2023, the last business day of the registrant’s most recently completed second fiscal quarter, was approximately $1,370.0 million, based on the closing price of the registrant’s common stock on the Nasdaq Global Select Market of $12.43 per share.
As of February 21, 2024, 132,305,845 shares of the registrant’s Class A common stock were outstanding and 119,094,026 shares of the registrant’s Class B common stock were outstanding.
DOCUMENTS INCORPORATED BY REFERENCE
The information required by Part III of this Report, to the extent not set forth herein, is incorporated herein by reference from the registrant’s definitive proxy statement relating to the Annual Meeting of Shareholders to be held in 2024, which definitive proxy statement shall be filed with the Securities and Exchange Commission within 120 days after the end of the fiscal year to which this Report relates.
TABLE OF CONTENTS
SPECIAL NOTE REGARDING FORWARD LOOKING STATEMENTS
This Annual Report on Form 10-K contains “forward-looking statements” within the meaning of the safe harbor provisions of the U.S. Private Securities Litigation Reform Act of 1995. Investors are cautioned that statements which are not strictly historical statements constitute forward looking statements, including, without limitation, statements under the captions “Risk Factors,” “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and “Business” and are identified by words like “believe,” “expect,” “may,” “will,” “should,” “seek,” “anticipate,” “intend,” “plan,” “goal,” “project,” “estimate,” “likely,” or “could” and similar expressions.
Forward-looking statements are neither historical facts nor assurances of future performance. Instead, they are based only on our current beliefs, expectations and assumptions regarding the future of our business, future plans and strategies, projections, anticipated events and trends, the economy and other future conditions. Because forward-looking statements relate to the future, they are subject to inherent uncertainties, risks and changes in circumstances that are difficult to predict and many of which are outside of our control. Our actual results and financial condition may differ materially from those indicated in the forward-looking statements. Therefore, you should not rely on any of these forward-looking statements. Important factors that could cause our actual results and financial condition to differ materially from those indicated include those discussed under the heading “Summary of Risk Factors” and “Item 1A. Risk Factors” as well as those discussed elsewhere in this Annual Report on Form 10-K.
Any forward-looking statement made by us in this report is based only on information currently available to us and speaks only as of the date of this report. We undertake no obligation to publicly update any forward-looking statement, whether written or oral, that may be made from time to time, whether as a result of new information, future developments or otherwise.
Part I.
Item 1. Business
Overview
Maravai LifeSciences Holdings, Inc. (also referred to in this document as “Maravai”, “we”, “us” or “the Company”) is a leading life sciences company providing critical products to enable the development of novel vaccines, drug therapies, and diagnostics, and to support research on human diseases. Our customers include the top global biopharmaceutical companies ranked by research and development expenditures according to industry consultants, and many other emerging biopharmaceutical and life sciences research companies, as well as leading academic research institutions and in vitro diagnostics companies. Our products address the key phases of biopharmaceutical development spanning research to commercialization and include complex nucleic acids for vaccine, therapeutic and diagnostic applications, custom enzymes for research and diagnostic use and antibody-based products to detect impurities during the production of biopharmaceutical products. We provide products that support our customers from discovery through commercialization of their vaccines, therapeutic agents and in vitro diagnostic products.
Our businesses principally address high growth market segments in biopharmaceutical development. In particular, the field of cell and gene therapy has emerged as one of the fastest growing treatment modalities to address a host of human conditions. There are more than 4,000 nucleic acid and cell and gene therapies in development or launched and sales in this category are expected to grow more than 3 times by 2030, according to industry consultants and management estimates. Our portfolio offers key products for each stage of the cell and gene therapy development lifecycle. For example, our messenger RNA (“mRNA”) products are used in drug development to assist in the production of immune-activating antigens and to deliver gene editing technologies in cell and gene therapy treatments; our CleanCap® technology is used to stabilize mRNA and streamlines mRNA manufacturing; we were one of the first companies to provide the essential modified uridine, N1-methyl-pseudouridine triphosphate, for research applications and under good manufacturing processes (“GMP”) conditions; our catalog mRNA products are frequently used by lipid developers to test and validate new mRNA delivery platforms; and our plasmid DNA products are used as templates for the production of our mRNA products. Our oligonucleotides, oligonucleotide supply products, enzymes and NTPs are also included in the supply chain of several diagnostic, next-gen sequencing (“NGS”) and research platforms, as well as DNA and RNA synthesis end-markets. We also provide biologics safety testing technology used to ensure the safety of the biological drug manufacturing process and drug products.
Our proprietary capabilities and products underpin the value we aim to provide to our customers. Among other capabilities, we are experts in RNA and mRNA products, which are challenging and often unstable molecules requiring significant chemical modifications and analytical expertise to ensure their stability and efficacy in our customers’ applications. Notably, according to research commissioned by us consisting of interviews with our current and former customers, our competitors and industry experts focused across our two ongoing business segments (the “Industry Analysis”), we believe CleanCap technology is viewed as a leading solution to incorporate the five prime (“5’”) cap into mRNA. CleanCap is a novel chemical approach to produce the 5’ cap analog, which, in addition to making mRNA more stable, aids in protein production and helps prevent an unwanted immune response to the mRNA. CleanCap analogs have been incorporated into several mRNA programs targeting
immunization against the coronavirus, SARS-CoV-2 (“COVID-19”). These programs include commercial programs led by Pfizer in partnership with BioNTech and by BioNTech in partnership with Fosun Pharma, as well as two COVID-19 vaccines which are now approved for use in Japan. In addition to these commercially approved programs, CleanCap has been incorporated into many research and clinical stage programs addressing stand-alone and combination vaccines for COVID-19 and other respiratory diseases, such as Influenza that have not yet been commercialized.
We estimate our CleanCap products have been incorporated in approximately 350 vaccine and therapeutic programs in development as of December 31, 2023, including at least 60 molecules for which our mRNA CDMO services group manufactured the mRNA APIs using CleanCap. These include vaccine development programs for infectious diseases, including Lyme disease, malaria, HIV, tuberculosis, shingles, rabies, yellow fever, respiratory syncytial virus (“RSV”) and Zika, as well as other medical conditions. In addition to infectious diseases, these programs address a number of disease states, including ornithine transcarbamylase deficiency, glycogen storage disorders, Alpha-1 antitrypsin deficiency, acute lymphoblastic leukemia, Hurler syndrome, ovarian cancer and cardiovascular disease. These therapeutic programs also use multiple therapeutic modalities, including CRISPR/Cas-9, transcription activator-like effector nuclease (TALEN), enzyme replacement therapies, allogeneic CAR-T cells and base editing. Should one or more of these programs proceed to commercialization, we believe we will continue to supply our customers and our products will likely be incorporated in customer regulatory filings.
mRNA is at the core of our capabilities. We developed our expertise in mRNA with a belief in its potential as a therapeutic modality. The first clinical trial for an mRNA therapeutic agent occurred in 2016. Now, more than 900 clinical trials are in the pipeline, principally focused on vaccines against viruses and cancer vaccines. With the COVID-19 pandemic, mRNA has shown its potential for more rapid vaccine design and manufacture when compared to traditional techniques involving culturing inactivated virus to elicit an immune response. COVID-19 has helped highlight the potential advantage of mRNA as a treatment modality and directed significant resources to the growing base of knowledge about mRNA. This knowledge is now being directed at future vaccine programs addressing infectious diseases as well as for therapeutic agents for a host of human diseases. We are positioned to serve our biopharmaceutical customers in the fast-growing field of mRNA across a range of clinical programs for a variety of diseases.
Forming long-term partnerships with our customers is core to our strategy. Today, we primarily serve our customers during the product development and process development phases. During product development, we collaborate with our customers to develop and synthesize nucleic acids, which in some cases comprise the active pharmaceutical ingredient (“API”) of our customers’ products in development. We also provide our customers a host of chemically complex and highly specialized raw materials. Process development is a complex phase that establishes highly validated procedures and determines the investment in facilities and equipment required to bring biopharmaceutical products to market. These decisions impact the viability of our customers’ products for use in clinical trials and commercialization. During process development, we provide enzyme-linked immunosorbent assays (“ELISAs”) that reduce the risk posed by impurities and contaminants in biological drugs, a critical step to ensure the safety of the drug product.
While we do not provide products that are themselves regulated as drugs or in vitro diagnostics, our customers frequently incorporate our products into their highly validated products and processes. For example, we provide oligonucleotides and antibody-based products used by in vitro diagnostic product manufacturers for their on-market products. Because of the extensive validation required for these products, these components are frequently purchased for the life of our customers’ products and we believe they are unlikely to be substituted. In addition, our analytical tools are used in the design and development of manufacturing processes and often will be used throughout the life cycle of our customers’ manufactured products. Once our services or products are qualified by our customers, they are often cited in regulatory documents and standard operating procedures. As a result, our customer relationships frequently span many years.
The nature of our products and their uses require that they be manufactured by highly trained personnel in state-of-the-art facilities following exacting procedures to ensure quality. As of December 31, 2023, approximately 24% of our workforce have earned advanced degrees and all receive rigorous training on our procedures. During 2023, the majority of our nucleic acid products were manufactured at our San Diego, California Wateridge facility (“Wateridge facility”). The Wateridge facility was purpose-built to address our customers’ needs for critical raw materials manufactured under certain GMP conditions and APIs for investigational use. Our raw material products are manufactured following the voluntary quality standards of ISO 9001:2015. Our GMP-grade raw materials follow ISO 9001:2015 standards, additional voluntary GMP quality standards and customer specific requirements. Our API products are manufactured following the voluntary quality standards of ISO 9001:2015, the International Council for Harmonisation’s GMP Guide, comparable GMP principles for the European Union and customer specific requirements. We believe our products are exempt from compliance with the current GMP (“cGMP”) regulations of the FDA, as our products are further processed or incorporated into final drug products by our customers and we do not make claims related to their safety or effectiveness. Our other facilities are similarly designed for specific applications with quality systems to match our customers’ requirements. All of our manufacturing facilities meet applicable ISO standards.
We built our business through a combination of acquisitions and subsequent investments in our acquired companies to grow their commercial capabilities, upgrade and expand their research and production facilities, deploy stringent quality systems, integrate their back-office functions, and develop the personnel and management to fuel continued growth. Today, we offer an integrated portfolio that enables innovation across the biopharmaceutical and academic markets. Mergers, acquisitions and strategic partnerships that complement our capabilities in cell and gene therapy and biopharmaceutical production remain core to our strategy. Our strategy aims to augment our strong organic growth with the addition of synergistic products and capabilities.
Our Portfolio and Capabilities
We provide critical raw materials that support our customers from discovery through commercialization of their vaccines, therapeutic agents and in vitro diagnostic products. Our products are frequently incorporated into our customers’ products, whether as research products or APIs used in development or products incorporated as raw materials into on-market products. They may also be incorporated into the manufacturing process itself. We are therefore a critical part of our customers’ supply chain and they frequently seek to maintain their supply relationship with us for the life of their products or development programs.
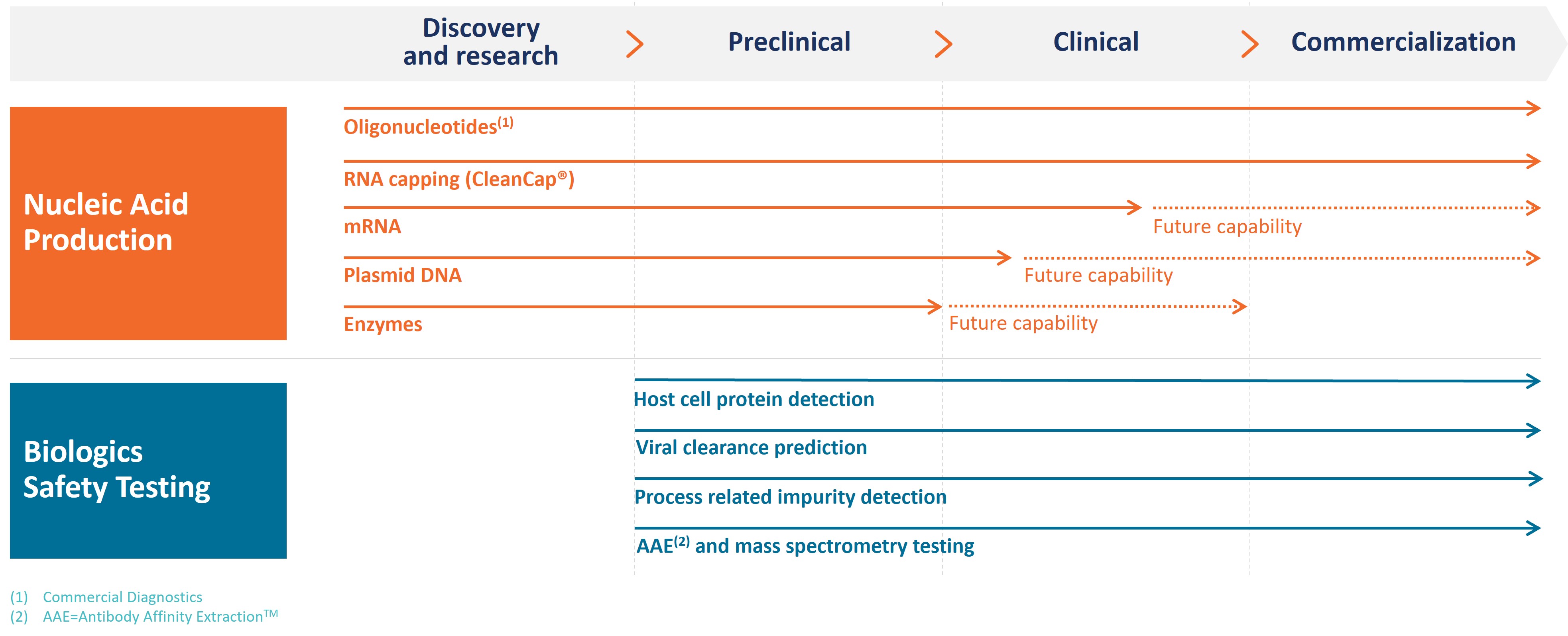
Our products address our customers’ needs for Nucleic Acid Production and Biologics Safety Testing, and our operations are aligned to these two segments. Within Nucleic Acid Production, we have four business units: TriLink Discovery, TriLink GMP, Glen Research and Alphazyme. Our Biologics Safety Testing business is comprised of Cygnus Technologies. Our brands, products and the end markets they serve are depicted in the following image:
Nucleic Acid Production (78% of Revenue for the Year Ended December 31, 2023)
We are a global provider of highly modified, complex nucleic acids and related products. We have recognized expertise in complex chemistries and products provided under exacting quality standards. Our core offerings include mRNA, long and short oligonucleotides, our proprietary CleanCap mRNA capping technology, mRNA building blocks, oligonucleotide building blocks and specialty enzymes. Our offerings address key customer needs for critical components, from research to GMP-grade raw materials and API manufacturing. We market our nucleic acid products under the TriLink BioTechnologies®, Glen Research and Alphazyme brands.
Since the 1961 discovery of mRNA and its role in converting genetic information into proteins, countless attempts had been made to harness mRNA for therapeutic purposes. Today, mRNA vaccines are globally recognized for their success in responding to the COVID-19 pandemic.
The success of COVID-19 vaccines has helped highlight the potential advantage of mRNA as a treatment modality and significant investments have been made industry wide to developing future mRNA vaccines as well as for therapeutic agents for
a host of human diseases. We are positioned to serve our biopharmaceutical customers in the fast-growing field of mRNA across a range of clinical programs for a variety of diseases.
We offer the following nucleic acid products: mRNA, RNA Capping (CleanCap), oligonucleotides, oligonucleotide inputs, nucleoside triphosphates, custom nucleic acid chemistry, plasmid DNA and specialty enzymes.
mRNA. mRNA is an intermediary molecule that translates the genetic information stored in DNA into proteins. The genetic information stored in DNA is transferred to mRNA in a cellular process called transcription. This process occurs in the nucleus of cells. DNA, a double stranded molecule, is unwound and copied as mRNA by the enzyme RNA polymerase. mRNA is then transferred out of the nucleus to the cytosol, a component of the cytoplasm of a cell, where it serves as a blueprint for making cellular proteins by a multi-component organelle complex called the ribosome.
mRNA has traditionally been a difficult molecule for vaccine and therapeutic purposes. mRNA is inherently unstable compared to DNA and is susceptible to degradation by ubiquitous enzymes called RNases. mRNAs are also physically and chemically fragile and can degrade at elevated temperatures and under shear forces that occur during downstream manufacturing processes. We have developed manufacturing processes that overcome many of these obstacles, resulting in highly effective mRNA.
We develop and manufacture mRNA products to support vaccine and therapeutic programs from pre-clinical development through and including clinical phases, including scale-up and analytical development services. The mRNA molecules may serve as APIs for diverse applications, such as enzyme replacement therapies, gene editing therapies and vaccines. We offer both research grade material and material made under GMP conditions for early phase clinical trials.
RNA Capping. Within the mRNA category, we also offer our patented CleanCap technology. CleanCap analogs principally serve the mRNA vaccine and therapeutics markets. Cap analogs are a component of mRNA that aids in protein production as well as in making mRNA more stable inside cells. For mRNA to serve as a template to make a protein, it requires a special cap at the 5’ end of the molecule. The cap structure also affects the stability of the mRNA. Lack of a cap can result in activation of the innate immune system, which can affect the production of the desired protein or elicit undesired biological effects. We offer a suite of CleanCap analogs that are specifically made for therapeutics and vaccines. CleanCap analogs are sold as a stand-alone reagents or bundled with other mRNA products. Based on the Industry Analysis, we believe our cap analogs are critical features of several mRNA vaccines and therapies in development.
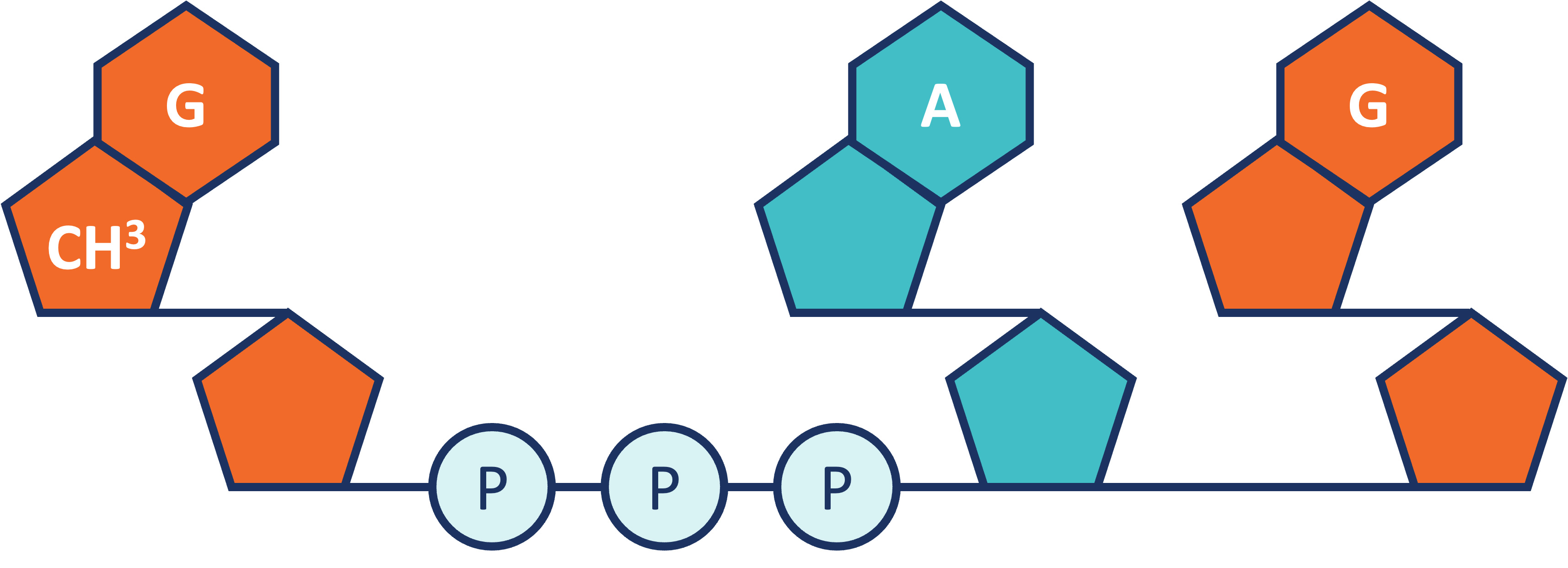
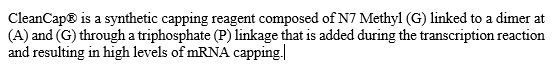
Traditionally, the 5’ cap has been added in one of two ways. The cap can be added post mRNA synthesis by an enzymatic process. This enzymatic method has several drawbacks, including the high cost of the capping enzymes as well as the need to perform additional processing steps to the mRNA to remove enzymes and byproducts of the capping reaction. While capping efficiency is usually high, the extra processing steps typically result in degradation and mRNA of poorer quality. The second method is to add a synthetic cap analog into the transcription reaction such that the mRNA is transcribed and capped in a single step. Anti-reverse cap analog (“ARCA”) is an example of a cap analog that is added to the transcription reaction. This avoids the workflow challenges of the enzymatic process, but typically results in lower yields.
Like ARCA, CleanCap analogs are synthetic, chemically-made mRNA 5’ cap analogs added to the transcription process in a single step. Unlike ARCA, however, CleanCap results in significantly higher levels of capping efficiency, resulting in very low levels of uncapped mRNA, which in turn minimizes the risk of activation of the innate immune system. In addition, CleanCap’s higher mRNA yields compared to ARCA result in lower cost of goods. When compared to enzymatic capping, CleanCap removes the additional downstream purification steps required.
We currently offer several variations of the CleanCap molecule, serving the needs of mRNA and self-amplifying RNA developers. CleanCap is available in two quality grades, research use only for discovery and development activities, and a GMP-grade for clinical and commercial applications. Our newest CleanCap analog, CleanCap M6, was introduced in May 2023 and is our most robust cap analog to date, enabling mRNA that delivers higher levels of protein production.
CleanCap mRNA products represented 69% of our Nucleic Acid Production revenue for the year ended December 31, 2023 (including the revenue from CleanCap products).
Oligonucleotides. The oligonucleotide product category supports broad customer applications, including therapeutics, in vitro diagnostics, NGS and CRISPR-based gene editing. Most of our TriLink BioTechnologies oligonucleotide products are custom manufactured DNA or RNA sequences, often highly modified and produced as research grade or under GMP conditions for use in development, clinical and commercial applications.
Oligonucleotide Synthesis Inputs. Our product offerings through Glen Research include reagents and support supplies for DNA and RNA oligonucleotide synthesis, labeling, modification and purification. We are a reputable and trusted vendor with a large portfolio, quality brand, knowledgeable technical support, and responsive customer service. In addition to oligonucleotide synthesis service providers, our customer base includes life science, biopharma, and diagnostic companies as well as academic institutions and government organizations, all of which internally manufacture their own oligonucleotide products.
Nucleoside triphosphates. Nucleoside triphosphates (“NTPs”) are the precursors to DNA and RNA. They are composed of a nitrogen base bound to either ribose or deoxyribose with three phosphate groups added to the sugar. We manufacture NTPs that are used in polymerase chain reactions (“PCR”), in sequencing reactions and in the manufacture of mRNA. The NTPs can be unmodified, composed of the four standard bases, or modified, with a base altered to enhance a particular biological property, such as the ability to evade the innate immune system in therapeutic applications. TriLink BioTechnologies NTPs are used by customers in both research and clinical trial applications. Our manufacturing capabilities for NTPs now includes both research use and GMP-grade.
Custom Nucleic Acid Chemistry. Through our acquisition of MyChem LLC in the first quarter of 2022, TriLink BioTechnologies expanded its synthetic chemistry expertise and added proprietary manufacturing processes allowing for the highest purity NTP, amidite and custom nucleotide services. We serve a diverse market of diagnostics and therapeutic developers that require novel molecules that are otherwise unavailable on the market. Typically, these molecules are initially manufactured in small quantities, and then scaled to meet the need of larger diagnostic platform or therapeutic applications once positive candidates have been identified by the customer.
Plasmid DNA. Unlike genomic DNA, which constitutes the chromosome, plasmid DNA exists outside the chromosome and represents small circular double-stranded constructs. Plasmid DNA is frequently used as a vector for replicating nucleic acid products. Plasmid DNA is integral to the production of mRNA, serving as the nucleic acid template for the DNA-dependent RNA polymerases that frequently are used in the manufacturing of mRNA. Our plasmid DNA offering allows us to ensure the quality and timeliness of the mRNA API manufacturing campaigns that we service for our customers.
Specialty Enzymes. Through our acquisition of Alphazyme in the first quarter of 2023, our Nucleic Acid Production capabilities now include specialized enzymes. Enzymes are critical to almost every phase of nucleic acid production. Alphazyme provides custom, scalable molecular biology enzymes with a full product line of IVT, NGS, life science and diagnostic enzyme solutions.
Discovery mRNA synthesis. Through the TriLink BioTechnologies Discovery Business unit, we offer a core set of products and services geared toward customers doing early-stage development work. We produce mRNA utilizing standard sequences for generalized research using customer supplied sequences for custom built constructs. We also provide process development services to optimize customers’ transcription and purification processes. These services can be integrated with our CleanCap, NTP products and enzyme portfolios and have access to our analytical and QC method development.
GMP mRNA synthesis. Our GMP mRNA manufacturing services offer a clear pathway for customers running clinical trials. We focus on building partnerships with our customers in the emerging market of cell and gene therapy to ensure we are well-positioned to be an extension of their development teams. Our services feature robust quality management systems and include process development and scale-up, phase-appropriate, high-quality plasmid DNA, regulatory submission support, and in-house analytical services for mRNA analysis and characterization.
Biologics Safety Testing (22% of Revenue for the Year Ended December 31, 2023)
We provide products and services under the Cygnus Technologies®, LLC (“Cygnus Technologies”) brand that ensure the purity of our customers’ biopharmaceutical products, including biological drugs. For over 25 years, the Cygnus Technologies brand has been associated with products and services that enable the detection of impurities present in bioproduction. Our biologics safety testing products are used during development and scale-up, during the regulatory approval process and
throughout commercialization. We are recognized globally for the detection of host cell proteins (“HCPs”) and process-related impurities during bioproduction.
Our customers in this segment manufacture a broad range of biopharmaceutical products. These include monoclonal antibodies and recombinant proteins, both as novel biologics and biosimilars, and recombinant vaccines, including oncolytic vaccines to treat cancer. We also provide products that support the development and commercialization of cell and gene therapies. Recombinant vaccines and cell and gene therapies rely on manufacturing of various viral vectors produced using recombinant nucleic acid and cell culture technologies. Viral vector manufacturing processes require rigorous analytics, including testing for process-related impurities such as HCPs, host cell DNA, purification leachates, growth media additives and enzymes used in viral vector purification processes. Of all process-related impurities, HCPs present the most complex impurity. Per regulatory requirements, viral vectors used as a component of CAR-T cell therapies or as gene therapies must be produced in certain cell lines, purified and tested for the presence of host cell proteins. All of the 19 existing FDA-and EMA-approved CAR-T Cell and Gene Therapies use Cygnus Host Cell Protein ELISA kits for HCP testing for commercial product lot release. Four of these 19 therapies were approved in 2023.
ELISA is the benchmark method for monitoring levels of process-related impurities during the purification process and in product release testing. The advantages of well-developed ELISA kits include the ability to measure very low levels of impurities in the presence of high amounts of drug product, and are readily transferable across an organization from process development to manufacturing and quality control bioanalytical groups. Though relatively simple to run, these ELISA kits require a high level of expertise to design, develop and qualify.
Customers establishing biopharmaceutical manufacturing processes may use off-the-shelf or generic HCP kits provided by manufacturers like ourselves, or they may choose to design their own in-house assays for their specific processes. Some customers may choose to use generic assays early in development and migrate to process-specific assays later. The trend in recent years has been for customers to increasingly use generic assays throughout their development and commercialization pathway, relying on our expertise, the established performance of our assays supported by our comprehensive state-of-the-art assay qualification services. If customers choose to develop process-specific assays, we offer custom antibody production and assay development as well as characterization services to meet their needs.
Our comprehensive catalog of Cygnus Technologies HCP ELISA kits cover 24 expression platforms and provides the specificity and sensitivity to detect impurities with reproducibility, which supports regulatory compliance. Our reputation for quality is recognized by the industry and global regulatory agencies, with Cygnus Technologies assays used as reference methods throughout the industry and to support manufacturing and quality control of commercialized biologics and gene therapy products.
Our customers in this segment are biopharmaceutical companies, contract research organizations (“CROs”), contract development and manufacturing organizations (“CDMOs”) and life science companies.
Cygnus Technologies product categories include HCP ELISA kits, other bioprocess impurity and contaminant ELISA kits, viral clearance prediction kits, ancillary reagents and custom services.
HCP ELISA kits. HCP ELISA kits are bioassays used to detect residual proteins from the expression system used in bioproduction. HCPs constitute a major group of process-related impurities produced using cell culture technology no matter what cell expression platform is used. HCPs pose potential health risks for patients and the risk of failure of safety endpoints for drug manufacturers. When present in the administered product, even at low levels, HCPs can induce an undesired immune response, interfere with drug efficacy and impact drug stability. HCPs are a critical quality attribute for biologics safety testing development and must be adequately removed during the downstream purification process.
Other impurity and contaminant kits. Products in this category include kits for measuring Protein A leachate, which results from the affinity purification method used for monoclonal antibody therapeutic agents; ELISA kits for measuring additives in growth media, such as bovine serum albumin; kits for measuring host cell DNA; ELISA kits to detect and quantify residual endonuclease impurities in recombinant viral vector and vaccine preparations: and ELISA kits to quantify residual AAV2, AAV8, AAV9 ligands resulting from affinity purification method used for adeno associated virus (AAV)-based gene therapies.
Viral Clearance Prediction kits. In 2020, Cygnus Technologies introduced the MockV® Minute Virus of Mice (“MVM”) kit, a novel, proprietary viral clearance prediction tool that includes a non-infectious “mock virus particle” mimicking the physicochemical properties of live virus that may be present endogenously in the drug substance or introduced during bioproduction. The kit enables manufacturers to conduct viral clearance assessments easily and economically and to predict outcomes in-house ahead of costly and logistically challenging live viral clearance studies. In 2022, Cygnus Technologies introduced MockV® RVLP Kit. This kit enables bioprocess scientists to quantify the removal of Retrovirus-like Particles (RVLPs) produced endogenously by Chinese Hamster Ovary (CHO) cell lines during biopharmaceutical manufacturing. The kit includes a highly purified and concentrated stock solution of RVLP, an actual non-infectious retrovirus-like contaminant generated during CHO production. In the early 1990s, global regulatory agencies such as the FDA realized the prevalence of this particle and became concerned about the retroviral safety of CHO-derived biopharmaceuticals. Since then, the biopharmaceutical industry has relied on CROs to propagate Xenotropic Murine Leukemia Virus (XMuLV) as a model retrovirus to demonstrate effective clearance. With the availability of the MockV® RVLP Kit, biopharmaceutical companies can now independently assess the removal of the original retroviral particle of regulatory concern, derived directly from CHO cells.
Ancillary reagents. These products include antibodies, antigens, sample diluents and other auxiliary products necessary to optimize applications for customer processes.
Custom services. We provide process-specific antibody and ELISA development, qualification and maintenance services. In addition, we have pioneered advanced orthogonal methods including antibody affinity extraction (AAE™) and mass spectrometry for HCP antibodies coverage analysis and HCP identification, which we provide as custom services.
Our Competitive Strengths
We believe we are a leader in providing nucleic acid products and services and biologics safety testing products and services to biopharmaceutical customers worldwide. Our success is built on the ability of our proprietary technologies and products, provided under exacting quality standards, to reliably serve our customers’ needs for critical raw materials, and the process innovation, quality, analytical expertise and reliability of our services.
Leading Supplier of Critical Solutions for Life Sciences from Discovery to Commercialization
We seek to be an important component of our customers’ supply chain by providing inputs that are central to the performance of their products and processes throughout the product lifecycle. By collaborating with customers early in the development phase, our products frequently follow our customers’ development path to commercialization and are likely to be incorporated as raw materials in their on-market products and processes. Our decades-long experience and track record, coupled with our ongoing investment in facilities and quality systems, allow our customers to rely on us for their critical products. Our approach is to be a trusted partner throughout the life cycle of our customers’ products.
Innovation, Proprietary Technologies and Expertise Underpin Our Portfolio
Our expertise in complex chemistries leads customers to seek our collaboration in designing complex products that meet high performance expectations. Based on the responses to the Industry Analysis, we believe the solutions we provide, in many cases, cannot be provided effectively by our competitors. In certain cases, like our CleanCap technology, our know-how features differentiated performance characteristics and is backed by intellectual property. In other cases, such as our HCP products, our antibodies are proprietary and therefore can only be supplied by us. We believe the proprietary nature of our expertise and products solidifies our long-term customer relationships.
Products with Outstanding Quality Performance
We believe our products stand out when compared to those of our competitors’ because they present innovative solutions to customer needs, as indicated by the responses to the Industry Analysis, while providing reliable performance and quality. CleanCap, for example, offers advantages over competing capping technologies in yield, process efficiency, stability and safety. Our oligonucleotides address complex chemistry challenges, which we believe few competitors can address. The results of the Industry Analysis indicate that our HCP ELISA kits have defined the market for impurity detection and we believe they have become a de facto standard in biologics safety testing.
Trusted Brands
Our TriLink BioTechnologies, Glen Research, Alphazyme and Cygnus Technologies brands are well known in their respective markets for innovation, consistent quality, and performance. This brand recognition has been earned over decades. Our manufacturing processes, quality standards, technical support and high-touch customer service ensure that we maintain the reputation of our brands.
State-of-the-Art Manufacturing Facilities
Our biopharmaceutical customers manufacture their products to meet stringent quality standards under strict regulatory guidelines and expect their critical suppliers to meet their exacting requirements. Our customers further expect that we have the production capacity to meet their needs in a timely manner.
Maravai has designed and constructed four world-class manufacturing facilities, and since 2022 has expanded the company’s facility footprint by over 95,000 square feet to support expanded capabilities and future growth.
As of December 31, 2023, we had invested approximately $93.2 million into our flagship San Diego, California Wateridge facility and its dedicated manufacturing suites to produce materials from Research Use Only (“RUO”) to GMP conditions, along with the required quality systems to meet requirements specified by our customers. This investment in our Wateridge facility allows us to meet our customers’ demand for our nucleic acid products, including CleanCap.
In January 2023, we moved into a new state-of-the-art facility in Leland, North Carolina to increase our manufacturing and development capacity to further support the Biologics Safety Testing business.
In March 2023, we expanded our San Diego, California manufacturing campus by making a significant investment in additional cleanroom and small molecule manufacturing space, implementing automation systems and adding support areas to augment production capacity with the Flanders 1 facility (“Flanders 1 facility”) for GMP small molecule production. This facility provides site redundancy for our Wateridge facility and features five GMP suites that can operate in parallel.
In June 2023, we finished construction and took occupancy of an additional 32,000 square-foot facility in San Diego (“Flanders 2 facility”) for GMP-grade mRNA manufacturing to support customers progressing into Phase II clinical trials and beyond.
In June 2023, we expanded the Alphazyme facility in Jupiter, Florida to add an additional research and development (“R&D”) space to support our specialized enzyme development.
Experienced Leaders and Talented Workforce
Our management includes experienced leaders with demonstrated records of success at Maravai and other highly regarded industry participants. They have decades of combined experience and expertise on the forefront of life sciences innovation. In addition, as of December 31, 2023, approximately 24% of our workforce have earned advanced degrees and all receive rigorous on the job training. We believe the quality of our personnel is critical to ensuring the collaborative, long-standing relationships we maintain with many of our customers.
Our Markets
We participate in two distinct market segments: Nucleic Acid Production and Biologics Safety Testing. Our businesses principally address high growth market segments in biopharmaceutical development. In particular, the field of cell and gene therapy has emerged as one of the fastest growing treatment modalities to address a host of human conditions.
Biopharmaceutical customers are increasingly relying on outside parties to provide important raw material inputs and services for their clinical research and manufacturing, a development driving growth for suppliers with unique capabilities, high quality and the ability to manufacture at a relevant scale to support customer programs. We believe that suppliers like ourselves, with
this rare combination of capabilities, proprietary products and the required investment in manufacturing and quality systems, are benefiting from rapid growth as biopharmaceutical customers seek to partner with a small number of trusted partners.
In addition to the continued trend toward outsourcing, several market developments should contribute to long-term growth in our addressable market segments, including:
•Pivot toward mRNA vaccines and therapeutics for non-COVID indications has been accelerated in part by COVID-19. The first two vaccines approved for use in combating the COVID-19 pandemic were mRNA vaccines, including the vaccine developed by Pfizer and BioNTech which uses our CleanCap product. Our CleanCap product is also incorporated into the booster vaccines developed by Pfizer and BioNTech. The mRNA platforms are gaining prominence as a result of their fast development time, lower relative manufacturing costs and proven safety profile. Pfizer and BioNTech are now developing a combined COVID-19 and Influenza vaccine that has been placed into a designation that can “fast-track” a regulatory decision by the FDA. In addition to the COVID-19 vaccines, mRNA technology is being investigated for a spectrum of other infectious diseases as well as cancer vaccines, including personalized medicine vaccines. RNA expertise is highly specialized, and customers seek partners with our expertise to provide these complex products. A small number of providers, like ourselves, with a successful track record for COVID-19, can provide this level of RNA capability.
•Rapid growth in development of cell and gene therapies. Seven new cell and gene therapy approvals (Omisirge, Vyjuvek, Roctavian, Lantidra, Casgevy, Lyfgenia, Elvidys) were all granted FDA or EMA approval in 2023 and have added clinical credibility to cell and gene therapies. Our internal analysis, supported by third-party research, projects that by 2027, 20-30% of the mRNA pipeline assets will be for in vivo gene editing and ex vivo gene-edited cell therapies. We support the development of cell and gene therapies by providing products used in gene editing and cell therapy research. For example, our host cell protein assays are used during the manufacturing of viral vectors and plasmid DNA. Further, we participate by providing the critical high quality synthetic guide RNA and mRNA that encodes for gene-editing enzymes, such as Cas9 that are used in vivo gene editing and ex vivo gene-edited cell therapies.
•Large and growing pipeline of protein-based therapeutics. In addition to cell and gene therapies, an increase in protein-based therapies is driving the need for impurity testing during process development and manufacturing from our Biologics Safety Testing business. Classical biologics are evolving to be expressed in vivo via mRNA. Our analysis suggests that therapeutic proteins and protein replacement may represent as much as 25% of the mRNA pipeline by 2027. We are well positioned to leverage our service capabilities and deep understanding of mRNA biology to serve our customers’ needs to express these large, complex, peptide-based molecules.
Nucleic Acid Production Market
The nucleic acid production market includes the production and synthesis of reagents for research and manufacturing of DNA and RNA-based biologics, including cell and gene therapies, mRNA therapeutics and synthetic biology approaches.
The field of mRNA-based drugs and vaccines has advanced dramatically within a few short years. Providers of technical expertise and manufacturing capabilities, like ourselves, with the facilities and quality systems demanded by biopharmaceutical customers, benefit from the demand created in the mRNA category.
Biologics Safety Testing Market
The biologics safety testing market includes the detection and clearance of downstream bioprocessing product-related and process-related impurities. We participate in the HCP and other process related impurities and viral contamination segments of this market for biopharmaceutical vaccine and therapeutics manufacturing. The growth in this market is driven by continued growth of biologics and biosimilars, viral vector manufacturing for rapidly-growing CAR-T and gene therapy modalities, and increased outsourcing of process development.
Our Strategy
Our customers strive to improve human health. Our goal is to provide them with products and services to accelerate their development efforts, from basic research through clinical trials and ultimately to commercialization for therapeutics, diagnostics and vaccines.
Supporting Biopharmaceutical Customers from Discovery Through to Commercialization
Our customers include both emerging and established biopharmaceutical leaders developing novel drugs, therapeutics, diagnostics and vaccines. Emerging biopharmaceutical customers frequently seek the support we can offer in our state-of-the-art facilities under our stringent quality standards, with the capabilities that result from the capital and process investments we have made over the last several years. We are capable of manufacturing reagents from research-grade to GMP-grade, which often exceeds the in-house capabilities of our pre-commercial customers. The results of the Industry Analysis indicate that our emerging and established customers also seek us out for our leading capabilities in nucleic acid chemistries and process control assays. We have expertise in complex chemistries, especially in highly modified nucleic acids and mRNA, and we believe we are a leader in applying these capabilities to the development of vaccines and therapeutics. We further support our customers as they transition from product development to commercialization by providing critical raw materials for their drugs. A core component of our strategy is the continued investment in facilities, quality standards and products and services that allow us to support our customers through the entire life cycle of their drugs.
Developing Proprietary Technologies that Deepen our Relationships with Our Customers
We believe we are experts in nucleic acids and our scientists aim to develop proprietary enabling technologies that become integral to our customers’ products. For example, CleanCap, our proprietary chemical capping technology, has demonstrated its advantages in terms of the stability of the associated mRNA and its efficiency in protein production when compared to traditional capping technologies. This efficiency has led biopharmaceutical customers to employ CleanCap in their vaccine and therapeutic programs. As those products proceed through development into commercialization, we believe CleanCap will be a critical input in on-market vaccines and therapeutics.
Forming Long-Term Partnerships for Critical Biopharmaceutical Components and Process Tests
Our products are frequently incorporated into regulated and highly validated therapeutic and diagnostic products and processes. Our biopharmaceutical customers expect us to provide them with consistent, high-quality products that meet narrow specifications, and that we ensure their supply chain for such products for the length of their programs. In many cases, we may be the sole source of the products we provide. We therefore take seriously our responsibility to our biopharmaceutical partners, and by extension the patients they serve. Our emphasis on partnership generally leads to long-term relationships with our customers.
Focusing Our Efforts on High Growth End Markets
While biopharmaceutical research and in vitro diagnostics markets are experiencing strong growth, we target the highest growth segments within those markets. Our product portfolio is well positioned to serve the biologic, cell and gene therapy and mRNA vaccine and therapeutic end markets, which are currently experiencing above-market growth. By investing in technologies at the forefront of biopharmaceutical and in vitro diagnostics, we aim to remain focused on the highest-growth applications.
Acquiring Leading Life Sciences Businesses and Supporting Their Continued Development
We built our business by acquiring established and emerging companies with strong scientific foundations in our target markets and investing in their systems, processes and people to accelerate their growth and expand their technologies. Going forward, we may pursue strategic acquisitions that we believe meet, or could meet after being acquired and expanded, the following criteria:
•address our core target markets;
•have a demonstrated adherence to high quality standards;
•be leaders in their market niche(s);
•have differentiated or proprietary products and processes that provide clear value to our biopharmaceutical and other customers; and
•have a track record of attractive rates of growth and compelling returns on invested capital.
Our acquisition strategy is to invest significantly in our acquired businesses. We strive to rapidly integrate their quality, human resources, information and financial systems into our shared services. All of our companies share a common enterprise resource planning system, and we implement our financial controls and reporting systems soon after acquisition. We seek opportunities to invest in their facilities and personnel to provide an operating foundation for growth. We also augment their commercial capabilities through a combination of sales and marketing resources dedicated to each business, supported by our global marketing infrastructure.
We will continue to seek a balance between driving growth organically and inorganically through acquisitions.
Commercial
We have relationships with the following categories of customers: developers of therapeutics and vaccines, other biopharmaceutical and life science research companies, academic institutions and molecular diagnostic companies. Our biopharmaceutical customers include startups, established biotechnology companies and large pharmaceutical companies developing enzyme replacement therapies, gene editing therapies, ex vivo therapies and vaccines.
Our commercial function includes direct sales, marketing, customer service, technical support and distributor management. We serve customers through direct sales in each business segment, with a primary focus on our biopharmaceutical and large diagnostics and commercial customers. We serve our academic customers via web, email and phone ordering as well as through key partnerships where our reagent products are included in their mRNA kits. We support all customers with live technical support and customer service.
We address customers outside the United States with a combination of direct sales and distributors. We serve many of our biopharmaceutical customers, especially in our nucleic acid production segment, via direct sales worldwide. Our distributors also sell our products in over 40 countries and provide customer service and local sales and marketing.
Competition
We compete with a range of companies across our segments.
Nucleic Acid Production
Within nucleic acid production, we compete with four primary types of companies: (1) chemistry companies that create and produce the basic monomers, amidites, and supports that go into the creation of an oligonucleotide; (2) oligonucleotide manufacturers that specialize in custom oligonucleotide development of varying complexities and scales; (3) mRNA biotechnology companies that create fully processed mRNA and specialize in custom, complex orders; and (4) CDMOs that have the capability to accept work from large biopharmaceutical companies and serve as the outsourcing entity for the development and manufacturing of nucleic acid products. However, it is important to note that CDMOs seldom offer proprietary products.
For mRNA capping analogs, we compete principally with Thermo Fisher Scientific, Aldevron (a subsidiary of Danaher), and New England BioLabs, who offer alternatives to CleanCap with enzymatic capping solutions. Many biopharmaceutical companies produce capping solutions in-house using enzymatic or ARCA processes. However, given CleanCap’s high yield and process efficiency, many customers who previously insourced these processes have begun to partner with us. Based on the Industry Analysis, we believe our products and services are more effective than those of our competitors. Deep scientific expertise, intellectual property protection and specialty equipment serve as barriers to entry in this space.
For our mRNA offerings, we compete with Aldevron, Patheon, eTheRNA, Lonza, Catalent, and Samsung Biologics, among others. Based on the Industry Analysis, we believe we have a reputation for our expertise in the RNA space with talented scientists who are constantly pushing the frontier of RNA science. This scientific expertise and the required high-cost equipment serve as barriers to entry. In addition to our expertise, we believe our GMP cleanroom manufacturing process differentiates us from competitors.
For custom oligonucleotides, we compete with a number of manufacturers. Custom oligonucleotide providers include those that provide complex, highly modified oligonucleotides and those that provide less complex offerings. In the custom oligonucleotide space, complexity is based on the length of the sequence and level of modification to the phosphate backbone. Large manufacturers like Integrated DNA Technologies, Thermo Fisher Scientific and EMD Millipore Corporation (“Millipore Sigma”) serve less complex customer needs while we, LGC Biosearch Technologies and GenScript Biotech Corporation serve more complex customer needs. In the custom oligonucleotide market, we have a reputation for accepting complex orders and delivering high purity products that reduce researcher re-work and save money. Quick turnaround times and the ability to produce at scale are essential requirements in this segment.
In the oligonucleotide synthesis inputs market, we compete against large distributor-manufacturers like Thermo Fisher and Millipore Sigma while also serving them as customers. Our Glen Research brand has a long history in this industry, which drives customer loyalty, and has a reputation for high-fidelity technical service, focusing on supplying and sourcing highly modified inputs for its customers.
For our specialty enzymes offering, we compete with New England Biolabs, Thermo Fisher, QIAGEN, and Roche, among others. We believe that Alphazyme is uniquely positioned in the market to address customers’ custom enzyme needs and has a reputation of being a flexible partner.
Biologics Safety Testing
For drugs in early development, we compete against other bioprocess impurity kit providers such as BioGenes (“BioGenes”) or Enzo Life Sciences (“Enzo”). Competitors generally offer fewer expression platforms (generally between one and three) compared to our offering of 24 expression platforms and over 100 different impurity detection kits. As a drug successfully moves forward to validation and approval stages, a customer may either continue with an off-the-shelf kit or they may begin the process to develop a custom assay that is tailored to meet their specific host cell and manufacturing process needs. During the entire drug development process, and especially during this decision, we are partners with the manufacturer and provide our expertise to help them make the best bioprocess quality control and testing-related decisions.
If a drug manufacturer continues with an off-the-shelf assay from development to validation and approval, they will generally stay with the incumbent kit provider due to the extensive validation they have conducted. For custom assay development, our main competitors are BioGenes, Rockland Immunochemicals and some CDMOs and CROs with custom assay development capabilities. The trend in recent years has been for CDMOs, CROs and large biopharmaceutical companies to focus on core competencies and outsource host cell protein assays or qualify off-the-shelf kits when possible.
Licenses and Collaborations
Broad Patent License Agreement
We (through TriLink BioTechnologies) entered into a Nonexclusive Patent License and Material Transfer Agreement with The Broad Institute, Inc. (“Broad”) effective as of July 5, 2017, and amended on September 29, 2017 (the “Broad Patent License Agreement”). Broad, together with a consortium of educational institutions (including Harvard University and the Massachusetts Institute of Technology), owns and controls certain patent rights relating to genome editing technology, including the CRISPR-Cas9 gene editing processes and have a licensing program for use and commercialization of technologies and products covered by the underlying patent rights. Under the Broad Patent License Agreement, Broad grants to us a non-exclusive, royalty-bearing, non-transferable and non-sublicensable, worldwide license under the licensed patent rights to manufacture and sell products and to perform certain in vitro processes or services on a fee-for-service basis, in each case, solely as research tools for research purposes (excluding human, clinical or diagnostic uses). We must use diligent efforts to develop products, introduce products into the commercial market and make products reasonably available to the public. We are obligated to pay a mid-five figure annual license maintenance fee and royalties in the range of 5% to 10% on net sales of covered products and processes.
The term of the Broad Patent License Agreement extends through the expiration of the last to expire claim of any of the licensed patents. We are entitled to terminate the Broad Patent License Agreement for convenience at any time on at least three (3) months written notice, in which case we must continue to pay license maintenance fees and royalties as noted above for the sale of products that are not covered by the specific claims of the licensed patent rights but are otherwise derived from such
licensed patent rights or from products covered by such licensed patent rights. Broad may terminate the license for our uncured failure to make payments, for our uncured material breach or if we bring a patent challenge against any of the institutional rights holders.
Manufacturing and Supply
We occupy facilities in San Diego, California, Leland, North Carolina, Sterling, Virginia, and Jupiter, Florida.
Our Wateridge facility in San Diego is engaged in the manufacture of reagents. The facility was designed and built by us in conjunction with the building owner to contain fully functional chemical and biological manufacturing operations from material receiving to product distribution and has its own loading dock, manufacturing gas delivery system, solvent delivery and waste system, ISO Class 8 and ISO Class 7 designated customer manufacturing suites and integrated building management systems for required site control.
In addition to the Wateridge facility, we have two facilities in San Diego, Flanders 1 and Flanders 2. Flanders 1 provides us with additional GMP manufacturing capacity and provides us the optionality downstream to manufacture materials beyond current quality requirements for mRNA raw materials, including CleanCap. Flanders 1 will also support an additional increase to batch run sizes and overall throughput. Flanders 2 was purpose built to support GMP-grade manufacturing and to support customers into Phase II clinical trials and beyond. Both the Flanders 1 and Flanders 2 facilities include the introduction of integrated manufacturing systems, quality of water improvements from Reverse Osmosis De-ionized grade water to WFI (“Water For Injection”), which is pharmaceutical grade water, and other facility infrastructure investments to support potential customer needs related to quality. We took occupancy of the Flanders facilities in 2023 and expect to begin manufacturing from both locations in 2024.
In early 2023, our Southport, North Carolina operations were relocated to a new state-of-the-art facility in Leland, North Carolina. This new facility more than doubles our operational square footage and has capacity to support future growth in the manufacture and processing of antibody and HCP ELISA kits. The operations include laboratory, manufacturing, kitting, cold storage, shipping and waste handling capabilities. The fully customized design includes a Mass Spectrometry Center of Excellence and specialized cell culture facilities. It significantly increases our manufacturing and development capacity while providing other R&D, laboratory and automation upgrades. Extensive process flow analysis has been incorporated into the facility design to optimize and enhance both our manufacturing and kit packaging operations.
Our Sterling, Virginia facility was designed to perform quality control, aliquoting, packaging and shipping and houses the appropriate space and systems.
Through our acquisition of Alphazyme in the first quarter of 2023, we acquired a purpose-built enzyme production facility in Jupiter, Florida, that can produce enzymes to kilogram quantities. The facility includes environmental controls such as HEPA filtration, pressure, temperature, and humidity monitoring, with vertical integration of all enzyme development, production, and testing operations. Our enzymes are produced under the controls of an ISO 13485:2016 compliant QMS.
Our supply chain is supported by a diverse network of specialized suppliers and transportation partners and undergoes regular evaluations to assess supplier quality and identify risks, including those associated with supply concentration. These proactive evaluations enable us to implement strategic measures to effectively manage and mitigate risks. By continuously optimizing our supply chain, we ensure operational resilience and maintain a steady supply of critical materials for our products.
Government Regulation
We provide products used for basic research or as raw materials used by biopharmaceutical customers for further processing, and active pharmaceutical ingredients used for preclinical and clinical studies. The quality of our products is critical to researchers looking to develop novel vaccines and therapies and for biopharmaceutical customers who use our products as raw materials or who are engaged in preclinical studies and clinical trials. Biopharmaceutical customers are subject to extensive regulations by the FDA and similar regulatory authorities in other countries for conducting clinical trials and commercializing products for therapeutic, vaccine or diagnostic use. This regulatory scrutiny results in our customers imposing rigorous quality requirements on us as their supplier through supplier qualification processes and customer contracts.
Our nucleic acid and biologics safety testing segments produce materials used in research and biopharmaceutical production, clinical trial vaccines and vaccine support products. We produce materials in support of our customers’ manufacturing businesses and to fulfill their validation requirements, as applicable. These customer activities are subject to regulation and consequently require these businesses to be inspected by the FDA and other national regulatory agencies under their respective cGMP regulations. These regulations result in our customers imposing quality requirements on us for the manufacture of our products, and maintain records of our manufacturing, testing and control activities. In addition, the specific activities of some of our businesses require us to hold specialized licenses for the manufacture, distribution and/or marketing of particular products.
All of our sites are subject to licensing and regulation, as appropriate under federal, state and local laws relating to:
•the surface and air transportation of chemicals, biological reagents and hazardous materials;
•the handling, use, storage and disposal of chemicals (including toxic substances), biological reagents and hazardous waste;
•the procurement, handling, use, storage and disposal of biological products for research purposes;
•the safety and health of employees and visitors to our facilities; and
•protection of the environment and general public.
Regulatory compliance programs at each of our businesses are managed by a dedicated group responsible for regulatory affairs and compliance, including the use of outside consultants. Our compliance programs are also managed by quality management systems, such as vendor supplier programs and training programs. Within each business, we have established Quality Management Systems (“QMS”) responsible for risk based internal audit programs to manage regulatory requirements and client quality expectations. Our QMS program ensures that management has proper oversight of regulatory compliance and quality assurance, inclusive of reviews of our system practices to ensure that appropriate quality controls are in place and that a robust audit strategy confirms requirements for compliance and quality assurance.
Research Products
Our products and operations may be subject to extensive and rigorous regulation by the FDA and other federal, state, or local authorities, as well as foreign regulatory authorities. The FDA regulates, among other things, the research, development, testing, manufacturing, clearance, approval, labeling, storage, recordkeeping, advertising, promotion, marketing, distribution, post-market monitoring and reporting, and import and export of pharmaceutical drugs. Certain of our products are currently marketed as research use only (“RUO”).
We believe that our products that are marketed as RUO products are exempt from compliance with GMP regulations under the FDCA. RUO products cannot make any claims related to safety, effectiveness or diagnostic utility and they cannot be intended for human clinical diagnostic use. In November 2013, the FDA issued a final guidance on products labeled RUO, which, among other things, reaffirmed that a company may not make any clinical or diagnostic claims about an RUO product. The FDA will also evaluate the totality of the circumstances to determine if the product is intended for diagnostic purposes. If the FDA were to determine, based on the totality of circumstances, that our products labeled and marketed for RUO are intended for diagnostic purposes, they would be considered medical products that will require clearance or approval prior to commercialization.
We do not make claims related to safety or effectiveness and they are not intended for diagnostic or clinical use. However, the quality of our products is critical to meeting customer needs, and we therefore voluntarily follow the quality standards outlined by the International Organization for Standardization for quality management systems (ISO 9001:2015) for the design, development, manufacture, and distribution of our products. Some biopharmaceutical customers desire extra requirements including quality parameters and product specifications, which are outlined in customer-specific quality agreements. These products are further processed and validated by customers for their applications. Customers qualify us as part of their quality system requirements, which can include a supplier questionnaire and on-site audits. Customers requalify us on a regular basis to ensure our quality system, processes and facilities continue to meet their needs and we are meeting requirements outlined in relevant customer agreements.
Active Pharmaceutical Ingredients (“APIs”) for Clinical Trials
We provide APIs to customers for use in preclinical studies through and including clinical trials. We hold a drug manufacturing license with the California Food and Drug Branch of the California Department of Public Health for manufacture of APIs for clinical use and are subject to inspection to maintain licensure. Manufacture of APIs for use in clinical trials is regulated under § 501(a)(2)(B) of the FDCA, but is not subject to the current GMP regulations in 21 CFR § 211 by operation of 21 CFR § 210. We follow the principles detailed in the International Council for Harmonisation (“ICH”) Q7, Good Manufacturing Practice Guide for Active Pharmaceutical Ingredients (Section 19, APIs For Use in Clinical Trials) in order to comply with the applicable requirements of the FDCA, and the comparable GMP principles for Europe; European Community, Part II, Basic Requirements for Active Substances Used as Starting Materials (Section 19, APIs For Use in Clinical Trials). APIs are provided to customers under customer contracts that outline quality standards and product specifications. As products advance through the clinical phases, requirements become more stringent, and we work with customers to define and agree on requirements and risks associated with their product.
Customers’ biopharmaceutical products early in their development have a high failure rate and often do not advance through the clinical stages to commercialization. Our customers are required to follow regulatory pathways that are not always known, which may cause additional unforeseen requirements placed on us as their contract manufacturer and delays in advancing to the next stage of product development. We also provide novel compounds for cell and gene therapy applications, which result in additional challenges for our customers attempting to obtain regulatory approval given that this field is relatively new, and regulations are evolving. Customer clinical trials rely on approval from institutional review boards (“IRBs”) and patient and volunteer enrollment, which makes timelines unpredictable for advancing to the next stage in product development. Preclinical studies and clinical trials conducted by our customers are also expensive and data may be negative or inconclusive causing customers to abandon projects that were expected to continue. Regulatory requirements in both the United States and abroad are always evolving and compliance with future laws may require significant investment to ensure compliance.
Other Regulatory Requirements
Environmental laws and regulations. We believe that our operations comply in all material respects with applicable laws and regulations concerning environmental protection. To date, there have been no material effects upon our earnings or competitive position resulting from our compliance with applicable laws or regulations enacted or adopted relating to the protection of the environment. Our capital and operating expenditures for pollution control in 2023 and 2022 were not material and are not expected to be material in 2024.
Intellectual Property
Our success depends in part on our ability to obtain and maintain intellectual property protection for our products and services, defend and enforce our intellectual property rights, preserve the confidentiality of our trade secrets, and operate without infringing, misappropriating or otherwise violating valid and enforceable intellectual property rights of others. We seek to protect the investments made into the development of our products and services by relying on a combination of patents, trademarks, copyrights, trade secrets, including know-how, and license agreements. We also seek to protect our proprietary products and services, in part, by requiring our employees, consultants, contractors and other third parties to execute confidentiality agreements and invention assignment agreements.
Patents. Our intellectual property strategy is focused on protecting through patents and other intellectual property rights our core products and services, including CleanCap, and related instrumentation and applications. In addition, we protect our ongoing research and development into critical reagents for cell and gene therapy through patents and other intellectual property rights. Our patent portfolio generally includes patents and patent applications relating to compositions and methods for the production of CleanCap, oligonucleotides, nucleic acids, immunofluorescence assays, and mock viral particles. We may own provisional patent applications, and provisional patent applications are not eligible to become issued patents until, among other things, we file national stage patent applications either directly or via the PCT within 12 or 30 to 32 months, respectively. If we do not timely file any national stage patent applications, we may lose our priority date with respect to our provisional patent applications and any patent protection on the inventions disclosed in such provisional patent applications. We cannot predict whether any such patent applications will result in the issuance of patents that provide us with any competitive advantage.
Issued patents extend for varying periods depending on the date of filing of the patent application or the date of patent issuance and the legal term of patents in the countries in which they are obtained. Generally, utility patents issued for applications are granted a term of 21 years from the earliest effective filing date of a non-provisional patent application. Issued patents may be extended beyond the natural 21 year term for regulatory or administrative delay in accordance with provisions of applicable local law. As a result, our patent portfolio may not provide us with sufficient rights to exclude others from commercializing products similar or identical to ours.
The following granted patents relate to our CleanCap products and technology.
| | | | | | | | | | | | | | | | | | | | |
| Jurisdiction | | Patent Number | | Title | | Expiration |
| United States | | 10494399 | | Compositions and methods for synthesizing 5′-Capped RNAs | | 2036 |
| United States | | 10519189 | | Compositions and methods for synthesizing 5′-Capped RNAs | | 2036 |
| United States | | 10913768C1 | | Compositions and methods for synthesizing 5′-Capped RNAs | | 2036 |
| United States | | 11414453 | | Compositions and methods for synthesizing 5′-Capped RNAs | | 2036 |
| United States | | 11878991 | | Compositions and methods for synthesizing 5′-Capped RNAs | | 2036 |
| United States | | 11578095 | | Compositions and methods for synthesizing 5′-Capped RNAs | | 2036 |
| Europe | | 3352584 | | Compositions and methods for synthesizing 5′-Capped RNAs | | 2036 |
| Europe | | 3954225 | | Compositions and methods for synthesizing 5′-Capped RNAs | | 2036 |
| Europe | | 3906789 | | Compositions and methods for synthesizing 5′-Capped RNAs | | 2036 |
| Europe | | 4104687 | | Compositions and methods for synthesizing 5′-Capped RNAs | | 2036 |
| Europe | | 4140491 | | Compositions and methods for synthesizing 5′-Capped RNAs | | 2036 |
| Australia | | 2016328645 | | Compositions and methods for synthesizing 5′-Capped RNAs | | 2036 |
| Australia | | 2021206780 | | Compositions and methods for synthesizing 5′-Capped RNAs | | 2036 |
Canada | | 2999274 | | Compositions and methods for synthesizing 5′-Capped RNAs | | 2036 |
China | | ZL 202310734863.0 | | Compositions and methods for synthesizing 5′-Capped RNAs | | 2036 |
Hong Kong | | 40080484 | | Compositions and methods for synthesizing 5′-Capped RNAs | | 2036 |
| Japan | | 6814997 | | Compositions and methods for synthesizing 5′-Capped RNAs | | 2036 |
| Japan | | 7082174 | | Compositions and methods for synthesizing 5′-Capped RNAs | | 2036 |
Korea, Republic of | | 10-2500198 | | Compositions and methods for synthesizing 5′-Capped RNAs | | 2036 |
The following patents relate to our MockV related products and technology.
| | | | | | | | | | | | | | | | | | | | |
| Jurisdiction | | Patent Number | | Title | | Expiration |
| United States | | 9632087 | | Methods for evaluating viral clearance from a biopharmaceutical solution employing mock viral particles | | 2034 |
| United States | | 10309963 | | Methods for evaluating viral clearance from a process solution employing mock viral particles | | 2034 |
| Europe | | 3044339 | | Methods and kits for quantifying the removal of mock virus particles from a purified solution | | 2034 |
| Australia | | 2014320015 | | Methods and kits for quantifying the removal of Mock Virus Particles from a purified solution | | 2034 |
| Australia | | 2021200484 | | Methods and kits for quantifying the removal of Mock Virus Particles from a purified solution | | 2034 |
| China | | 105899684 | | Methods and kits for quantifying pseudoviral particles removed from purified solution | | 2034 |
| Japan | | 6549126 | | Methods and kits for removal of mock virus particles from a purified solution | | 2034 |
United States | | 11754565 | | Methods and kits for removal of mock virus particles from a purified solution | | 2034 |
Trademarks. Our trademark portfolio is designed to protect the brands of our current and future products and includes U.S. trademark registrations for our company name, Maravai LifeSciences, subsidiary names Cygnus Technologies and TriLink Biotechnologies and various product names, such as CleanCap and MockV.
Trade Secrets. We also rely on trade secrets, including know-how, unpatented technology and other proprietary information, to strengthen our competitive position. We have determined that certain technologies, such as the production of antibodies for biologics safety testing, are better kept as trade secrets, rather than pursuing patent protection. To prevent disclosure of trade secrets to others, it is our policy to enter into nondisclosure, invention assignment and confidentiality agreements with parties who have access to trade secrets, such as our employees, collaborators, outside scientific collaborators, consultants, advisors
and other third parties. These agreements also provide that all inventions resulting from work performed for us or relating to our business and conceived or completed during the period of employment or assignment, as applicable, are our exclusive property. In addition, we take other appropriate precautions, such as physical and technological security measures, to guard against misappropriation of our proprietary information by third parties.
We intend to pursue additional intellectual property protection to the extent we believe it would advance our business objectives. Notwithstanding these efforts, there can be no assurance that we will adequately protect our intellectual property or provide any competitive advantage. We cannot provide any assurance that any patents will be issued from our pending or any future patent applications or that any issued patents will adequately protect our products or technology. Our intellectual property rights may be invalidated, held unenforceable, circumvented, narrowed or challenged. In addition, the laws of various foreign countries where our products are distributed may not protect our intellectual property rights to the same extent as laws in the United States. Furthermore, it may be difficult to protect our trade secrets. While we have confidence in the measures we take to protect and preserve our trade secrets, they may be inadequate and can be breached, and we may not have adequate remedies for violations of such measures. In addition, our trade secrets may otherwise become known or be independently discovered by competitors. Moreover, our invention assignment agreements with employees, collaborators, outside scientific collaborators, consultants, advisors and other third parties may not be self-executing or otherwise provide meaningful protection for our intellectual property rights. If we do not adequately protect our intellectual property, third parties, including our competitors, may be able to use our technologies to produce and market products that compete with us and erode our competitive advantage. For more information regarding risks related to intellectual property, please see Item 1A. “Risk Factors—Risks Related to our Intellectual Property.”
Human Capital Management: Empowering Our Future
At Maravai, we understand that our strength lies in the expertise and commitment of our people.
As of December 31, 2023, our team had over 650 full-time employees. Following the reduction in force, which was completed on January 5, 2024, we had approximately 570 full-time employees. Our workforce represents a diversity of backgrounds, with 44% identifying as female, 56% as male, and 52% as ethnically or racially diverse as of December 31, 2023. We take pride in the fact that, as of December 31, 2023, 24% of our team held advanced degrees, underscoring our emphasis on science and innovation.
Our compensation and comprehensive benefits packages are designed to attract and retain the talent we need to be competitive in the markets we serve. Believing in the value of ownership, we extend equity awards to all full-time employees through our 2020 Omnibus Incentive Plan, alongside opportunities to participate in our 2020 Employee Stock Purchase Plan. Our commitment to excellence ensures every employee receives thorough on-the-job training. We also understand that great people managers are the key to enabling and unlocking the potential of our employees. In 2023, we initiated a bi-monthly “Leading Together” people leader series with all levels of our people leaders to ensure they had the critical knowledge, perspective, and tools to develop their people and align their teams towards company goals and objectives.
We actively work to foster direct and open lines of communication between all levels of staff through our all-employee engagement survey, our quarterly all-employee town halls, management skip level meetings, and an emphasis on our core values (Connected, Open, Driven, and Empowered). Our 2023 company-wide engagement survey reached a participation rate of 97% and all levels of leadership engage in action planning based on the results.
In 2023, all of our sites participated in process excellence initiatives and we launched “Find a Better Way Day” to highlight the efforts of our employees to drive quality and safety improvements and reduce cost.
As a leading life sciences company, we are committed to the health, safety and well-being of our employees. All employees that could be exposed to potential hazards are required to complete annual health and safety training, including laboratory chemical safety, hazard communication and hazardous waste management trainings. In 2023, we upgraded our electronic Safety Data Sheet (SDS) system to more readily provide chemical safety data electronically to employees and customers.
Available Information
Our website is located at www.maravai.com, and our investor relations website is located at investors.maravai.com. Our Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q, and Current Reports on Form 8-K, and any amendments to these reports, are available through our investor relations website, free of charge, as soon as reasonably practicable after we electronically file or furnish them with the SEC. Our filings with the SEC are also available, free of charge, on the SEC's website at www.sec.gov. We webcast via our investor relations website our earnings calls and certain events we participate in or host with members of the investment community. Our investor relations website also provides notifications of news or announcements regarding our financial performance and other items that may be material or of interest to our investors,
including SEC filings, investor events, press and earnings releases, and blogs. The contents of our website are not incorporated by reference into this Annual Report on Form 10-K or in any other report or document we file with the SEC, and any references to our website are intended to be inactive textual references only.
Item 1A. Risk Factors
In addition to the other information in this report and our other filings with the SEC, you should carefully consider the risks and uncertainties described below, which could materially and adversely affect our business operations, financial condition and results of operations. The risks and uncertainties described below are not the only ones we face. Additional risks and uncertainties that we are unaware of, or that we currently believe are not material, may also become important factors that affect us. If any of the following risks occur, our business, financial condition, results of operations and prospects could be materially and adversely affected. In that event, the price of our Class A common stock could decline, and you could lose all or part of your investment.
Summary of Risk Factors
The following is a summary of the material risks we and/or our shareholders face in the normal course of our business operations. The list below is not exhaustive, and is qualified in its entirety by reference to the full risk factor discussion that follows this summary.
Risks Related to Our Business and Strategy
•The level of our customers’ spending on and demand for outsourced nucleic acid production and biologics safety testing products and services.
•Uncertainty regarding the extent and duration of our revenue associated with COVID-19-related products and services and the dependency of such revenue, in important respects, on factors outside our control.
•The impact of ongoing macroeconomic challenges and changes in economic conditions, including adverse developments affecting banks and financial institutions, follow-on effects of those events and related systemic pressures, on our and our customers’ current and future business operations.
•The effects of our recent reduction in force, including on our ability to attract and/or retain qualified key personnel.
•Use of our products by customers in the production of vaccines and therapies, some of which represent relatively new and still-developing modes of treatment, and the impact of unforeseen adverse events, negative clinical outcomes, development of alternative therapies, or increased regulatory scrutiny of these modes of treatment and their financial cost on our customers’ use of our products and services.
•Competition with life science, pharmaceutical and biotechnology companies who are substantially larger than us and potentially capable of developing new approaches that could make our products, services and technology obsolete.
•The potential failure of our products and services to not perform as expected and the reliability of the technology on which our products and services are based.
•The risk that our products do not comply with required quality standards.
•Market acceptance of our life science reagents.
•Significant fluctuations and unpredictability in our quarterly and annual operating results, which make our future operating results difficult to predict and could cause our operating results to fall below expectations or any guidance we may provide.
•Our ability to implement our strategic plan successfully.
•Natural disasters, geopolitical instability (including the ongoing military conflicts in Ukraine and the Gaza Strip) and other catastrophic events.
•Risks related to our acquisitions, including whether we achieve the anticipated benefits of acquisitions of businesses or technologies.
•Product liability lawsuits.
•Our dependency on a limited number of customers for a high percentage of our revenue and our ability to maintain our current relationships with such customers.
•Our reliance on a limited number of suppliers or, in some cases, sole suppliers, for some of our raw materials and the risk that we may not be able to find replacements or immediately transition to alternative suppliers.
•The risk that our products become subject to more onerous regulation by the FDA or other regulatory agencies in the future.
Risks Related to Our Intellectual Property and Technology
•Our ability to obtain, maintain and enforce sufficient intellectual property protection for our current or future products.
•The risk that a future cyber-attack or security breach cannot be prevented.
•Our ability to protect the confidentiality of our proprietary information
•The risk that one of our products may be alleged (or found) to infringe on the intellectual property rights of third parties.
•Compliance with our obligations under intellectual property license agreements.
•Our or our licensors’ failure to maintain the patents or patent applications in-licensed from a third party.
•Our ability to adequately protect our intellectual property and proprietary rights throughout the world.
Risks Related to Our Indebtedness
•Our existing level of indebtedness and our ability to raise additional capital on favorable terms.
•Our ability to generate sufficient cash flow to service all of our indebtedness.
•Our potential failure to meet our debt service obligations.
•Restrictions on our current and future operations under the terms applicable to the Credit Agreement.
Risks Related to Our Organizational Structure
•Our dependence, by virtue of our principal asset being our interest in Maravai Topco Holdings, LLC (“Topco LLC”), on distributions from Topco LLC to pay our taxes and expenses, including payments under a tax receivable agreement with the former owners of Topco LLC (the “Tax Receivable Agreement” or “TRA”) together with various limitations and restrictions that impact Topco LLC’s ability to make such distributions.
•The risk that conflicts of interest could arise between our shareholders and Maravai Life Sciences Holdings, LLC (“MLSH 1”), the only other member of Topco LLC, and impede business decisions that could benefit our shareholders.
•The substantial future cash payments we may be required to make under the Tax Receivable Agreement to MLSH 1 and Maravai Life Sciences Holdings 2, LLC (“MLSH 2”), an entity through which certain of our former owners hold their interests in the Company and the negative effect of such payments.
•The fact that our organizational structure, including the TRA, confers certain benefits upon MLSH 1 and MLSH 2 that will not benefit our other common shareholders to the same extent as they will benefit MLSH 1 and MLSH 2.
•Our ability to realize all or a portion of the tax benefits that are expected to result from the tax attributes covered by the Tax Receivable Agreement.
•The possibility that we will receive distributions from Topco LLC significantly in excess of our tax liabilities and obligations to make to make payments under the Tax Receivable Agreement.
•Unanticipated changes in effective tax rates or adverse outcomes resulting from examination of our income or other tax returns.
Risks Related to Being a Public Company
•Risks related to our annual assessment of the effectiveness of our internal control over financial reporting, including the potential existence of any material weakness or significant deficiency.
Risks Related to Our Class A Common Stock
•The fact that investment entities affiliated with GTCR, LLC (“GTCR”) currently control a majority of the voting power of our outstanding common stock, and it may have interests that conflict with ours or yours in the future.
•Risks related to our “controlled company” status within the meaning of the corporate governance standards of NASDAQ.
•The potential anti-takeover effects of certain provisions in our corporate organizational documents.
•Potential sales of a significant portion of our outstanding shares of Class A common stock.
•Potential preferred stock issuance and the anti-takeover impacts of any such issuances.
Risks Related to Our Business and Strategy
We are dependent on the level of our customers’ spending on and demand for outsourced nucleic acid production and biologics safety testing products and services. A reduction in spending or change in spending priorities of our customers could significantly reduce demand for our products and services and could have a material adverse effect on our business, financial condition, results of operations, cash flows and prospects.
The success of our business depends primarily on the number and size of contracts with our customers, primarily pharmaceutical and biotechnology companies, for our products and services. For example, during the COVID-19 pandemic we benefited from a significant increase in demand for our products and service, including our proprietary CleanCap® analogs that are used by our customers in the production of COVID-19 vaccines, and also benefited during 2021 and 2022, more generally, from the overall growth of the global biologics market, higher research and development budgets of our customers and a greater degree of outsourcing by our customers. The level of our customers’ spending on and demand for our products and services is also subject to, among other things, their own financial performance, changes in their available resources, the timing of their commercial manufacturing initiatives, their decisions to acquire in-house manufacturing capacity (rather than outsource), their spending priorities, including research and development budgets, and their budgetary policies and practices, which, in turn, are dependent upon a number of factors outside of our control.
Our customers determine their research and development budgets based on several factors, including their need to develop new biological products, their competitors’ discoveries, developments and commercial manufacturing initiatives and the anticipated market, clinical and reimbursement scenarios for specific products and therapeutic areas. In addition, consolidation in the industries in which our customers operate may have an impact on our customers’ spending as they integrate acquired operations, including research and development departments and associated budgets.
Access to capital is critical to many of our customers’ ability to fund research and development, particularly early-stage biotechnology and pharmaceutical companies, and historically, these companies have funded their research and development activities by raising capital privately or in the equity markets. Declines and uncertainties in the capital markets, including as a result of ongoing negative macroeconomic challenges, rising interest rates, recent instability in the banking sector, and volatile credit markets, have limited access to capital and negatively affected companies’ ability to fund research and development efforts. While 2021 and 2022 saw a significant level of investment in venture- and private equity-backed startup companies, funding for companies at all stages, and particularly early- and late-stage companies, contracted considerably during 2023, which, together with broader economic uncertainty, led certain of our customers to implement more stringent budgetary policies designed to conserve capital, which in turn, caused a reduction in research and development spending and a decline in further purchases of our products and services. We have no assurance as to whether, or when, such research and development spending may stabilize or increase, if at all. Further, if the funding of venture- and private equity-backed biotechnology and pharmaceutical companies remains weak or weakens further, the research and development budgets of our customers may be further reduced or eliminated altogether, which could impact future demand for our products and services.
If our customers reduce their spending on our products and services as a result of any of these or other factors, our business, financial condition, results of operations, cash flows and prospects would be materially and adversely affected.
Moreover, we have no control over the timing and volume of purchases by our customers, and as a result, our operating results may fluctuate significantly, and our future revenue and operating results can be difficult to forecast. Our inability to forecast fluctuations in demand could harm our business, financial position and future results of operations. See also “—Our operating results may fluctuate significantly in the future, which makes our future operating results difficult to predict and could cause our operating results to fall below expectations or any guidance we may provide” below.
The extent and duration of our revenue associated with COVID-19 related products and services are uncertain and are dependent, in important respects, on factors outside our control.
Certain of our products, including our proprietary CleanCap® analogs, are used by our customers in the production of COVID-19 vaccines. During each of the years ended December 31, 2022, 2021 and 2020, our results of operations and cash flows were significantly and positively impacted by a strong demand for our proprietary CleanCap® analogs and highly modified RNA products, particularly mRNA. However, as a result of the general decrease in market demand for COVID-19 related products and services, including the supply and manufacture of COVID-19 vaccines, and in particular, following the end of U.S. federal public health emergency declaration and World Health Organization declaration of the end of the pandemic in early May 2022, we experienced substantial declines in COVID-19 related revenue during the year ended December 31, 2023. For the years ended December 31, 2023, 2022 and 2021, we estimate that revenue from COVID-19 related products and services represented approximately 21.0%, 67.9% and 69.7%, respectively, of our total revenues. We expect to experience
further declines in COVID-19 related revenue for the aforementioned reasons, as well as a result of unused inventory of our products that our customers have on hand, which are not indication-specific. We are currently unable to fully estimate the impact of this unused inventory on our future revenues, nor are we able to predict when or if our customers will resume purchasing COVID-19 related products. Our longer-term revenue prospects for COVID-19 related products are highly uncertain but are expected to be substantially less than pandemic highs. Additionally, the ongoing manufacture and supply of COVID-19 vaccines (including bivalent booster doses) by our customers is uncertain and subject to various political, social, economic, and regulatory factors that are outside of our control, including the emergence, duration and intensity of new virus variants; regional resurgences of the virus globally; the availability and administration of pediatric and booster vaccinations, vaccine supply constraints, vaccine hesitancy and the effectiveness of vaccines against new virus strains; competition faced by our customers from other COVID-19 vaccine manufacturers and the development and availability of antiviral therapeutic alternatives; the lapsing of the public health emergency declaration made pursuant to Section 319 of the Public Health Service Act in January 2020 with respect to the COVID-19 pandemic; political and social debate relating to the need for, efficacy of, or side effects related to one or more specific COVID-19 vaccines; and the U.S. economy and global economy, including impacts resulting from supply chain constraints, labor market shortages and inflationary pressures. As the supply and manufacture of COVID-19 vaccines by our customers slows, or becomes no longer necessary, including if COVID-19 vaccines by our customers’ competitors are determined or perceived to be more effective, we expect that demand for our COVID-19 related products and services will significantly decrease, which would have a material adverse effect on our revenue, results of operations and financial condition.
Ongoing macroeconomic challenges and changes in economic conditions, including adverse developments affecting banks and financial institutions, follow-on effects of those events and related systemic pressures, could negatively impact, directly or indirectly, our and our customers’ current and future business operations and our financial condition, revenue and earnings.
Our reagents are sold primarily to biopharmaceutical and academic organizations developing novel vaccines and therapies and performing basic research. Research and development spending by our customers and the availability of government research funding can fluctuate due to changes in available resources, mergers of pharmaceutical and biotechnology companies, spending priorities, general economic conditions and institutional and governmental budgetary policies. Our biologics safety testing customers are biopharmaceutical companies, contract research organizations (“CROs”), contract development and manufacturing organizations (“CDMOs”) and life science companies, which largely serve the biopharmaceutical industry. Our nucleic acid production customers are largely vaccine and therapeutic drug makers or diagnostics manufacturers, which rely in part on government healthcare-related policies and funding. As a result, changes in government funding for certain research, decreases in or the imposition of limits on government spending more generally (including as a result of the ongoing appropriations process for the US. federal government’s fiscal year 2024), or reductions in overall healthcare spending could negatively impact us or our customers and, correspondingly, our sales to them. In particular, if the U.S. Congress fails to pass appropriate appropriations measures or enact another continuing resolution, reimbursements we are eligible to receive under the Cooperative Agreement we entered into with the U.S. Department of Defense may be jeopardized, which would negatively affect our business, operations and financial condition.
Currently, the U.S. and global economies are experiencing ongoing macroeconomic challenges, including labor shortages, supply chain disruptions and persistent inflation, which have led to increasing interest rates, volatility in the capital and credit markets, and fiscal and monetary policy uncertainty. Our business operations, as well as our customers’ and suppliers’ business operations, have been impacted, and are expected to continue to be impacted, by these negative conditions. In particular, labor shortages and wage inflation have affected our ability to hire, develop and retain our talented and diverse workforce, to maintain performance levels (especially cost and schedule), and to maintain our corporate culture. Further, if our raw material and other laboratory material suppliers experience operational challenges as a result of labor shortages, limited material availability, logistics delays and transportation capacity constraints, or are unable to access adequate capital to support their working capital requirements, they may be unable to provide raw materials or other laboratory materials to us in a timely manner or at a reasonable cost, which could adversely affect our profit margins and results of operations.
Additionally, demand for our products and services could be adversely impacted if these ongoing macroeconomic challenges cause customers to reduce their operating budgets, adversely impact our customers’ ability to commit funds to purchase our products, or otherwise cause customers to delay, cancel, decrease or forego purchases of our products and services. Further, since the majority of our customers’ contracts can be terminated, delayed or reduced in scope upon short notice or no notice, this may require us to carry excess inventory to manage through unevenness in order activity and lead to unanticipated fluctuations in our quarterly revenue and earnings. If we are not able to forecast and adequately manage through changes in our customers’ order requirements, our productivity, profitability, results of operations, cash flows and financial position could be negatively impacted. Further deterioration or a protracted extension of these negative macroeconomic conditions, a potential economic downturn or recession, or a significant reduction or delay in governmental funding as a result of U.S. federal budget
issues, or the perception that any of these events may occur, could cause a decline in demand for our products and services and adversely affect our performance and result in declines in our revenue and earnings.
Our recent reduction in force may have unintended consequences, including business disruption, cause us to experience difficulties attracting and/or retaining qualified key personnel, which could negatively impact our ability to develop and market our products and services and our overall performance.
On November 7, 2023, we announced a workforce reduction of approximately 15% of our total full-time workforce (the “Reduction in Force”). The Reduction in Force resulted in the elimination of 102 full-time positions. Although the majority of the positions eliminated were intended to address excess manufacturing capacity, relative to current demand, this reduction has resulted in certain reallocations of employee duties. As a result, inefficiencies related to task unfamiliarity, heavier workloads, loss of knowledge and unfilled gaps may arise, especially if we are unable to effectively manage and implement the transition of impacted employees’ duties and responsibilities. Any such inefficiencies may cause disruption or delay in our business activities. The Reduction in Force and resulting job reassignments could also negatively affect employee morale and make it more difficult to motivate and retain our remaining personnel.
In addition, our future success depends largely upon the continued service of our management and scientific staff and our ability to attract, retain and motivate highly skilled technical, scientific, management and marketing personnel, who deliver high-quality and timely services to our customers and keep pace with cutting-edge technologies and developments in biologics. We face significant competition in the hiring and retention of such personnel from other companies, other providers of outsourced biologics services, research and academic institutions, government and other organizations who have superior funding and resources and who may use these resources to pursue personnel more aggressively than we are. Additionally, certain highly skilled personnel that we seek to employ may be subject to non-competition or other restrictive covenants restricting their ability to work for us or within certain aspects of our business for a period of time. Although some jurisdictions (including the State of California) prohibit non-competition agreements as a matter of law, and the U.S. Federal Trade Commission has issued a notice of proposed rulemaking that would prohibit employers in the U.S. from using non-compete agreements, if we hire certain employees from competitors or other companies, those former employers may attempt to assert that these employees and/or we have breached certain legal obligations, resulting in a diversion of our time and resources.
We have, from time to time, experienced, and we expect to continue to experience, difficulty in hiring and retaining employees with appropriate qualifications. In recent years, recruiting, hiring and retaining employees with expertise in our industry and in the geographies where we operate has become increasingly difficult as the demand for skilled professionals has increased and as a result of labor shortages believed to have resulted from actions taken during the onset of the COVID-19 pandemic, but which remained following the recovery and which we expect will continue beyond the near-term. These difficulties may be heightened as a result of the Reduction in Force. The loss of key personnel or our inability to hire and retain skilled personnel could materially adversely affect the development of our products and services and our business, financial condition, results of operations, cash flows and prospects.
Certain of our products are used by customers in the production of vaccines and therapies, some of which represent relatively new and still-developing modes of treatment. Unforeseen adverse events, negative clinical outcomes, or increased regulatory scrutiny of these and their financial cost may damage public perception of the safety, utility, or efficacy of these vaccines and therapies or other modes of treatment and may harm our customers’ ability to conduct their business. Such events may negatively impact our revenue and have an adverse effect on our performance.
Gene therapy and nucleic acid vaccines remain relatively new and are under active development, with only a few gene therapies and nucleic acid vaccines, including those for COVID-19, approved to date by regulatory authorities. Public perception may be influenced by claims that gene therapy or nucleic acid vaccines are unsafe or ineffective, and gene therapy may not gain the acceptance of the public or the medical community. Following the release of nucleic acid COVID-19 vaccines, including those that incorporate our CleanCap® products, segments of the population have criticized their safety and efficacy impacting vaccine demand. In addition, ethical, social, legal and financial concerns about gene therapy and nucleic acid vaccines, including COVID-19 vaccines, could result in additional regulations or limitations or even prohibitions on certain gene therapies or vaccine-related products. Our customers’ use of our products and services in therapeutic and vaccine development programs for other (non-COVID-19-related) indications could be impacted by more restrictive regulations or negative public perception, which could negatively affect our business prospects, revenue and results of operation.
We compete with life science, pharmaceutical and biotechnology companies who are substantially larger than we are and potentially capable of developing new approaches that could make our products, services and technology obsolete.
The market for pharmaceutical, reagent, therapeutic and diagnostic products and services is intensely competitive, rapidly evolving, significantly affected by new product introductions and other market activities by industry participants and subject to rapid technological change. We also expect increased competition as additional companies enter our market and as more
advanced technologies become available. We compete with other providers of outsourced biologics products and services. We also compete with the in-house discovery, development and commercial manufacturing functions of pharmaceutical and biotechnology companies. Many of our competitors are large, well-capitalized companies with significantly greater resources and market share than we have. As a consequence, these competitors are able to spend more aggressively on product and service development, marketing, sales and other initiatives than we can. Many of these competitors also have:
•broader name recognition;
•longer operating histories and the benefits derived from greater economies of scale;
•larger and more established distribution networks;
•additional product and service lines and the ability to bundle products and services to offer higher discounts or other incentives to gain a competitive advantage;
•more experience in conducting research and development, manufacturing and marketing;
•more experience in entering into collaborations or other strategic partnership arrangements; and
•more financial, manufacturing and human resources to support product development, sales and marketing and patent and other intellectual property litigation.
These factors, among others, may enable our competitors to market their products and services at lower prices or on terms more advantageous to customers than we can offer. Competition may result in price reductions, reduced gross margins and loss of market share, any of which could have a material adverse effect on our business, financial condition, results of operations, cash flows and prospects. Additionally, our current and future competitors, including certain of our customers, may at any time develop additional products and services that compete with our products and services and new approaches by these competitors may make our products, services, technologies and methodologies obsolete or noncompetitive. We may not be able to compete effectively against these organizations.
In addition, to develop and market our new products, services, technologies and methodologies successfully, we must accurately assess and meet customers’ needs, make significant capital expenditures, optimize our development and manufacturing processes to predict and control costs, hire, train and retain the necessary personnel, increase customer awareness and acceptance of our services, provide high-quality services in a timely manner, price our products and services competitively and effectively integrate customer feedback into our business planning. If we fail to create demand for our new products, services or technologies, our future business could be harmed.
If our products and services do not perform as expected or the reliability of the technology on which our products and services are based is questioned, we could experience lost revenue, delayed or reduced market acceptance of our products and services, increased costs and damage to our reputation.
Our success depends on the market’s confidence that we can provide reliable, high-quality life science reagents. We believe that customers in our target markets are likely to be particularly sensitive to product defects and errors. Our reputation and the public image of our products, services and technologies may be impaired if our products or services fail to perform as expected.
Although our products are tested prior to shipment, defects or errors could nonetheless occur. Our operating results depend on our ability to execute and, when necessary, improve our quality management strategy and systems and our ability to effectively train and maintain our employee base with respect to quality management. A failure of our quality control systems could result in problems with facility operations or preparation or provision of products. In each case, such problems could arise for a variety of reasons, including equipment malfunction, failure to follow specific protocols and procedures, problems with raw materials or environmental factors and damage to, or loss of, manufacturing operations. Such problems could affect production of a particular batch or series of batches of products, requiring the destruction of such products or a halt of facility production altogether. Furthermore, some of the products that we manufacture are subsequently incorporated into products that are sold by other life sciences companies and we have no control over the manufacture and production of those products.
In addition, in the event we, or our suppliers, fail to meet required quality standards and if our products experience, or are perceived to experience, a material defect or error, our products could be recalled or we may be unable to timely deliver products to our customers, which in turn could damage our reputation for quality and service. In the past, certain of our custom mRNA and CleanCap® reagent products have been sold with insufficient capping efficiency or with incorrect transcription instructions. Additionally, several lots of our host cell protein (“HCP”) enzyme-linked immunosorbent assay (“ELISA”) biologics safety testing kits have experienced a possible instability drift and decrease in accuracy. Although we have taken steps to improve our quality review, product documentation and reference testing procedures, we cannot guarantee that we will not experience quality assurance issues with our products in the future. Any such failure could, among other things, lead to increased costs, delayed or lost revenue, delayed market acceptance, damaged reputation, diversion of development resources,
legal claims, reimbursement to customers for lost drug product, starting materials and active pharmaceutical ingredients, other customer claims, damage to and possibly termination of existing customer relationships, increased insurance costs, time and expense spent investigating the cause and, depending on the cause, similar losses with respect to other batches or products, any of which could harm our business, financial condition, results of operations, cash flows and prospects. Such defects or errors could also narrow the scope of the use of our products, which could hinder our success in the market.
Even after any underlying concerns or problems are resolved, any lingering concerns in our target markets regarding our technology or any manufacturing defects or performance errors in our products or services could continue to result in lost revenue, delayed market acceptance, damage to our reputation and claims against us.
In addition, we may be unable to maintain the quality, reliability, robustness and expected turnaround times of our products and services to continue to satisfy customer demand as we grow. To effectively manage our growth, we must continue to improve our operational, manufacturing and quality control systems and processes and other aspects of our business and continue to effectively expand, train and manage our personnel. The time and resources required to improve our existing systems and procedures, implement new systems and procedures and to adequately staff such existing and new systems and procedures is uncertain, and failure to complete this in a timely and efficient manner could adversely affect our operations and negatively impact our business and financial results. We may need to purchase additional equipment, some of which can take several months or more to procure, set up and validate, establish new production processes and increase our personnel levels to meet increased demand. There can be no assurance that any of these increases in scale, personnel expansion or equipment or process enhancements will be successfully implemented, or that we will have adequate space, including in our laboratory and production facilities, to accommodate such required expansion. Failure to manage this growth or transition could result in delays in turnaround times, higher product costs, declining product quality, deteriorating customer service and slower responses to competitive challenges. A failure in any one of these areas could make it difficult for us to meet market expectations for our products and services and could damage our reputation and our business, financial condition, results of operations, cash flows and prospects could be adversely affected.
Our products are highly complex and are subject to quality control requirements.
Whether a product is produced by us or purchased from outside suppliers, it is subject to quality control procedures, including the verification of stability and performance and, for certain products, additional validation required by certain GMP that we voluntarily follow, European Conformity (“CE”) marking and ISO 9001:2015 compliance, prior to final packaging. Certain of our products are manufactured following the voluntary GMP quality standards of the International Council for Harmonisation’s GMP Guide, comparable GMP principles for the European Union and customer-specific requirements. We believe these products are exempt from compliance with the Food, Drug, and Cosmetic Act (“FDCA”) and the current GMP (“cGMP”) regulations of the Food and Drug Administration (“FDA”), as our products are further processed and incorporated into final drug products by our customers and we do not make claims related to their safety or effectiveness. In the event we, or our suppliers, produce products that fail to comply with required quality standards, we may incur delays in fulfilling orders, write-downs, damages resulting from product liability claims and harm to our reputation.
Our operating results may fluctuate significantly in the future, which makes our future operating results difficult to predict and could cause our operating results to fall below expectations or any guidance we may provide.
We have no control over the timing and volume of purchases by our customers. As a result,our quarterly and annual operating results may fluctuate significantly, which makes it difficult for us to predict our revenues and future operating results. These fluctuations may be driven by a variety of factors, many of which are outside of our control, including, but not limited to:
•unused inventory of our products that our customers have on hand, which are not indication-specific, and our lack of insight as to the amount of unused inventory of our products that such customers have on hand;
•changes in the level of our customers’ spending on and demand for our products and services, including as a result of, among other things, their own financial performance, changes in their available resources, timing of their commercial manufacturing initiatives, their decision to acquire in-house manufacturing capacity (rather than outsource), their spending priorities, including research and development budgets, and their budgetary policies and practices;
•our ability to increase penetration in our existing markets and expand into new markets;
•our customers accelerating, canceling, reducing or delaying orders as a result of developments related to their pre-clinical studies and clinical trials;
•the relative reliability and robustness of our products and services;
•changes in governmental regulations or the regulatory posture toward our business;
•the volume and mix of the products and services we sell;
•changes in the production or sales costs related to our products and services;
•the ongoing success of our newer products, such as our CleanCap® and mRNA products;
•the rate of introduction of other new products or product enhancements by us or others in our industry;
•the timing and amount of expenditures that we may incur to acquire, develop or commercialize additional products, services and technologies or for other purposes, such as the expansion of our facilities;
•changes in governmental and academic funding of life sciences research and developments or changes that impact budgets, budget cycles or seasonal spending patterns of our customers;
•future accounting pronouncements or changes in our accounting policies;
•difficulties encountered by our commercial carriers in delivering our products, whether as a result of external factors such as weather or negative macroeconomic conditions or internal issues such as labor disputes;
•the timing and magnitude of any adjustments to the Tax Receivable Agreement liability;
•changes in the assessment of the realizability of our deferred tax assets;
•general market conditions and other factors outside of our control, such as natural disasters, geopolitical unrest, war, terrorism, public health issues or other catastrophic events; and
•the other factors described in this “Risk Factors” section.
The impact of any one of the factors discussed above, or the cumulative effects of a combination of such factors, could result in significant fluctuations and unpredictability in our quarterly and annual operating results. As a result, comparisons of our operating results on a period-to-period basis may not be meaningful. Investors should not rely on our past results as an indication of our future performance.
As a result of variability and unpredictability, we may also fail to meet the expectations of industry or financial analysts or investors for any period. If our revenue or operating results fall short of the expectations of analysts or investors or any guidance we may provide, or if the guidance we provide falls short of the expectations of analysts or investors, the price of our Class A common stock could decline substantially. Such a stock price decline could occur even when we have met or exceeded any previously publicly stated guidance we may have provided.
If we are unable to manufacture in specific quantities, our operating results will be harmed.
Our revenue and other operating results depend in large part on our ability to manufacture and ship our products in sufficient quantities. Any interruptions we experience in the manufacturing or shipping of our products could delay our ability to recognize revenue in a particular quarter. Manufacturing problems can and do arise, and as demand for our products increases, any such problems could have an increasingly significant impact on our operating results. While we have not generally experienced problems with, or delays in, our production capabilities that resulted in delays in our ability to ship finished products, there can be no assurance that we will not encounter such problems in the future. We may not be able to quickly ship products and recognize anticipated revenue for a given period if we experience significant delays in the manufacturing process. In addition, we must maintain sufficient production capacity in order to meet anticipated customer demand, and we may be unable to offset the associated fixed costs if orders slow, which would adversely affect our operating margins. If we are unable to manufacture and ship our products consistently, in sufficient quantities and on a timely basis, our revenue, cash flow, gross margins and our other results of operations will be materially and adversely affected.
Natural disasters, geopolitical unrest, war, terrorism, public health issues or other catastrophic events could disrupt the supply, delivery or demand of products and services, as well as our sites, which could negatively affect our operations and performance.
We are subject to the risk of disruption by earthquakes, hurricanes, floods and other natural disasters, fire, power shortages, geopolitical unrest, war (including any escalation of the ongoing military conflicts in Ukraine or the Gaza Strip), terrorist attacks and other hostile acts, public health issues, epidemics or pandemics and other events beyond our control and the control of the third parties on which we depend. Any of these catastrophic events, whether in the United States or abroad, may have a significant negative impact on the global economy, our employees, facilities, partners, suppliers, distributors or customers, and could decrease demand for our products and services, create delays and inefficiencies in our supply chain and make it difficult or impossible for us to deliver products and services to our customers.
We rely upon our internal manufacturing, packaging and distribution operations to produce many of the products we sell and our warehouse facilities to store products pending sale. Any significant disruption of those operations for any reason, such as
labor disputes or social unrest, power interruptions, fire, hurricanes, a public health crisis (such as a pandemic), earthquakes or other events beyond our control, could adversely affect our sales and customer relationships and therefore adversely affect our business and results of operations. We have significant operations in California, near major earthquake faults, which make us susceptible to earthquake risk.
In addition, a catastrophic event that results in damage to specific equipment that would be difficult to replace, the destruction or disruption of our research and production facilities or our critical business or information technology systems would severely affect our ability to conduct normal business operations and, as a result, our operating results would be adversely affected.
Strategic transactions or acquisitions may require us to seek additional financing, which we may not be able to secure on favorable terms, if at all.
We plan to continue a strategy of growth and development for our business. To this end, we actively evaluate various strategic transactions on an ongoing basis, including licensing or acquiring complementary products, technologies or businesses that would complement our existing portfolio of products and services. In order to complete such strategic transactions, we may need to seek additional financing to fund these investments and acquisitions. Should we need to do so, we may not be able to secure such financing, or obtain such financing on favorable terms, for reasons including rising interest rates and continued volatility and uncertainty in the U.S. and global capital and credit markets. Our credit agreement also contains a number of restrictive covenants that impose significant restrictions on our ability to make acquisitions or certain other investments, as well as to incur additional indebtedness to finance such acquisitions or other investments. In addition, future acquisitions may require the issuance or sale of additional equity, or equity-linked securities, which may result in additional dilution to our shareholders.
Our commercial success depends on the market acceptance of our life science reagents. Our reagents may not achieve or maintain significant commercial market acceptance.
Our commercial success is dependent upon our ability to continue to successfully market and sell our life science reagents. Our ability to achieve and maintain commercial market acceptance of our products and services and provide customers access to our life science reagents will depend on a number of factors, including:
•our ability to increase awareness of the capabilities of our technology and solutions;
•our customers’ willingness to adopt new products, services and technologies;
•whether our products and services reliably provide advantages over legacy and other alternative technologies and are perceived by customers to be cost effective;
•our ability to execute on our strategy to scale-up our CleanCap® technology to meet increasing demand and provide channels to access our CleanCap® technology and life science reagents;
•the rate of adoption of our products and services by biopharmaceutical companies, academic institutions and others;
•the relative reliability and robustness of our products and services as a whole and the components of our life science offerings, including, for example, CleanCap® and our assays for detecting host cell proteins;
•our ability to develop new tools and solutions for customers;
•whether competitors develop and commercialize products and services that provide comparable features and benefits at scale;
•the impact of our investments in product innovation and commercial growth;
•negative publicity regarding our or our competitors’ products resulting from defects or errors; and
•our ability to further validate our technology through research and accompanying publications.
We cannot assure you that we will be successful in addressing these criteria or other criteria that might affect the market acceptance of our products and services. If we are unsuccessful in achieving and maintaining market acceptance of our products and services, our business, financial condition, results of operations, cash flows and prospects could be adversely affected.
The market may not be receptive to our new products and services upon their introduction.
We expect a portion of our future revenue growth to come from introducing new products, including plasmid DNA and GMP-grade mRNA. The commercial success of all of our products and services will depend upon their acceptance by the life science and biopharmaceutical industries. Some of the products and services that we are developing are based upon new technologies or approaches. As a result, there can be no assurance that these new products and services, even if successfully developed and
introduced, will be accepted by customers. If customers do not adopt our new products, services and technologies, our results of operations may suffer and, as a result, the market price of our Class A common stock may decline.
It may be difficult for us to implement our strategies for revenue growth in light of competitive challenges.
We face significant competition across many of our product lines. In addition, consolidation trends in the pharmaceutical, biotechnology and diagnostics industries have served to create fewer customer accounts and to concentrate purchasing decisions for some customers, resulting in increased pricing pressure on us. Moreover, customers may believe that larger companies are better able to compete as sole source vendors, and therefore prefer to purchase from such businesses. Failure to anticipate and respond to competitors’ actions may impact our future revenue and profitability.
Our estimates of market opportunity and forecasts of market growth may prove to be inaccurate, and even if the market in which we compete achieves the forecasted growth, our business could fail to grow at similar rates, if at all.
Addressable market estimates and growth forecasts are subject to significant uncertainty and are based on assumptions and estimates that may not prove to be accurate. These estimates and forecasts are based on a number of complex assumptions and third-party estimates and other business data, including assumptions and estimates relating to our ability to generate revenue from existing products and services and the development of new products and services. Our estimates and forecasts relating to the size and expected growth of our markets may prove to be inaccurate. Even if the markets in which we compete meet our size estimates and growth forecasts, our business could fail to grow at the rate we anticipate, if at all.
If we are unable to successfully implement our strategic plan on a timely basis or at all, our business and future result of operations may be adversely impacted.
Our strategic plan was developed based upon market and technology trends that we currently believe present revenue growth opportunities, and in turn, long-term shareholder value creation. Our strategic plan includes a series of strategic priorities and cost realignment initiatives designed to drive growth and improve operational efficiency. Our ability to achieve our strategic initiatives is subject to a number of risks, including those discussed herein under the heading “Risks Related to Our Business and Strategy,” as well as challenges we face with executing multiple initiatives simultaneously. For example, our commercial initiatives may not succeed, or we may lose market share due to challenges in choosing the right products to develop or the right customers to target for these products, or integrating products of acquired companies into our sales and marketing strategy. We cannot assure you that we will overcome the risks associated with our strategic initiatives. If we fail to manage or overcome those risks, we may not realize the intended benefits of our strategic plan and may incur additional expenses without related revenue growth. Our business, financial position and results of operations will be adversely affected if we fail to successfully implement our strategic initiatives or if we invest resources in a growth strategy that ultimately proves to be unsuccessful.
Product liability lawsuits against us could cause us to incur substantial liabilities, limit sales of our existing products and limit commercialization of any products that we may develop.
Our business exposes us to the risk of product liability claims that are inherent in the development, production, distribution, and sale of biotechnology products. We face an inherent risk of product liability exposure related to the use of certain of our products in our customers’ human clinical trials and product liability lawsuits may allege that our products or services identified inaccurate or incomplete information or otherwise failed to perform as designed. We may also be subject to liability for errors in, a misunderstanding of or inappropriate reliance upon, the information we provide in the ordinary course of our business activities. If any of our products harm people due to our negligence, willful misconduct, unlawful activities or material breach, or if we cannot successfully defend ourselves against claims that our products caused injuries, we could incur substantial liabilities. Regardless of merit or eventual outcome, liability claims may result in the following, any of which could impact our business, financial condition, results of operations, cash flows and prospects:
•decreased demand for our products and any products that we may develop;
•injury to our reputation;
•costs to defend the related litigation;
•loss of revenue; and
•the inability to commercialize products that we may develop.
We maintain product liability insurance, but this insurance is subject to deductibles, limits and exclusions and may not fully protect us from the financial impact of defending against product liability claims or the potential loss of revenue that may result.
Any product liability claim brought against us, with or without merit, could increase our insurance rates or prevent us from securing insurance coverage in the future.
We may be unable to efficiently manage growth as a larger and more geographically diverse organization.
Our strategic acquisitions, the continued expansion of our commercial sales operations and our organic growth have increased the scope and complexity of our business. As a result, we will face challenges inherent in efficiently managing a more complex business with an increased number of employees over large geographic distances, including the need to implement appropriate systems, policies, benefits and compliance programs. Our inability to manage successfully the geographically more diverse and substantially larger combined organization could materially adversely affect our operating results.
Opportunistic acquisitions may pose risks and challenges that could adversely affect our business, and we may not achieve the anticipated benefits of acquisitions of businesses or technologies.
We have made in the past, and may make in the future, selected opportunistic acquisitions of complementary businesses, products, services or technologies. In January 2022, we acquired MyChem LLC, a provider of proprietary, ultra-pure nucleotides to customers in the diagnostics, pharma, genomics and research markets to complement our nucleic acid business and in January 2023, we completed the acquisition of Alphazyme, LLC, an original equipment manufacturer provider of custom molecular biology enzymes, servicing customers in the genetic analysis and nucleic acid synthesis markets to complement our nucleic acid production business. However, we may be unable to continue to identify or complete promising acquisitions for many reasons, including competition among buyers, the high valuations of businesses in our industry, the need for regulatory and other approvals and the availability of capital, particularly during a period of disruption and volatility within the global capital and credit markets.
Any acquisition involves numerous risks, uncertainties and operational, financial, and managerial challenges, including the following, any of which could adversely affect our business, financial condition, results of operations, cash flows and prospects:
•difficulties in integrating new operations, systems, technologies, products, services and personnel of acquired businesses effectively and in a timely manner;
•difficulties in implementing and maintaining controls, procedures and policies with respect to our financial accounting systems, including disclosure controls and procedures and internal control over financial reporting, at acquired businesses that, prior to the acquisition, had lacked such controls, procedures and policies;
•lack of synergies or the inability to realize expected synergies and cost-savings, including enhanced revenue, technology, human resources, cost savings, operating efficiencies and other synergies;
•difficulties in obtaining and verifying the financial statements and other business information of acquired businesses;
•difficulties in managing geographically dispersed operations, including risks associated with entering new or foreign markets in which we have no or limited prior experience;
•underperformance of any acquired technology, product, or business relative to our expectations and the price we paid;
•negative near-term impacts on financial results after an acquisition, including acquisition-related earnings charges;
•the potential loss of key employees, customers, contractual relationships, and strategic partners of acquired companies;
•declining employee morale and retention issues affecting employees of businesses that we acquire, which may result from changes in compensation, or changes in management, reporting relationships, future prospects or the direction of the acquired business;
•claims by terminated employees and shareholders of acquired companies or other third parties related to the transaction;
•the assumption or incurrence of historical liabilities, obligations and expenses of the acquired business, including unforeseen and contingent or similar liabilities that are difficult to identify or accurately quantify, or other litigation-related liabilities and regulatory actions;
•the assumption or incurrence of additional debt obligations or expenses, or use of substantial portions of our cash;
•the issuance of equity or equity-linked securities to finance or as consideration for any acquisitions that dilute the ownership of our shareholders;
•the issuance of equity securities to finance or as consideration for any acquisitions may not be an option if the price of our Class A common stock is low or volatile which could preclude us from completing any such acquisitions;
•the assumption of certain collaboration, strategic alliance and licensing arrangement may require us to relinquish valuable rights to our technologies or products, or grant licenses on terms that are not favorable to us;
•disruption of our ongoing operations, diversion of management’s attention and company resources from existing operations of the business, and the dedication of significant efforts and expense across all operational areas, including sales and marketing, research and development, manufacturing, finance, legal and information technologies;
•the impairment of intangible assets as a result of technological advancements, or worse-than-expected performance of acquired companies;
•the need to later divest acquired assets at a loss if an acquisition does not meet our expectations;
•risks associated with acquiring intellectual property, including potential disputes regarding acquired companies’ intellectual property; and
•difficulties relating to operating with increased leverage and incurring additional interest expense as a result of financing acquisitions with additional indebtedness, which could make us more vulnerable to downturns.
There can be no assurance we will identify promising acquisition opportunities. Even if we do, there can be no assurance that any of the acquisitions we have made, or that we may make, will be successful or will be, or will remain, profitable. Our failure to successfully address the foregoing risks may prevent us from achieving the anticipated benefits from any past or future acquisition in a reasonable time frame, or at all.
Our ability to use net operating loss and tax credit carryforwards and certain built-in losses to reduce future tax payments is limited by provisions of the Internal Revenue Code, and it is possible that changes in laws or certain transactions or a combination of certain transactions may result in material additional limitations on our ability to use our net operating loss and tax credit carryforwards.
Sections 382 and 383 of the Internal Revenue Code of 1986, as amended, contain rules that limit the ability of a company that undergoes an ownership change, which is generally any change in ownership of more than 50% of its stock over a three-year period, to utilize its net operating loss and tax credit carryforwards and certain built-in losses recognized in years after the ownership change. These rules generally operate by focusing on ownership changes involving stockholders owning directly or indirectly 5% or more of the stock of a company and any change in ownership arising from a new issuance of stock by the company. Generally, if an ownership change occurs, the yearly taxable income limitation on the use of net operating loss and tax credit carryforwards and certain built-in losses is equal to the product of the applicable long-term, tax-exempt rate and the value of the company’s stock immediately before the ownership change. As a result, following any such ownership change, we might be unable to offset our taxable income with losses, or our tax liability with credits, before such losses and credits expire, in which event we could incur larger federal and state income tax liabilities than we would have had we not experienced an ownership change. In addition, under the 2017 Tax Cuts and Jobs Act (“TCJA”), tax losses generated in taxable years beginning after December 31, 2017, may be utilized to offset no more than 80% of taxable income annually. On March 27, 2020, the Coronavirus Aid Relief, and Economic Security Act (“CARES Act”) was signed into law and changed certain provisions of the TCJA. Under the CARES Act, NOLs arising in taxable years beginning after December 31, 2017 and before January 1, 2021 may be carried back to each of the five taxable years preceding the tax year of such loss, but NOLs arising in taxable years beginning after December 31, 2020 may not be carried back. In addition, the CARES Act eliminates the limitation on the deduction of NOLs to 80% of current year taxable income for taxable years beginning before January 1, 2021, but the 80% limitation applies to tax years beginning after December 31, 2020. As such, we may not be able to realize a tax benefit from the use of our NOLs.
We may be required to record a significant charge to earnings if our goodwill and other amortizable intangible assets, or other investments become impaired.
We are required under U.S. generally accepted accounting principles (“GAAP”) to test goodwill for impairment at least annually and to review our goodwill, amortizable intangible assets and other assets acquired through merger and acquisition activity for impairment when events or changes in circumstance indicate the carrying value may not be recoverable. Factors that could lead to impairment of goodwill, amortizable intangible assets and other assets acquired via acquisitions include significant adverse changes in the business climate and actual or projected operating results (affecting our company as a whole or affecting any particular segment) and declines in the financial condition of our business. We may be required in the future to record additional charges to earnings if our goodwill, amortizable intangible assets or other investments become impaired. Any such charge would adversely impact our consolidated financial results.
Changes in accounting principles and guidance could result in unfavorable accounting charges or effects.
We prepare our consolidated financial statements in accordance with GAAP. These principles are subject to interpretation by the SEC and various bodies formed to create and interpret appropriate accounting principles and guidance. A change in these principles or guidance, or in their interpretations, may have a material effect on our reported results, as well as our processes and related controls, and may retroactively affect previously reported results.
Our revenue recognition and other factors may impact our financial results in any given period and make them difficult to predict.
We recognize revenue when our performance obligations have been satisfied in an amount that reflects the consideration that we expect to receive in exchange for those performance obligations. Our revenue includes revenue from the sale of manufactured products, including products that can be purchased out of a catalog and custom manufactured products, and services, including custom antibody and assay development contracts, antibody affinity extraction and stability and feasibility studies, as well as certain licensing and royalty arrangements. The majority of our contracts include only one performance obligation, namely the delivery of products, both custom and catalog, and services. We also recognize revenue from other contracts that may include a combination of products and services, the provision of solely services, or from license fee arrangements which may be associated with the delivery of product. Our application of the revenue recognition accounting guidance with respect to the nature of future contractual arrangements could impact the forecasting of our revenue for future periods, as both the mix of products and services we will sell in a given period, as well as the size of contracts, is difficult to predict.
Furthermore, the presentation of our financial results requires us to make estimates and assumptions that may affect revenue recognition. In some instances, we could reasonably use different estimates and assumptions, and changes in estimates may occur from period to period. See Part II, Item 7, “Management’s Discussion and Analysis of Financial Condition and Results of Operations—Critical Accounting Estimates—Revenue Recognition.”
Given the foregoing factors, comparing our revenue and operating results on a period-to-period basis may not be meaningful, and our past results may not be indicative of our future performance.
Fluctuations in our effective tax rate may adversely affect our results of operations and cash flows.
We are subject to a variety of tax liabilities, including federal, state, foreign and other taxes such as income, sales/use, payroll, withholding, and ad valorem taxes. Changes in tax laws or their interpretations could decrease our net income, the value of any tax loss carryforwards, the value of tax credits recorded on our balance sheet and our cash flows, and accordingly could have a material adverse effect on our business, financial condition, results of operations, cash flows and prospects. In addition, our tax liabilities are subject to periodic audits by the relevant taxing authority, which could increase our tax liabilities.
Our business is subject to a number of environmental risks.
Our manufacturing business involves the controlled use of hazardous materials and chemicals and is therefore subject to numerous environmental and safety laws and regulations and to periodic inspections for possible violations of these laws and regulations. The costs of compliance with environmental and safety laws and regulations are significant. Any violations, even if inadvertent or accidental, of current or future environmental and safety laws or regulations and the cost of compliance with any resulting order or fine could adversely affect our operations.
Risks Related to Our Reliance on Third Parties
We depend on a limited number of customers for a high percentage of our revenue. If we cannot maintain our current relationships with customers, fail to sustain recurring sources of revenue with our existing customers, or if we fail to enter into new relationships, our future operating results will be adversely affected.
Revenue from our largest customers were 19.3%, 61.2% and 68.1% of total revenue for the years ended December 31, 2023, 2022 and 2021, respectively. The revenue attributable to our top customers has fluctuated in the past and may fluctuate in the future, which could have a material adverse effect on our business, financial condition, results of operations, cash flows and prospects. In addition, the termination of these relationships, including following any failure to renew a long-term contract, could result in a temporary or permanent loss of revenue. See also “—The extent and duration of our revenue associated with COVID-19 related products and services are uncertain and are dependent, in important respects, on factors outside our control.”
Our future success depends on our ability to maintain these relationships, to increase our penetration among these existing customers and to establish new relationships. We engage in conversations with other companies and institutions regarding
potential commercial opportunities on an ongoing basis, which can be time consuming. There is no assurance that any of these conversations will result in a commercial agreement, or if an agreement is reached, that the resulting relationship will be successful. Speculation in the industry about our existing or potential commercial relationships can be a catalyst for adverse speculation about us, our products, our services and our technology, which can adversely affect our reputation and our business. In addition, if our customers order our products or services, but fail to pay on time or at all, our liquidity, financial condition, results of operations, cash flows and prospects could be materially and adversely affected.
We cannot assure investors that we will be able to further penetrate our existing markets or that our products or services will gain adequate market acceptance. Any failure to increase penetration in our existing markets would adversely affect our ability to improve our operating results.
We rely on distribution arrangements to market and sell our products and services, including in certain international markets, and our failure to maintain and successfully manage these arrangements or to renew or identify and implement additional arrangements on favorable terms, if at all, may impair our ability to effectively distribute and market our products and adversely impact our revenues and future results of operations.
We rely on certain distributors in order to market and sell our products and services in in certain international markets, particularly our biologics safety testing products and services in China. Our distributor in China accounted for 4.8% of our total revenues in the year ended December 31, 2023. If we are unable to maintain this distributor or enter into a similar arrangement with another distributor, or our current or future distributors do not perform adequately, our revenues and results of operations would likely be adversely impacted, at least temporarily. Additionally, changes in the inventory levels of our products owned and held by our distributors can result in significant variability in our revenues. Furthermore, our revenues from such distributors could be negatively impacted by macroeconomic conditions specific to the geographic markets in which our products and services are marketed and sold, geopolitical risks and other risks described below under “We are subject to financial, operating, legal and compliance risks associated with global operations.”
We may pursue additional arrangements regarding the sales and marketing and distribution of one or more of our products and services, including if we intend to grow our business internationally in certain geographic markets, and the success of our strategic initiatives and our future revenue growth may depend, in part, on our ability to enter into and maintain arrangements with other companies having sales, marketing and distribution capabilities and the ability of such companies to successfully market and sell any such products and services. Any failure to enter into such arrangements and marketing alliances on favorable terms, if at all, could delay or impair our ability to distribute or market our products and services and could increase our costs of distribution and marketing.
Our use of distribution arrangements and marketing alliances to commercialize our products and services subject us to a number of risks, including the following:
•we may be required to relinquish important rights to our products;
•we may not be able to control the amount and timing of resources that our distributors or collaborators may devote to the distribution or marketing of our products;
•our distributors or collaborators may experience financial difficulties; and
•business combinations or significant changes in a collaborator’s business strategy may adversely affect a collaborator’s willingness or ability to complete its obligations under any arrangement.
We rely on a limited number of suppliers or, in some cases, sole suppliers, for some of our raw materials and may not be able to find replacements or immediately transition to alternative suppliers.
Certain of our raw materials are sourced from a limited number of suppliers and some materials, including a proprietary DNA reagent, certain packaging materials, specific cell lines for Cygnus Technologies’ operations and certain raw materials used in our nucleic acid production products, as well as those raw materials sold under the Glen Research brand, are sole sourced. Delays or difficulties in securing these raw materials or other laboratory materials could result in an interruption in our production operations if we cannot obtain an acceptable substitute. Since the onset of the COVID-19 pandemic, global supply chains have faced challenges, including material availability, global logistics delays and constraints arising from, among other things, the transportation capacity of ocean shipping containers, and these challenges have been exacerbated by the ongoing macroeconomic conditions as discussed above. Any interruption of our supply chain could significantly affect our business, financial condition, results of operations, cash flows and prospects. While we may identify other suppliers, raw materials furnished by such replacement suppliers may require us to alter our production operations or perform extensive validations, which may be time consuming and expensive. There can be no assurance that we will be able to secure alternative materials and revalidate them without experiencing interruptions in our workflow. If we should encounter delays or difficulties in obtaining raw materials, our business, financial condition, results of operations, cash flows and prospects could be adversely affected.
We depend on a stable and adequate supply of quality raw materials from our suppliers, and price increases or interruptions of such supply could have an adverse impact on our business, financial condition, results of operations, cash flows and prospects.
Our operations depend upon our ability to obtain raw materials at reasonable prices. Cost and wage inflation, ongoing supply disruptions and logistics capacity constraints have increased, or may increase, our costs to manufacture and distribute our products and services. If we are unable to obtain the materials we need at a reasonable price due to inflationary pressures or other factors, we may not be able to produce certain of our products at marketable prices or at all, which could have a material adverse effect on our results of operations.
Although we believe that we have stable relationships with our existing suppliers, we cannot assure you that we will be able to secure a stable supply of raw materials going forward. Our suppliers may not be able to keep up with our pace of growth or may reduce or cease their supply of raw materials to us at any time. In addition, we cannot assure you that our suppliers have obtained and will be able to obtain or maintain all licenses, permits and approvals necessary for their operations or comply with all applicable laws and regulations, and failure to do so by them may lead to interruption in their business operations, which in turn may result in shortages of raw materials supplied to us. Some of our suppliers are based overseas and therefore may need to maintain export or import licenses. If the supply of raw materials is interrupted, due to ongoing supply chain disruptions or other factors, our business, financial condition, results of operations, cash flows and prospects may be adversely affected.
Because we rely heavily on third-party package-delivery services, a significant disruption in these services, damages or losses sustained during shipping or significant increases in prices could adversely affect our business, financial condition, results of operations, cash flows and prospects.
We ship a significant portion of our products to our customers through independent package delivery companies, such as World Courier, FedEx, UPS and DHL. If one or more of these third-party package-delivery providers were to experience a major work stoppage, preventing our products from being delivered in a timely fashion or causing us to incur additional shipping costs we could not pass on to our customers, our costs could increase and our relationships with certain of our customers could be adversely affected. In addition, if one or more of these third-party package-delivery providers were to increase prices, and we were not able to find comparable alternatives or make adjustments in our delivery network, our profitability could be adversely affected. Furthermore, if one or more of these third-party package-delivery providers were to experience performance problems or other difficulties, it could negatively impact our operating results and our customers’ experience. In the past, some of our products have sustained serious damage in transit such that they were no longer usable. Although we have taken steps to improve our packaging and shipping containers, there is no guarantee our products will not become damaged or lost in transit in the future. If our products are damaged or lost in transit, it may result in a substantial delay in the fulfillment of our customer’s order and, depending on the type and extent of the damage, it may result in a substantial financial loss. If our products are not delivered in a timely fashion or are damaged or lost during the delivery process, our customers could become dissatisfied and cease using our products or our services, which would adversely affect our business, financial condition, results of operations, cash flows and prospects.
Risks Related to Laws and Regulations
Our products could become subject to more onerous regulation by the FDA or other regulatory agencies in the future, which could increase our costs and delay or prevent commercialization of our products, thereby materially and adversely affecting our business, financial condition, results of operations, cash flows and prospects.
We make certain of our products available to customers as research-use-only (“RUO”) products. RUO products are regulated by the FDA as medical devices, and include in vitro diagnostic products in the laboratory research phase of development that are being shipped or delivered for an investigation that is not subject to the FDA’s investigational device exemption requirements. Although medical devices are subject to stringent FDA oversight, products that are intended for RUO and are labeled as RUO are exempt from compliance with most FDA requirements, including premarket clearance or approval, manufacturing requirements, and others. A product labeled RUO but which is actually intended for clinical diagnostic use may be viewed by the FDA as adulterated and misbranded under the FDCA, and subject to FDA enforcement action. The FDA has indicated that when determining the intended use of a product labeled RUO, the FDA will consider the totality of the circumstances surrounding distribution and use of the product, including how the product is marketed and to whom. The FDA could disagree with our assessment that our products are properly marketed as RUO, or could conclude that products labeled as RUO are actually intended for clinical diagnostic use, and could take enforcement action against us, including requiring us to stop distribution of our products until we are in compliance with applicable regulations, which would reduce our revenue, increase our costs and adversely affect our business, prospects, results of operations and financial condition. In the event that the FDA requires us to obtain marketing authorization of our RUO products in the future, there can be no assurance that the FDA will grant any clearance or approval requested by us in a timely manner, or at all.
Our raw material products are manufactured following the voluntary quality standards of ISO 9001:2015. Our GMP-grade raw material products follow ISO 9001:2015 standards, additional voluntary GMP quality standards and customer specific requirements. We believe these raw material products, including our GMP-grade raw material products, are exempt from compliance with the FDCA and the cGMP regulations of the FDA, as our products are further processed by our customers and we do not make claims related to their safety or effectiveness. We provide API products to customers for use in preclinical studies through and including clinical trials. Our API products are manufactured following the principles detailed in the International Council for Harmonisation (ICH) Q7, Good Manufacturing Practice Guide for Active Pharmaceutical Ingredients (Section 19, APIs For Use in Clinical Trials) in order to comply with the applicable requirements of the FDCA, and the comparable GMP principles for Europe; European Community, Part II, Basic Requirements for Active Substances Used as Starting Materials (Section 19, APIs For Use in Clinical Trials). Manufacture of APIs for use in clinical trials is regulated under § 501(a)(2)(B) of the FDCA, but is not subject to the current GMP regulations in 21 CFR § 211 by operation of 21 CFR § 210. Our API products are provided to customers under customer contracts that outline quality standards and product specifications. As products advance through the clinical phases, requirements become more stringent and we work with customers to define and agree on requirements and risks associated with their product.
The FDA could disagree with our assessment that our products are exempt from current GMP regulations. In addition, the FDA could conclude that the raw material and API products we provide to our customers are actually subject to the pharmaceutical or drug quality-related regulations for manufacturing, processing, packing or holding of drugs or finished pharmaceuticals, and could take enforcement action against us, including requiring us to stop distribution of our products until we are in compliance with applicable regulations, which would reduce our revenue, increase our costs and adversely affect our business, prospects, results of operations and financial condition. In the event that the FDA requires us to comply with FDA regulations, for our raw material and API products in the future, including the FDA’s current GMP regulations, there can be no assurance that the FDA will find our operations are in compliance in a timely manner, or at all.
We are subject to stringent privacy laws, information security laws, regulations, policies and contractual obligations related to data privacy and security and changes in such laws, regulations, policies and contractual obligations could adversely affect our business, financial condition, results of operations, cash flows and prospects.
We are subject to data privacy and protection laws and regulations that apply to the collection, transmission, storage and use of proprietary information and personally identifiable information (“PII”), which among other things, imposes certain requirements relating to the privacy, security and transmission of certain individually identifiable information.
Numerous other federal and state laws, including state security breach notification laws, state health information privacy laws and federal and state consumer protection laws, govern the collection, use, disclosure and security of personal information. These laws continue to change and evolve and are increasing in breadth and impact. Failure to comply with any of these laws and regulations could result in enforcement action against us, including fines, imprisonment of company officials and public censure, claims for damages by affected individuals, damage to our reputation and loss of goodwill, any of which could have a material adverse effect on our business, financial condition, results of operations, cash flows and prospects. Additionally, if we are unable to properly protect the privacy and security of personal information, we could be found to have breached our contracts.
Many states in which we operate have laws that protect the privacy and security of personal information. For example, the California Consumer Privacy Act of 2018 (“CCPA”), which increases privacy rights for California residents and imposes obligations on companies that process their personal information, came into effect on January 1, 2020. Among other things, the CCPA requires covered companies to provide new disclosures to California consumers and provide such consumers new data protection and privacy rights, including the ability to opt-out of certain sales of personal information. The CCPA provides for civil penalties for violations, as well as a private right of action for certain data breaches that result in the loss of personal information. Further, the California Privacy Rights Act (the “CPRA”), which took effect on January 1, 2023 (with certain provisions having retroactive effect to January 1, 2022), amended the CCPA. Amongst other things, the CPRA and eliminated the “employee exemption” under the CCPA, makes a distinction between “personal information” and “sensitive personal information,” imposing heightened protections for “sensitive personal information,” and brings business-to-business transactions under its purview. These laws and others like it are yet to be tested and may subject us to increased regulatory scrutiny, litigation, and overall risk. Further, there is discussion in Congress of a new federal data protection and privacy law to which we would become subject, if it is enacted.
Various foreign countries in which we operate also have, or are developing, laws that govern the collection, use, disclosure, security and cross-border transmission of personal information. For example, in the European Union (the “EU”) and the United Kingdom, the collection and use of personal data is governed by the provisions of the General Data Protection Regulation (“GDPR”), in addition to other applicable laws and regulations. The GDPR came into effect in May 2018,and has resulted in, and will continue to result in, significantly greater compliance burdens and costs for companies like us. Any data security breach could require notifications to the data subject and/or owners under U.S. federal, U.S. state, and/or international data
breach notification laws and regulations. Other jurisdictions outside the EU are similarly introducing or enhancing privacy and data security laws, rules and regulations, which could increase our compliance costs and the risks associated with noncompliance. We cannot guarantee that we are, or will be, in compliance with all applicable international regulations as they are enforced now or as they evolve.
It is possible that these laws may be interpreted and applied in a manner that is inconsistent with our practices and our efforts to comply with the evolving data protection rules may be unsuccessful. We must devote significant resources to understanding and complying with this changing landscape. Failure to comply with federal, state and international laws regarding privacy and security of personal information could expose us to penalties under such laws, orders requiring that we change our practices, claims for damages or other liabilities, regulatory investigations and enforcement action, litigation and significant costs for remediation, any of which could adversely affect our business. Even if we are not determined to have violated these laws, government investigations into these issues typically require the expenditure of significant resources and generate negative publicity, which have a material adverse effect on our business, financial condition, results of operations, cash flows and prospects.
We are subject to export and import control laws and regulations that could impair our ability to compete in international markets or subject us to liability if we violate such laws and regulations.
We are subject to U.S. export controls and sanctions regulations that restrict the shipment or provision of certain products and services to certain countries, governments and persons. While we take precautions to prevent our products and services from being exported in violation of these laws, we cannot guarantee that the precautions we take will prevent violations of export control and sanctions laws. If we are found to be in violation of U.S. sanctions or export control laws, it could result in substantial fines and penalties for us and for the individuals working for us. We may also be adversely affected through other penalties, reputational harm, loss of access to certain markets, or otherwise. Complying with export control and sanctions regulations may be time consuming and may result in the delay or loss of sales opportunities or impose other costs. Any change in export or import regulations, economic sanctions or related legislation, or change in the countries, governments, persons or technologies targeted by such regulations, could result in our decreased ability to export or sell certain products and services to existing or potential customers in affected jurisdictions.
Changes in political, economic or governmental regulations may reduce demand for our products and services or increase our expenses.
We compete in many markets in which we and our customers must comply with federal, state, local and international regulations, such as environmental, health and safety and food and drug regulations. We develop, configure and market our products and services to meet customer needs created by those regulations. The U.S. and international healthcare industry is subject to changing political, economic and regulatory influences that could significantly affect the drug development process, research and development costs and the pricing and reimbursement for pharmaceutical products, and also may increase the likelihood of legislative or regulatory changes that could impact us or our business operations. Any significant change in regulations could have an adverse effect on both our customers’ business and our business, which could result in reduced demand for our products and services or increases in our expenses. For example, we provide products and services used for basic research, raw materials used by biopharmaceutical customers for further processing, and active pharmaceutical ingredients used for preclinical studies and clinical trials.
Changes in the FDA’s regulation of the drug discovery and development process may have a negative impact on the ability of our customers to conduct and fund clinical trials, which could have a material adverse effect on the demand for the products and services we provide these customers. Additionally, the U.S. government and governments worldwide have increased efforts to expand healthcare coverage while at the same time curtailing and better controlling the increasing costs of healthcare. If cost-containment efforts limit our customers’ profitability, they may decrease research and development spending, which could decrease the demand for our products and services and materially adversely affect our growth prospects. Any of these factors could harm our customers’ businesses, which, in turn, could materially adversely hurt our business, financial condition, results of operations, cash flows and prospects.
We are subject to financial, operating, legal and compliance risk associated with global operations.
We engage in business globally, with approximately 51%, 62% and 60% of our revenue for the years ended December 31, 2023, 2022 and 2021, respectively, coming from outside the U.S. In addition, one of our strategies is to expand geographically, both through distribution and through direct sales. This subjects us to a number of risks, including international economic, political, and labor conditions; currency fluctuations; tax laws (including U.S. taxes on income earned by foreign subsidiaries); increased financial accounting and reporting burdens and complexities; unexpected changes in, or impositions of, legislative or regulatory requirements; failure of laws to protect intellectual property rights adequately; inadequate local infrastructure and difficulties in managing and staffing international operations; delays resulting from difficulty in obtaining export licenses for
certain technology; tariffs, quotas and other trade barriers and restrictions; transportation delays; operating in locations with a higher incidence of corruption and fraudulent business practices; and other factors beyond our control, including terrorism, war, natural disasters, climate change and diseases.
The application of laws and regulations implicating global transactions is often unclear and may at times conflict. Compliance with these laws and regulations may involve significant costs or require changes in our business practices that result in reduced revenue and profitability. Non-compliance could also result in fines, damages, criminal sanctions, prohibited business conduct, and damage to our reputation. We incur additional legal compliance costs associated with our global operations and could become subject to legal penalties in foreign countries if we do not comply with local laws and regulations, which may be substantially different from those in the U.S.
We may expand our operations in countries with developing economies, where it may be common to engage in business practices that are prohibited by anti-corruption and anti-bribery laws and regulations that apply to us, such as the U.S. Foreign Corrupt Practices Act (“FCPA”), the U.S. Travel Act, and the UK Bribery Act 2010, which prohibit improper payments or offers of payment to foreign governments and political parties by us for the purpose of obtaining or retaining business. Although we implement policies and procedures designed to ensure compliance with these laws, there can be no assurance that all of our employees, contractors, distributors and agents, including those based in foreign countries where practices which violate such U.S. laws may be customary, will comply with our internal policies. Any such non-compliance, even if prohibited by our internal policies, could have an adverse effect on our business and result in significant fines or penalties.
Our activities are and will continue to be subject to extensive government regulation, which is expensive and time consuming.
We are subject to various local, state, federal, foreign and transnational laws and regulations, and, in the future, any changes to such laws and regulations could adversely affect us.
We provide products and services used for basic research, raw materials and life science reagents used by biopharmaceutical customers for further processing, assays for biologics safety testing and active pharmaceutical ingredients used for preclinical studies and clinical trials. The quality of our products and services is critical to researchers looking to develop novel vaccines and therapies and for biopharmaceutical customers who use our products as raw materials or who are engaged in preclinical studies and clinical trials. Biopharmaceutical customers are subject to extensive regulations by the FDA and similar regulatory authorities in other countries for conducting clinical trials and commercializing products for therapeutic or diagnostic use. This regulatory scrutiny results in our customers imposing rigorous quality requirements on us as their supplier through supplier qualification processes and customer contracts.
Additionally, regulatory authorities and our customers may conduct scheduled or unscheduled periodic inspections of our facilities to monitor our regulatory compliance or compliance with our quality agreements with our customers. There are significant risks at each stage of the regulatory scheme for our customers.
Regulatory agencies may in the future take action against us or our customers for failure to comply with applicable regulations governing clinical trials and the development and testing of therapeutic products. Failure by us or by our customers to comply with the requirements of these regulatory authorities, including without limitation, remediating any inspectional observations to the satisfaction of these regulatory authorities, could result in warning letters, product recalls or seizures, monetary sanctions, injunctions to halt manufacture and distribution, restrictions on our operations, civil or criminal sanctions, or withdrawal of existing or denial of pending approvals, including those relating to products or facilities. In addition, such a failure could expose us to contractual or product liability claims, contractual claims from our customers, including claims for reimbursement for lost or damaged active pharmaceutical ingredients, as well as ongoing remediation and increased compliance costs, any or all of which could be significant.
We are also subject to a variety of federal, state, local and international laws and regulations that govern, among other things, the importation and exportation of products, the handling, transportation and manufacture of substances that could be classified as hazardous, and our business practices in the U.S. and abroad such as anti-corruption and anti-competition laws. Any noncompliance by us with applicable laws and regulations or the failure to maintain, renew or obtain necessary permits and licenses could result in criminal, civil and administrative penalties and could have an adverse effect on our results of operations.
Increasing scrutiny and changing expectations from investors, lenders, customers, government regulators and other market participants with respect to our Environmental, Social and Governance (“ESG”) policies and activities may impose additional costs on us or expose us to additional risks.
Companies across all industries and around the globe are facing increasing scrutiny relating to their ESG policies, initiatives and activities by investors, lenders, customers, government regulators and other market participants. In particular, these constituencies are increasingly focusing on environmental stewardship, including climate change, water use, deforestation,
waste, and other sustainability concerns, as well as diversity and inclusion, workplace conduct, support for local communities, and other human capital and social issues.
There is no guarantee that any ESG or sustainability goals set forth in our ESG initiatives will be achieved on the desired timeframe or at all, and the achievement of any such goals may require the incurrence of additional costs or the implementation of operational changes, any of which could adversely affect the Company’s results of operations.
Additionally, changes in legal and regulatory requirements related to ESG have been issued in the E.U., its Member States and other countries, particularly with respect to climate change, emission reduction and environmental stewardship in the U.S., amongst other regulatory efforts, the SEC has proposed rules to enhance and standardize climate-related disclosures in public company filings. We expect legal, regulatory and reporting requirements related to ESG matters to continue to expand globally and increase our costs of compliance.
If we are unable to meet our ESG initiatives or evolving investor, industry, or customer expectations and standards, or we are perceived to have not responded adequately on any number of ESG matters, we risk damage to our brand and reputation, adverse impacts to our ability to secure government contracts, decreased desirability of our common stock to investors, or limited access to capital markets and other sources of financing.
Risks Related to Our Intellectual Property and Technology
If we are unable to obtain, maintain and enforce intellectual property protection for our current or future products, or if the scope of our intellectual property protection is not sufficiently broad, our ability to commercialize our products successfully and to compete effectively may be materially adversely affected.
Our success depends on our ability to obtain and maintain patent and other intellectual property protection in the United States and other countries with respect to our current and future proprietary products. We rely upon a combination of patents and trade secret protection to protect the intellectual property related to our technology, manufacturing processes, and products. Our commercial success depends in part on obtaining and maintaining patent and trade secret protection for our current and future products, if any, and the methods used to manufacture them, as well as successfully defending and protecting such patents and trade secrets against third-party challenges. Our ability to stop third parties from making, using, selling, offering to sell or importing our products is dependent upon the extent to which we have rights under valid and enforceable patents and other intellectual property that covers these activities.
The patent prosecution process is expensive and time consuming, and we may not be able to file and prosecute all necessary or desirable patent applications at a reasonable cost or in a timely manner or in all jurisdictions where protection may be commercially advantageous. It is also possible that we may fail to identify patentable aspects of our research and development output before it is too late to obtain patent protection. In addition, we or our collaborators may only pursue, obtain or maintain patent protection in a limited number of countries. There is no assurance that all potentially relevant prior art relating to our patents and patent applications has been found. We may be unaware of prior art that could be used to invalidate or narrow the scope of an issued patent or prevent our pending patent applications from issuing as patents. This may be (1) because patent applications in the United States, Europe and many other non-U.S. jurisdictions are typically not published until 18 months after filing, or in some cases not at all, (2) because publications of discoveries in scientific literature lag behind actual discoveries, and (3) because we cannot be certain that we or our licensors were the first to make the inventions claimed in any of our owned or any in-licensed issued patents or pending patent applications, or that we or our licensors were the first to file for protection of the inventions set forth in our patents or patent applications. As a result, we may not be able to obtain or maintain protection for certain inventions. Even if patents do successfully issue, such patents may not adequately protect our intellectual property, provide exclusivity for our current or future products, prevent others from designing around our claims or otherwise provide us with a competitive advantage. We cannot offer any assurances about which, if any, patents will issue, the breadth of any such patents or whether any issued patents will be found invalid or unenforceable or will be threatened by third parties. In addition, third parties may challenge the validity, enforceability, ownership, inventorship or scope of any of our patents. Any successful challenge to any of our patents could deprive us of rights necessary for the successful commercialization of our current or future products and could impair or eliminate our ability to collect future revenue and royalties with respect to such products. If any of our patent applications with respect to our current or future products fail to result in issued patents, if their breadth or strength of protection is narrowed or threatened, or if they fail to provide meaningful exclusivity or competitive position, it could dissuade companies from collaborating with us or otherwise adversely affect our competitive position.
The patent positions of life science companies can be highly uncertain and involve complex legal and factual questions for which important legal principles remain unresolved. No consistent policy regarding the breadth of claims allowed in life science patents has emerged to date in the United States. The standards applied by the United States Patent and Trademark Office (the “USPTO”) and foreign patent offices in granting patents are not always applied uniformly or predictably and can change.
Additionally, the laws of some foreign countries do not protect intellectual property rights to the same extent as the laws of the United States, and many companies have encountered significant problems in protecting and defending such rights in foreign jurisdictions. The legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents and other intellectual property rights, particularly those relating to biotechnology, which could make it difficult for us to stop the infringement, misappropriation, or other violation of our patents or other intellectual property, including the unauthorized reproduction of our manufacturing or other know-how or the marketing of competing products in violation of our intellectual property rights generally. Any of these outcomes could impair our ability to prevent competition from third parties, which may have a material adverse effect on our business, financial condition, results of operations, cash flows and prospects.
Further, the existence of issued patents does not guarantee our right to practice the patented technology or commercialize products covered by such a patent. Third parties may have or obtain rights to patents which they may use to prevent or attempt to prevent us from practicing our patented technology or commercializing our patented products. If any of these other parties are successful in obtaining valid and enforceable patents, and establishing our infringement of those patents, we could be prevented from selling our products unless we were able to obtain a license under such third-party patents, which may not be available on commercially reasonable terms or at all. In addition, third parties may seek approval to market their own products similar to or otherwise competitive with our products. In these circumstances, we may need to defend or assert our patents, including by filing lawsuits alleging patent infringement. In any of these types of proceedings, a court or agency of competent jurisdiction may find our patents invalid or unenforceable. Our competitors and other third parties may also be able to circumvent our patents by developing similar or alternative products in a non-infringing manner. Any of the foregoing could have a material adverse effect on our business, financial condition, results of operations, cash flows and prospects.
In addition, competitors may use our technologies in jurisdictions where we have not obtained or are unable to adequately enforce patent protection to develop their own products and further, may export otherwise infringing products to territories where we have patent protection, but enforcement is not as strong as that in the United States and Europe. These products may compete with our products, and our patents or other intellectual property rights may not be effective or sufficient to prevent them from competing with us. Proceedings to enforce our patent rights, whether or not successful, could result in substantial costs and divert our efforts and attention from other aspects of our business, could put our patents at risk of being invalidated or held unenforceable, or interpreted narrowly and our patent applications at risk of not issuing, and could provoke third parties to assert claims against us. We may not prevail in any lawsuits that we initiate, and the damages or other remedies awarded, if any, may not be commercially meaningful. Accordingly, our efforts to enforce our intellectual property rights around the world may be inadequate to obtain a significant commercial advantage from the intellectual property that we develop, acquire or license.
Intellectual property that we own or in-license may be subject to a reservation of rights by one or more third parties. For example, one of our patents is co-owned with third parties and some of our patent rights in the future may be co-owned with third parties. If we are unable to obtain an exclusive license to any such third-party co-owners’ interest in such patent rights, such co-owners may be able to license their rights to other third parties, including our competitors, and our competitors could market competing products and technology. In addition, we may need the cooperation of any such co-owners of such patent rights in order to enforce such patent rights against third parties, and such cooperation may not be provided to us. Any of the foregoing could have a material adverse effect on our competitive position, business, financial conditions, results of operations, and prospects.
Moreover, the research resulting in certain of our patents and technology was funded in part by the U.S. government. As a result, the U.S. government has certain rights to such patent rights and technology, which include march-in rights. When new technologies are developed with government funding, in order to secure ownership of such patent rights, the recipient of such funding is required to comply with certain government regulations, including timely disclosing the inventions claimed in such patent rights to the U.S. government and timely electing title to such inventions. Additionally, the U.S. government generally obtains certain rights in any resulting patents, including a nonexclusive license authorizing the government to use the invention or to have others use the invention on its behalf. Accordingly, we or our licensors have granted the U.S. government a nonexclusive, nontransferable, irrevocable, paid-up license to practice or have practiced for or on behalf of the United States, the inventions described in the patents and patent applications relating to such inventions. If the U.S. government decides to exercise these rights, it is not required to engage us as its contractor in connection with doing so. The government’s rights may also permit it to disclose our confidential information to third parties and to exercise march-in rights to use or allow third parties to use such government-funded technology. The government can exercise its march-in rights if it determines that action is necessary because we fail to achieve practical application of the government-funded technology, or because action is necessary to alleviate health or safety needs, to meet requirements of federal regulations, or to give preference to U.S. industry. In addition, our rights in such inventions may be subject to certain requirements to manufacture products embodying such inventions in the United States. If we fail to comply with those requirements, we could lose our ownership of or other rights to any patents subject to such regulations. Any exercise by the government of any of the foregoing rights or by any third party of its reserved rights could have a material adverse effect on our competitive position, business, financial condition, results of operations, and prospects.
Furthermore, patents have a limited lifespan. In the United States, the unextended expiration of a patent is generally 20 years after its non-provisional application filing date. Various extensions may be available, however, the life of a patent and the protection it affords is limited. Given the amount of time required for the development, testing, regulatory review and approval of new products, our patents protecting such candidates might expire before or shortly after such candidates are commercialized. If we encounter delays in obtaining regulatory approvals, the period of time during which we could market a product under patent protection could be further reduced. Even if patents covering our future products are obtained, once such patents expire, we may be vulnerable to competition from similar products. The launch of a similar version of one of our products would likely result in an immediate and substantial reduction in the demand for our product. As a result, our patent portfolio may not provide us with sufficient rights to exclude others from commercializing products similar or identical to ours. Any of the foregoing could have a material adverse effect on our business, financial condition, results of operations, cash flows and prospects.
If we are prevented from enforcing our intellectual property rights because of governmental regulatory policies or political pressure or action, our sales and profitability may be materially adversely affected.
Our ability to maintain and grow our product sales and profitability depends, in part, on our ability to maintain and enforce our patents and other intellectual property rights. Proposed actions to waive intellectual property protections for COVID-19 vaccines and associated technology, such as those under discussion at the World Trade Organization, which are supported by the U.S. government, may impact our ability to fully assert our intellectual property rights related to our CleanCap® product in connection with the production of COVID-19 vaccines. Further, these policy actions may complicate our analysis and decision-making with respect to both research and development and capital investment, given the potential for lower returns on those investments that could result from our inability to fully protect our intellectual property. If we are unable to successfully navigate these considerations, the future revenues and profitability of our business could be negatively impacted. We are unable to estimate the impact of these potential policies given that they remain undefined and their adoption is uncertain.
Our internal computer systems, or those of our customers, collaborators or other contractors, have been and may in the future be subject to cyber-attacks or security breaches, which could result in a material disruption of our product development programs or otherwise adversely affect our business, financial condition, results of operations, cash flows and prospects.
Despite the implementation of security measures, our internal computer systems and those of our customers are vulnerable to damage from computer viruses and unauthorized access. Cyber-attacks are increasing in their frequency, sophistication and intensity, and have become increasingly difficult to detect. Cyber-attacks could include the deployment of harmful malware, ransomware, denial-of-service attacks, social engineering and other means to affect service reliability and threaten the confidentiality, integrity and availability of information. Cyber-attacks also could include phishing attempts or e-mail fraud to cause unauthorized payments or information to be transmitted to an unintended recipient. A material cyber-attack or security breach could cause interruptions in our operations and could result in a material disruption of our business operations, damage to our reputation, financial condition, results of operations, cash flows and prospects.
In the ordinary course of our business, we collect and store sensitive data, including, among other things, personally identifiable information about our employees, intellectual property, and proprietary business information. Any cyber-attack or security breach that leads to unauthorized access, use or disclosure of personal or proprietary information could harm our reputation, cause us not to comply with federal and/or state breach notification laws and foreign law equivalents and otherwise subject us to liability under laws and regulations that protect the privacy and security of personal information. In addition, we could be subject to risks caused by misappropriation, misuse, leakage, falsification or intentional or accidental release or loss of information maintained in the information systems and networks of our company and our vendors, including personal information of our employees, and company and vendor confidential data. In addition, outside parties have previously attempted and may in the future attempt to penetrate our systems or those of our vendors or fraudulently induce our personnel or the personnel of our vendors to disclose sensitive information in order to gain access to our data and/or systems or make unauthorized payments to third parties. Like other companies, we have on occasion experienced, and will continue to experience, data security incidents involving access to company data, unauthorized payments and threats to our data and systems, including malicious codes and viruses, phishing, business email compromise attacks, or other cyber-attacks. The number and complexity of these threats continue to increase over time. If a material breach of our information technology systems or those of our vendors occurs, the market perception of the effectiveness of our security measures could be harmed and our reputation and credibility could be damaged.
We could be required to expend significant amounts of money and other resources to respond to these threats or breaches and to repair or replace information systems or networks and could suffer financial loss or the loss of valuable confidential information. In addition, we could be subject to regulatory actions and/or claims made by individuals and groups in private litigation involving privacy issues related to data collection and use practices and other data privacy laws and regulations,
including claims for misuse or inappropriate disclosure of data, as well as unfair or deceptive practices. Although we develop and maintain systems and controls designed to prevent these events from occurring, and we have a process to identify and mitigate threats, the development and maintenance of these systems, controls and processes is costly and requires ongoing monitoring and updating as technologies change and efforts to overcome security measures become increasingly sophisticated. Moreover, despite our efforts, the possibility of these events occurring cannot be eliminated entirely and there can be no assurance that any measures we take will prevent cyber-attacks or security breaches that could adversely affect our business, financial condition, results of operations, cash flows and prospects.
If we are unable to protect the confidentiality of our proprietary information, the value of our technology and products could be materially adversely affected.
We also may rely on trade secrets to protect our technology, especially where we do not believe patent protection is appropriate or obtainable. To maintain the confidentiality of trade secrets and other proprietary information, we enter into confidentiality agreements with our employees, consultants, contractors, collaborators, CDMOs, CROs and others upon the commencement of their relationships with us. These agreements require that all confidential information developed by the individual or entity or made known to the individual or entity by us during the course of the individual’s or entity’s relationship with us be kept confidential and not disclosed to third parties. Our agreements with employees as well as our personnel policies also generally provide that any inventions conceived by the individual in the course of rendering services to us shall be our exclusive property or that we may obtain full rights to such inventions at our election. However, trade secrets are difficult to protect. Although we use reasonable efforts to protect our trade secrets, our employees, consultants, contractors, collaborators, CDMOs, CROs and others may unintentionally or willfully disclose our information to competitors. We also face the risk that present or former employees could continue to hold rights to intellectual property used by us, demand the registration of intellectual property rights in their name, and seek payment of damages for our use of such intellectual property.
Enforcing a claim that a third party illegally obtained or is using any of our trade secrets is expensive and time consuming, and the outcome is unpredictable. We may not have adequate remedies in the event of unauthorized use or disclosure of our trade secrets or other proprietary information in the case of a breach of any such agreements and our trade secrets and other proprietary information could be disclosed to third parties, including our competitors. Many of our partners also collaborate with our competitors and other third parties. The disclosure of our trade secrets to our competitors, or more broadly, would impair our competitive position and may materially harm our business, financial condition, results of operations, cash flows and prospects. Costly and time-consuming litigation could be necessary to enforce and determine the scope of our rights, and failure to maintain trade secret protection could adversely affect our competitive business position. The enforceability of confidentiality agreements may vary from jurisdiction to jurisdiction. Courts outside the United States are sometimes less willing to protect trade secrets. Moreover, our competitors may independently develop substantially equivalent or superior knowledge, methods and know-how, and the existence of our own trade secrets affords no protection against such independent discovery.
We may become involved in lawsuits to protect or enforce our patents, which could be expensive, time-consuming and unsuccessful and could result in a court or administrative body finding our patents to be invalid or unenforceable.
Even if the patent applications we own or license are issued, third parties may challenge or infringe upon our patents. To counter infringement, we may be required to file infringement claims, which can be expensive and time-consuming. In patent litigation in the United States, defendant counterclaims alleging invalidity or unenforceability are commonplace. Grounds for a validity challenge could be an alleged failure to meet any of several statutory requirements, including novelty, non-obviousness (or inventive step), written description or enablement. In addition, patent validity challenges may, under certain circumstances, be based upon non-statutory obviousness-type double patenting, which, if successful, could result in a finding that the claims are invalid for obviousness-type double patenting or the loss of patent term if a terminal disclaimer is filed to obviate a finding of obviousness-type double patenting. Grounds for an unenforceability assertion could be an allegation that someone connected with prosecution of the patent withheld information material to patentability from the USPTO, or made a misleading statement, during prosecution.
Third parties may raise similar claims before administrative bodies in the United States or abroad, even outside the context of litigation. Such mechanisms include re-examination, post-grant review, inter partes review, interference proceedings, derivation proceedings, and equivalent proceedings in foreign jurisdictions (e.g., opposition proceedings). Such proceedings could result in the revocation or cancellation of or amendment to our patents in such a way that they no longer cover our current or future products or provide any competitive advantage. The outcome following legal assertions of invalidity and unenforceability is unpredictable. If a third party were to prevail on a legal assertion of invalidity or unenforceability, we could lose part or all of the patent protection on one or more of our current or future products, which could result in our competitors and other third parties using our technology to compete with us. Such a loss of patent protection could have a material adverse impact on our business, financial condition, results of operations, cash flows and prospects.
Interference proceedings, or other similar enforcement and revocation proceedings, provoked by third parties or brought by us may be necessary to determine the priority of inventions with respect to our patents or patent applications. An unfavorable outcome could require us to cease using the related technology or to attempt to license rights to it from the prevailing party. Our business could be harmed if the prevailing party does not offer us a license on commercially reasonable terms. Our defense of litigation or interference proceedings may fail and, even if successful, may result in substantial costs and distract our management and other employees. We may not be able to prevent, alone or with our licensors, infringement, misappropriation or other violation of our intellectual property rights, particularly in countries where the laws may not protect those rights as fully as in the United States.
In an infringement proceeding, even one initiated by us, there is a risk that a court will decide that our patents are not valid and that we do not have the right to stop the other party from using the inventions they describe. There is also the risk that, even if the validity of such patents is upheld, the court will refuse to stop the other party on the ground that such other party’s activities do not infringe our rights to these patents.
Some of our competitors may be able to sustain the costs of complex patent litigation more effectively than we can because they have substantially greater resources. In addition, any uncertainties resulting from the initiation and continuation of any litigation could have a material adverse effect on our ability to raise the funds necessary to continue our operations. In addition, patent holding companies that focus solely on extracting royalties and settlements by enforcing patent rights may target us, especially as we gain greater visibility and market exposure as a public company.
An adverse outcome in a litigation or proceeding involving our patents could limit our ability to assert our patents against competitors, affect our ability to receive royalties or other licensing consideration from our licensees, and may curtail or preclude our ability to exclude third parties from making, using and selling similar or competitive products. Any of these occurrences could have a material adverse effect on our business, financial condition, results of operations, cash flows and prospects.
If we are sued for infringing, misappropriating, or otherwise violating intellectual property rights of third parties, such litigation could be costly and time consuming and could prevent or delay us from developing or commercializing our current or future products.
Our products may infringe on, or be accused of infringing on, one or more claims of an issued patent or may fall within the scope of one or more claims in a published patent application that may be subsequently issued and to which we do not hold a license or other rights.
Because patent applications in the United States and many foreign jurisdictions are typically not published until 18 months after filing, or in some cases not at all, and publications in the scientific literature often lag behind actual discoveries, we cannot be certain that others have not filed patent applications for technology covered by our issued patents or our pending applications, or that we were the first to invent the technology. Others, including our competitors, may have filed, and may in the future file, patent applications covering technology similar to ours. Any such patent application may have priority over our patent applications or patents, which could further require us to obtain rights to issued patents by others covering such technologies. If another party has filed a U.S. patent application on inventions similar to ours, we may have to participate in an interference proceeding declared by the USPTO to determine priority of invention in the United States. The costs of these proceedings could be substantial, and it is possible that such efforts would be unsuccessful if, unbeknownst to us, the other party had independently arrived at the same or similar invention prior to our own invention, resulting in a loss of our U.S. patent position with respect to such inventions.
Additionally, pending patent applications that have been published can, subject to certain limitations, be later amended in a manner that could cover our current or future products or the use of our current or future products. After issuance, the scope of patent claims remains subject to construction based on interpretation of the law, the written disclosure in a patent and the patent’s prosecution history. Our interpretation of the relevance or the scope of a patent or a pending application may be incorrect. In addition, third parties may obtain patents in the future and claim that use of our technologies infringes upon these patents. These third parties could bring claims against us or our collaborators that would cause us to incur substantial expenses and, if successful against us, could cause us to pay substantial damages.
The life sciences industry has produced a proliferation of patents, and it is not always clear to industry participants, including us, which patents cover various types of products or methods of use. Because the patent granting process is imperfect, the manufacture, distribution, or sale of our products may require us to challenge intellectual property rights by third parties that we believe to have been improperly granted. The coverage of patents is subject to interpretation by the courts, and the interpretation is not always uniform. If we are sued for patent infringement, we would need to demonstrate that our products or methods of use either do not infringe the patent claims of the relevant patent and/or that the patent claims are invalid or unenforceable, and we may not be able to do this. Proving invalidity, in particular, is difficult since it requires a showing of clear and convincing evidence in trial court litigation to overcome the presumption of validity enjoyed by issued patents. Third parties have, and may
in the future have, U.S. and non-U.S. issued patents and pending patent applications that may cover our current or future products. Such a third party may claim that we or our manufacturing or commercialization partners are using inventions covered by the third party’s patent rights and may go to court or a tribunal to stop us from engaging in our normal operations and activities, including making or selling our current or future products. In the event that any of these patent rights were asserted against us, we believe that we have defenses against any such action, including that such patents would not be infringed by our current or future products and/or that such patents are not valid. However, if any such patent rights were to be asserted against us and our defenses to such assertion were unsuccessful, unless we obtain a license to such patents, we could be liable for damages, which could be significant and include treble damages and attorneys’ fees if we are found to willfully infringe such patents, and we could be precluded from commercializing any future products that were ultimately held to infringe such patents, any of which could have a material adverse effect on our business, financial condition, results of operations, cash flows and prospects.
If we are found to infringe the patent rights of a third party, or in order to avoid potential claims, we or our collaborators may choose or be required to seek a license from a third party and be required to pay license fees or royalties or both. These licenses may not be available on reasonable terms, or at all. In particular, any of our competitors that control intellectual property that we are found to infringe may be unwilling to provide us a license under any terms. Even if we or our collaborators were able to obtain a license, the rights may be nonexclusive, which could result in our competitors gaining access to the same intellectual property. Ultimately, we could be prevented from commercializing a product, or be forced to cease some aspect of our business operations, if, as a result of actual or threatened patent infringement claims, we or our collaborators are unable to enter into licenses on acceptable terms. In addition, we could be found liable for monetary damages, including treble damages and attorneys’ fees if we are found to have willfully infringed a patent. Further, if a patent infringement suit is brought against us or our third-party service providers and if we are unable to successfully obtain rights to required third-party intellectual property, we may be required to expend significant time and resources to redesign our current or future products, or to develop or license replacement technology, all of which may not be feasible on a technical or commercial basis, and may delay or require us to abandon our development, manufacturing or sales activities relating to our current or future products. A finding of infringement could prevent us from commercializing our future products or force us to cease some of our business operations, which could harm our business. Claims that we have misappropriated the confidential information or trade secrets of third parties could have a similar negative impact on our business. Any of the foregoing could have a material adverse effect on our business, financial condition, results of operations, cash flows and prospects.
Intellectual property litigation and other proceedings could cause us to spend substantial resources and distract our personnel from their normal responsibilities.
Even if resolved in our favor, intellectual property litigation or other legal proceedings relating to our, our licensors’ or other third parties’ intellectual property claims may cause us to incur significant expenses and could distract our personnel from their normal responsibilities. Patent litigation and other proceedings may also absorb significant management time. If not resolved in our favor, litigation may require us to pay any portion of our opponents’ legal fees. Such litigation or proceedings could substantially increase our operating losses and reduce the resources available for development activities or any future sales, marketing, or distribution activities. We may not have sufficient financial or other resources to conduct such litigation or proceedings adequately. Our competitors or other third parties may be able to sustain the cost of such litigation and proceedings more effectively than we can because of their substantially greater resources. Uncertainties resulting from our participation in patent litigation or other proceedings could have a material adverse effect on our ability to compete in the marketplace. Furthermore, because of the substantial amount of discovery required in certain jurisdictions in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during this type of litigation. There could also be public announcements of the results of hearings, motions or other interim proceedings or developments. If securities analysts or investors perceive these results to be negative, the perceived value of our current or future products or intellectual property could be diminished. Accordingly, the market price of our Class A common stock may decline. Uncertainties resulting from the initiation and continuation of patent litigation or other proceedings could have a material adverse effect on our business, financial condition, results of operations, and prospects.
If we fail to comply with our obligations under any license agreements, disagree over contract interpretation, or otherwise experience disruptions to our business relationships with our licensors, we could lose intellectual property rights that are necessary to our business.
We rely, in part, on intellectual property and technology which we have in-licensed. We may also need to obtain additional licenses in the future to advance our research or allow commercialization of our future products and it is possible that we may be unable to do so at a reasonable cost or on reasonable terms, if at all. Moreover, such licenses may not provide exclusive rights to use such intellectual property and technology in all relevant fields of use and in all territories in which we may wish to develop or commercialize our future products.
In addition, our existing license agreements impose, and any future license agreements we enter into may impose, various development, commercialization, funding, milestone, royalty, diligence, sublicensing, insurance, patent prosecution and enforcement or other obligations on us. Our license agreements, and any future license agreement we enter into, may also impose restrictions on our ability to license certain of our intellectual property to third parties or to develop or commercialize certain current or future products or technologies. In spite of our best efforts, our counterparties may conclude that we have breached our obligations under our agreements, or that we have used the intellectual property licensed to us in an unauthorized manner, in which case, we may be required to pay damages and the counterparty may have the right to terminate the agreement. Any of the foregoing could result in us being unable to develop, manufacture and sell products that are covered by the licensed intellectual property or technology, or enable a competitor to gain access to the licensed intellectual property or technology.
We might not have the necessary rights or the financial resources to develop, manufacture or market our current or future products without the rights granted under our license agreements, and the loss of sales or potential sales in current or future products covered by such license agreements could have a material adverse effect on our business, financial condition, results of operations, cash flows and prospects.
Disputes may arise regarding intellectual property subject to license agreements, including:
•the scope of rights granted under the license agreement and other interpretation related issues;
•the extent to which our technology and processes infringe on intellectual property of the licensor that is not subject to the license agreement;
•the sublicensing of patent and other rights under our collaborative development relationships;
•our diligence obligations under the license agreement and what activities satisfy those diligence obligations;
•our financial obligations under the license agreement;
•the inventorship and ownership of inventions and know-how resulting from the joint creation or use of intellectual property by our licensors and us and our partners; and
•the priority of invention of patented technology.
In addition, the agreements under which we currently license intellectual property or technology to or from third parties are complex, and certain provisions in such agreements may be susceptible to multiple interpretations. The resolution of any contract interpretation disagreement that may arise could narrow what we believe to be the scope of our rights to the relevant intellectual property or technology, or increase what we believe to be our financial or other obligations under the relevant agreement, either of which could have a material adverse effect on our business, financial condition, results of operations, and prospects. Moreover, if disputes over intellectual property that we have licensed prevent or impair our ability to maintain our current licensing arrangements on commercially acceptable terms, we may be unable to successfully develop and commercialize the affected future products.
In some cases, we may not have primary control over prosecution, maintenance, enforcement and defense of patents and patent applications that we have in-licensed from third parties, and instead we rely on our licensors for these activities. We cannot be certain that such activities have been or will be conducted in compliance with applicable laws and regulations or in a manner consistent with the best interests of our business. If we do undertake any enforcement of our in-licensed patents or defense of any claims asserting the invalidity of such patents, such actions may be subject to the cooperation of our licensors or other third parties. If our licensors or other third parties fail to prosecute, maintain, enforce and defend intellectual property licensed to us, or lose their own rights to such intellectual property, the rights we have licensed may be impaired or eliminated and our ability to develop and commercialize any of our products that are subject to such rights could be adversely affected.
In-licensing or acquisition of third-party intellectual property is a competitive area and a number of more established companies are also pursuing strategies to in-license or acquire third-party intellectual property rights that we may consider attractive or necessary for our business. These companies may have a competitive advantage over us due to their size, cash resources and greater capabilities with respect to clinical development and commercialization. Furthermore, companies that perceive us as a competitor may be unwilling to assign or license rights to us. If we are unable to successfully obtain rights to required third-party intellectual property rights or maintain the existing intellectual property rights we have on reasonable terms or at all, we may have to abandon development of the relevant program or current or future product and our business, financial condition, results of operations, cash flows and prospects could suffer.
Changes to the patent law in the United States and other jurisdictions could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents, thereby impairing our ability to protect our technologies and current or future products.
As is the case with other life sciences companies, our success is heavily dependent on intellectual property, particularly patents. Obtaining and enforcing patents in the life sciences industry involves both technological and legal complexity and is therefore costly, time consuming and inherently uncertain. Changes in either the patent laws or in interpretations of patent laws in the United States and other countries may increase the uncertainties and costs surrounding the prosecution of patent applications and the enforcement or defense of issued patents.
For example, the Leahy-Smith America Invents Act (the “America Invents Act”), was signed into law on September 16, 2011, and many of the substantive changes became effective on March 16, 2013. The America Invents Act and its implementation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents, all of which could have a material adverse effect on our business, financial condition, results of operations, and prospects. Specifically, the America Invents Act reforms United States patent law in part by changing the U.S. patent system from a “first to invent” system to a “first inventor to file” system. Under a “first inventor to file” system, assuming the other requirements for patentability are met, the first inventor to file a patent application generally will be entitled to the patent on an invention regardless of whether another inventor was the first to invent the invention. This will require us to be cognizant going forward of the time from invention to filing of a patent application and be diligent in filing patent applications. Circumstances may arise that could prevent us from promptly filing patent applications on our inventions and allow third parties to file patents claiming our inventions before we are able to do so. The America Invents Act also includes a number of significant changes that affect the way patent applications will be prosecuted and may also affect patent litigation. These include allowing third-party submission of prior art to the USPTO during patent prosecution and additional procedures to attack the validity of a patent by the USPTO administered post grant proceedings, including reexamination proceedings, inter partes review, post grant review and derivation proceedings. These adversarial proceedings at the USPTO review patent claims without the presumption of validity afforded to U.S. patents in lawsuits in U.S. federal courts, and use a lower burden of proof than used in litigation in U.S. federal courts. Therefore, it is generally considered easier for a competitor or third party to have a U.S. patent invalidated in a USPTO post-grant review or inter partes review proceeding than in a litigation in a U.S. federal court.
In addition, the patent positions of companies in the life sciences industry are particularly uncertain. Recent U.S. Supreme Court rulings have narrowed the scope of patent protection available in certain circumstances and weakened the rights of patent owners in certain situations. This combination of events has created uncertainty with respect to the validity and enforceability of patents, once obtained. Depending on future actions by the U.S. Congress, the federal courts, and the USPTO, the laws and regulations governing patents could change in unpredictable ways. In addition, the complexity and uncertainty of European patent laws have also increased in recent years. Complying with these laws and regulations could have a material adverse effect on our existing patent portfolio and our ability to protect and enforce our intellectual property in the future.
Obtaining and maintaining our patent protection depends on compliance with various procedural, documentary, fee payment and other requirements imposed by governmental patent agencies, and our patent protection could be reduced or eliminated for noncompliance with these requirements.
Periodic maintenance fees, renewal fees, annuity fees and various other governmental fees on patents and patent applications will be due to be paid to the USPTO and various government patent agencies outside the United States over the lifetime of our patents and patent applications and any patent rights we may own or license in the future. Additionally, the USPTO and various government patent agencies outside the United States require compliance with a number of procedural, documentary, fee payment and other similar provisions during the patent application process. In certain cases, an inadvertent lapse can be cured by payment of a late fee or by other means in accordance with rules applicable to the particular jurisdiction. However, there are situations in which noncompliance can result in abandonment or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction. If we or our licensors fail to maintain the patents and patent applications covering or otherwise protecting our current or future products, it could have a material adverse effect on our business. In addition, to the extent that we have responsibility for taking any action related to the prosecution or maintenance of patents or patent applications in-licensed from a third party, any failure on our part to maintain the in-licensed intellectual property could jeopardize our rights under the relevant license and may have a material adverse effect on our business, financial condition, results of operations, cash flows and prospects.
We may be subject to claims by third parties asserting that our employees, consultants, independent contractors or we have misappropriated their intellectual property, or claiming ownership of what we regard as our own intellectual property and proprietary technology.
Many of our employees were previously employed at universities or other life science, biotechnology or pharmaceutical companies, including our competitors or potential competitors. We try to ensure that our employees do not use the proprietary information or know-how of others in their work for us. We may, however, be subject to claims that we or these employees have inadvertently or otherwise used or disclosed intellectual property, trade secrets or other proprietary information of any such employee’s former employer or that patents and applications we have filed to protect inventions of these individuals, even those related to one or more of our current or future products, are rightfully owned by their former or concurrent employer. Litigation may be necessary to defend against these claims. Even if we are successful in defending ourselves, such litigation could result in substantial costs to us or be distracting to our management. If we fail to defend any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights or personnel or we could be required to obtain a license from such third party to commercialize our technology or products. Such a license may not be available on an exclusive basis or on commercially reasonable terms or at all.
In addition, while we typically require our employees, consultants and independent contractors who may be involved in the development of intellectual property to execute agreements assigning such intellectual property to us, we may be unsuccessful in executing such an agreement with each party who in fact develops intellectual property that we regard as our own, or such agreements may be breached or alleged to be ineffective, and the assignment may not be self-executing, which may result in claims by or against us related to the ownership of such intellectual property or may result in such intellectual property becoming assigned to third parties. If we fail in enforcing or defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights. Even if we are successful in prosecuting or defending against such claims, litigation could result in substantial costs and be a distraction to our senior management and scientific personnel. Any of the foregoing could have a material adverse effect on our business, financial condition, results of operations, cash flows and prospects.
We may not be able to protect our intellectual property and proprietary rights throughout the world.
Filing, prosecuting, and defending patents on current or future products in all countries throughout the world would be prohibitively expensive, and the laws of foreign countries may not protect our rights to the same extent as the laws of the United States. For example, patent scope or coverage varies between countries based on the differences between the respective patent laws in each country or jurisdiction. Consequently, we may not be able to prevent third parties from practicing our inventions in all countries outside the United States, or from selling or importing products made using our inventions in and into the United States or other jurisdictions. Third parties may use our technologies in jurisdictions where we have not obtained or are unable to adequately enforce patent protection to develop their own products and, further, may export otherwise infringing products to territories where we have patent protection but enforcement is not as strong as that in the United States. These products may compete with our products, and our patents or other intellectual property rights may not be effective or sufficient to prevent them from competing.
Many companies have encountered significant problems in protecting and defending intellectual property rights in foreign jurisdictions. The legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents, trade secrets, and other intellectual property protection, particularly those relating to biotechnology products, which could make it difficult for us to stop the infringement of our patents or marketing of competing products in violation of our intellectual property and proprietary rights generally. Proceedings to enforce our intellectual property and proprietary rights in foreign jurisdictions could result in substantial costs and divert our efforts and attention from other aspects of our business, could put our patents at risk of being invalidated or interpreted narrowly, could put our patent applications at risk of not issuing, and could provoke third parties to assert claims against us. We may not prevail in any lawsuits that we initiate, and the damages or other remedies awarded, if any, may not be commercially meaningful. Accordingly, our efforts to enforce our intellectual property and proprietary rights around the world may be inadequate to obtain a significant commercial advantage from the intellectual property that we develop or license.
Many countries have compulsory licensing laws under which a patent owner may be compelled to grant licenses to third parties. In addition, many countries limit the enforceability of patents against government agencies or government contractors. In these countries, the patent owner may have limited remedies, which could materially diminish the value of such patent. If we or any of our licensors is forced to grant a license to third parties with respect to any patents relevant to our business, our competitive position may be impaired, and our business, financial condition, results of operations, cash flows and prospects may be adversely affected.
We rely on confidentiality agreements that, if breached, may be difficult to enforce and could have a material adverse effect on our business and competitive position.
Our policy is to enter agreements relating to the non-disclosure and non-use of confidential information with third parties, including our contractors, consultants, advisors and research collaborators, as well as agreements that purport to require the disclosure and assignment to us of the rights to the ideas, developments, discoveries and inventions of our employees and consultants while we employ them. However, these agreements can be difficult and costly to enforce. Moreover, to the extent that our contractors, consultants, advisors and research collaborators apply or independently develop intellectual property in connection with any of our projects, disputes may arise as to the proprietary rights to the intellectual property. If a dispute arises, a court may determine that the right belongs to a third party, and enforcement of our rights can be costly and unpredictable. In addition, we rely on trade secrets and proprietary know-how that we seek to protect in part by confidentiality agreements with our employees, contractors, consultants, advisors or others. Despite the protective measures we employ, we still face the risk that:
•these agreements may be breached;
•these agreements may not provide adequate remedies for the applicable type of breach; or
•our trade secrets or proprietary know-how will otherwise become known.
Any breach of our confidentiality agreements or our failure to effectively enforce such agreements would have a material adverse effect on our business and competitive position.
If our trademarks, trade dress, and trade names are not adequately protected, we may not be able to build name recognition in our markets of interest and our business, financial condition, results of operations, cash flows and prospects may be adversely affected.
Our trademarks, trade dress, or trade names may be challenged, infringed, circumvented or declared generic or determined to be infringing on other marks. We may not be able to protect our rights to these trademarks and trade names or may be forced to stop using these names or marks which we need for name recognition by potential partners or customers in our markets of interest. During trademark registration proceedings, we may receive rejections. Although we would be given an opportunity to respond to those rejections, we may be unable to overcome such rejections. In addition, in the USPTO and in comparable agencies in many foreign jurisdictions, third parties are given an opportunity to oppose pending trademark applications and to seek to cancel registered trademarks. Opposition or cancellation proceedings may be filed against our trademarks, and our trademarks may not survive such proceedings. If we are unable to establish name recognition based on our trademarks and trade names, we may not be able to compete effectively and our business, financial condition, results of operations, cash flows and prospects may be adversely affected.
Intellectual property rights do not necessarily address all potential threats.
The degree of future protection afforded by our proprietary and intellectual property rights is uncertain because such rights offer only limited protection and may not adequately protect our rights or permit us to gain or keep our competitive advantage. For example:
•others may be able to develop products that are similar to, or better than, our current or future products in a way that is not covered by the claims of the patents we license or may own currently or in the future;
•we, or our licensing partners or current or future collaborators, might not have been the first to make the inventions covered by issued patents or pending patent applications that we license or may own currently or in the future;
•we, or our licensing partners or current or future collaborators, might not have been the first to file patent applications for certain of our or their inventions;
•our pending owned or in-licensed patent applications may not lead to issued patents;
•we may choose not to file a patent for certain trade secrets or know-how, and a third party may subsequently file a patent covering such intellectual property;
•our competitors or other third parties might conduct research and development activities in countries where we do not have patent rights and then use the information learned from such activities to develop competitive products for sale in our major commercial markets;
•it is possible that there are prior public disclosures that could invalidate our or our licensors’ patents;
•the patents of third parties or pending or future applications of third parties, if issued, may have an adverse effect on our business;
•any patents that we obtain may not provide us with any competitive advantages or may ultimately be found not to be owned by us, invalid or unenforceable; or
•we may not develop additional proprietary technologies that are patentable.
Should any of these events occur, they could significantly harm our business, financial conditions, results of operations, cash flows and prospects.
Risks Related to Our Indebtedness
Our existing level of indebtedness may increase and adversely affect our business and growth prospects, growth prospects, and financial condition, as well as our ability to raise additional capital on favorable terms, which could, in turn, limit our ability to develop or acquire new products, services, technologies and methodologies.
As of December 31, 2023, we had total current and long-term indebtedness outstanding of approximately $524.1 million, including term loans of $533.1 million, and unamortized debt issuance costs of $9.0 million. We may incur significant additional indebtedness in the future. If we increase our current indebtedness levels, the risks related to our indebtedness as set forth herein could intensify.
Our indebtedness, or any additional indebtedness we may incur, could require us to divert funds identified for other purposes for debt service and impair our liquidity position. If we cannot generate sufficient cash flow from operations to service our debt, we may need to refinance our debt, dispose of assets or issue equity to obtain necessary funds. We do not know whether we will be able to take any of these actions on a timely basis, on terms satisfactory to us or at all.
Our indebtedness, the cash flow needed to satisfy our debt and the covenants contained in the Credit Agreement have important consequences, including:
•limiting funds otherwise available for financing our capital expenditures by requiring us to dedicate a portion of our cash flows from operations to the repayment of debt and the interest on this debt;
•limiting our ability to incur or prepay existing indebtedness, pay dividends or distributions, dispose of assets, engage in mergers and consolidations, make acquisitions or other investments and make changes in the nature of the business, among other things;
•making us more vulnerable to rising interest rates, as certain of our borrowings, including borrowings under the Credit Agreement, bear variable rates of interest; and
•making us more vulnerable in the event of a downturn in our business.
Our level of indebtedness may place us at a competitive disadvantage to our competitors that are not as highly leveraged. Fluctuations in interest rates can increase borrowing costs. Increases in interest rates may directly impact the amount of interest we are required to pay and reduce earnings accordingly. In addition, tax laws, including the disallowance or deferral of tax deductions for interest paid on outstanding indebtedness, could have an adverse effect on our liquidity and our business, financial condition, results of operations, cash flows and prospects. Further, our Credit Agreement contains customary affirmative and negative covenants and certain restrictions on operations that could impose operating and financial limitations and restrictions on us, including restrictions on our ability to enter into particular transactions and to engage in other actions that we may believe are advisable or necessary for our business.
Variable rate indebtedness that we have incurred or may in the future incur will subject us to interest rate risk, which could cause our debt service obligations to increase significantly.
Certain borrowings under our Credit Agreement bear variable rates of interest. Increase in interest rates directly increase the amount of interest we are required to pay, and negatively impacts our net income and cash flows, including cash available for servicing our indebtedness more generally.
We may not be able to generate sufficient cash flow to service all of our indebtedness and may be forced to take other actions to satisfy our debt service obligations, which actions may not be adequate or may impose additional restrictions on us.
Our ability to make scheduled debt service payments or to refinance outstanding debt obligations depends on our financial and operating performance, which is subject to prevailing economic, industry and competitive conditions and certain financial, business, economic and other factors beyond our control, including those discussed under “Risks Related to Our Business and Strategy” above. We may not be able to maintain a sufficient level of cash flow from operating activities to permit us to pay the principal, premium, if any, and interest on our indebtedness. If we cannot meet our debt service obligations, the holders of our indebtedness would have the right to accelerate such indebtedness and, to the extent such indebtedness is secured, foreclose on our assets. This could have serious consequences to our business, financial condition and results of operations and could cause us to become bankrupt or insolvent. Even if this does not occur, any failure to make payments of interest and principal on our outstanding indebtedness on a timely basis would likely result in a reduction of our creditworthiness, which would also harm our ability to incur additional indebtedness.
If our cash flows and other capital resources are insufficient to fund our debt service obligations, we may be forced to reduce or delay capital expenditures and acquisitions, sell assets, raise additional capital or seek to restructure or refinance our indebtedness. If we issue additional equity to repay all or a portion of our indebtedness, our shareholders may experience significant dilution of their equity interests. Any refinancing of our indebtedness could be at higher interest rates and may require us to comply with more onerous covenants, including the requirement to maintain specified liquidity or other ratios or restrictions on our ability to pay dividends or make acquisitions. If these alternative measures are not successful, we may be required to sell material assets or operations to attempt to meet our debt service obligations. Further, we may not be able to consummate these asset sales (including as a result of restrictions imposed on us under the Credit Agreement) or sell assets at prices and on terms that we believe are fair, and any proceeds that we do receive may not be adequate to meet any debt service obligations then due.
The terms of the financing documents governing our Credit Agreement restrict our current and future operations, particularly our ability to respond to changes or to take certain actions.
The financing documents governing our Credit Agreement contain a number of restrictive covenants that impose significant operating and financial restrictions on us and may limit our ability to engage in acts that may be in our long-term best interests, including restrictions on our ability to:
•incur additional indebtedness;
•incur liens;
•merge, dissolve, liquidate, amalgamate, consolidate or sell all or substantially all of our assets;
•declare or pay certain dividends, payments or distribution or repurchase or redeem certain capital stock;
•permit our subsidiaries to enter into agreements restricting their ability to pay dividends, make loans, incur liens and sell assets; and
•make certain investments.
These restrictions could limit, potentially significantly, our operational flexibility and affect our ability to finance our future operations or capital needs or to execute our business strategy.
Risks Related to Our Organizational Structure
Our principal asset is our interest in Maravai Topco Holdings LLC (“Topco LLC”), and, accordingly, we depend on distributions from Topco LLC to pay our taxes and expenses, including payments under the Tax Receivable Agreement. Topco LLC’s ability to make such distributions may be subject to various limitations and restrictions.
We are a holding company and have no material assets other than our ownership of equity interests in Topco LLC. As such, we have no independent means of generating revenue or cash flow, and our ability to pay our taxes, satisfy our obligations under the Tax Receivable Agreement and pay operating expenses or declare and pay dividends, if any, in the future depends on the financial results and cash flows of Topco LLC and its subsidiaries and distributions we receive from Topco LLC. There can be no assurance that Topco LLC and its subsidiaries will generate sufficient cash flow to distribute funds to us or that applicable state law and contractual restrictions, including negative covenants in debt instruments of Topco LLC and its subsidiaries, will permit such distributions.
Topco LLC is treated as a partnership for U.S. federal income tax purposes and, as such, is not subject to any entity-level U.S. federal income tax. For U.S. federal income tax purposes, taxable income of Topco LLC is allocated to the LLC Unitholders of Topco LLC, including us. Accordingly, we incur income taxes on our distributive share of any net taxable income of Topco LLC. Under the terms of the Topco LLC operating agreement (the “LLC Operating Agreement”), Topco LLC is obligated to make tax distributions to LLC Unitholders, including us. In addition to tax and dividend payments, we also incur expenses related to our operations, including obligations to make payments under the Tax Receivable Agreement. Due to the uncertainty of various factors, we cannot estimate the likely tax benefits we may realize as a result of our purchase of LLC Units in Topco LLC (the “LLC Units”) and LLC Unit exchanges, and the resulting amounts we are likely to pay out to LLC Unitholders pursuant to the Tax Receivable Agreement; however, such payments may be substantial. Under the LLC Operating Agreement, tax distributions shall be made on a pro rata basis among the LLC Unitholders, and will be calculated without regard to any applicable basis adjustment under Section 743(b) of The Internal Revenue Code (“the Code”).
We expect Topco LLC will continue to make cash distributions to the owners of LLC Units in amounts sufficient to (1) fund all or part of their tax obligations in respect of taxable income allocated to them and (2) cover our operating expenses, including payments under the Tax Receivable Agreement.
However, Topco LLC’s ability to make such distributions may be subject to various limitations and restrictions, such as restrictions on distributions that would violate either any contract or agreement to which Topco LLC or its subsidiaries is then a party, including debt agreements, or any applicable law, or that would have the effect of rendering Topco LLC or its subsidiaries insolvent. In addition, effective for taxable years beginning after December 31, 2017, liability for adjustments to a partnership’s tax return may be imputed on the partnership itself in certain circumstances, absent an election to the contrary. Topco LLC may be subject to material liabilities pursuant to this legislation and related guidance if, for example, its calculations of taxable income are incorrect. If we do not have sufficient funds to pay tax or other liabilities or to fund our operations, we may have to borrow funds, which could materially adversely affect our liquidity and financial condition and subject us to various restrictions imposed by any such lenders. To the extent that we are unable to make payments under the Tax Receivable Agreement, such payments generally will be deferred and will accrue interest until paid. Nonpayment for a specified period, however, may constitute a breach of a material obligation under the Tax Receivable Agreement and therefore accelerate payments due under the Tax Receivable Agreement, unless, generally, such nonpayment is due to a lack of sufficient funds.
Payments under the Tax Receivable Agreement will be based on the tax reporting positions we determine. Although we are not aware of any issue that would cause the IRS to challenge existing tax basis, a tax basis increase or other tax attributes subject to the Tax Receivable Agreement, if any subsequent disallowance of tax basis or other benefits were so determined by the IRS, we would not be reimbursed for any payments previously made under the applicable Tax Receivable Agreement (although we would reduce future amounts otherwise payable under such Tax Receivable Agreement). In addition, the actual state or local tax savings we realize may be different than the amount of such tax savings we are deemed to realize under the Tax Receivable Agreement, which will be based on an assumed combined state and local tax rate applied to our reduction in taxable income as determined for U.S. federal income tax purposes as a result of the tax attributes subject to the Tax Receivable Agreement. As a result, payments could be made under the Tax Receivable Agreement in excess of the tax savings we realize in respect of the attributes to which the Tax Receivable Agreement relate.
Conflicts of interest could arise between our shareholders and Maravai Life Sciences Holdings, LLC (“MLSH 1”), which may impede business decisions that could benefit our shareholders.
MLSH 1, which is controlled by GTCR, LLC (“GTCR”) and is the only holder of LLC Units other than us, has the right to consent to certain amendments to the LLC Operating Agreement, as well as to certain other matters. MLSH 1 may exercise these voting rights in a manner that conflicts with the interests of our shareholders. Circumstances may arise in the future when the interests of MLSH 1 conflict with the interests of our shareholders. As we control Topco LLC, we have certain obligations to MLSH 1 as an LLC Unitholder in Topco LLC that may conflict with fiduciary duties our officers and directors owe to our shareholders. These conflicts may result in decisions that are not in the best interests of shareholders.
The Tax Receivable Agreement requires us to make cash payments to MLSH 1 and MLSH 2 in respect of certain tax benefits to which we may become entitled, and we expect that the payments we may be required to make could be substantial.
Pursuant to the Tax Receivable Agreement we are required to make cash payments to MLSH 1 and MLSH 2, collectively, equal to 85% of the tax benefits, if any, that we actually realize, or, in some circumstances, are deemed to realize, as a result of (i) certain increases in the tax basis of assets of Topco LLC and its subsidiaries resulting from purchases or exchanges of LLC Units, (ii) certain tax attributes related to the LLC Units held by the corporations that merged into our corporate structure as part of the Organizational Transactions (as discussed in Note 11 to our consolidated financial statements), Topco LLC and subsidiaries of Topco LLC that existed prior to our initial public offering and (iii) certain other tax benefits related to our entering into the Tax Receivable Agreement, including tax benefits attributable to payments that we make under the Tax
Receivable Agreement. Any payments made by us to MLSH 1 and MLSH 2 under the Tax Receivable Agreement will generally reduce the amount of overall cash flow that might have otherwise been available to us. To the extent that we are unable to make payments under the Tax Receivable Agreement, such payments generally will be deferred and will accrue interest until paid. Nonpayment for a specified period, however, may constitute a breach of a material obligation under the Tax Receivable Agreement and therefore accelerate payments due under the Tax Receivable Agreement, unless, generally, such nonpayment is due to a lack of sufficient funds. Furthermore, our future obligation to make payments under the Tax Receivable Agreement could make us a less attractive target for an acquisition, particularly in the case of an acquirer that cannot use some or all of the tax benefits that may be deemed realized under the Tax Receivable Agreement. The payments under the Tax Receivable Agreement are also not conditioned upon MLSH 1 maintaining a continued ownership interest in Topco LLC.
Estimating the amount and timing of our realization of tax benefits subject to the Tax Receivable Agreement is by its nature imprecise. The actual amount and timing of any payments under the Tax Receivable Agreement will vary depending upon a number of factors, including the timing of exchanges by MLSH 1, the amount of gain recognized by MLSH 1, the amount and timing of the taxable income we generate in the future and the federal tax rates then applicable. Accordingly, estimating the amount and timing of payments that may become due under the Tax Receivable Agreement is also by its nature imprecise.
We expect that the aggregate payments that we may be required to make under the Tax Receivable Agreement may be substantial. Assuming no material changes in the relevant tax law, we expect that probable future payments under the Tax Receivable Agreement relating to the purchase by Maravai LifeSciences Holdings, Inc. of LLC Units from MLSH 1 and the corresponding tax attributes to be approximately $7.1 million. This determination is based on our estimate of taxable income for the year ended December 31, 2023. Future payments in respect of subsequent exchanges or financings and tax attributes relating to the purchase by the Company of LLC Units from MLSH 1 would be in addition to this amount and may be substantial. The foregoing numbers are merely estimates—the actual payments could differ materially. It is possible that future transactions or events could increase or decrease the actual tax benefits realized and the corresponding Tax Receivable Agreement payments. There may be a material negative effect on our liquidity if, as a result of timing discrepancies or otherwise, the payments under the Tax Receivable Agreement exceed the actual benefits we realize in respect of the tax attributes subject to the Tax Receivable Agreement and/or distributions to Maravai LifeSciences Holdings, Inc. by Topco LLC are not sufficient to permit Maravai LifeSciences Holdings, Inc. to make payments under the Tax Receivable Agreement after it has paid taxes.
Payments under the Tax Receivable Agreement will be based on the tax reporting positions that we determine. Although we are not aware of any issue that would cause the Internal Revenue Service (“IRS”) to challenge a tax basis increase or the availability of tax attributes of the corporations merged into our corporate structure as part of the Organizational Transactions, if any, we will not be reimbursed for any cash payments previously made to MLSH 1 and MLSH 2 pursuant to the Tax Receivable Agreement if any tax benefits initially claimed by us are subsequently disallowed, in whole or in part, by the IRS or other applicable taxing authority. For example, if the IRS later asserts that we did not obtain a tax basis increase or disallows (in whole or in part) the availability of Net Operating Losses (“NOLs”) due to a potential ownership change under Section 382 of the Internal Revenue Code (“IRC” or “the Code”), among other potential challenges, then we would not be reimbursed for any cash payments previously made to MLSH 1 and MLSH 2 pursuant to the Tax Receivable Agreement with respect to such tax benefits that we had initially claimed. Instead, any excess cash payments made by us pursuant to the Tax Receivable Agreement will be netted against any future cash payments that we might otherwise be required to make under the terms of the Tax Receivable Agreement. Nevertheless, any tax benefits initially claimed by us may not be disallowed for a number of years following the initial time of such payment or, even if challenged early, such excess cash payment may be greater than the amount of future cash payments that we might otherwise be required to make under the terms of the Tax Receivable Agreement. Accordingly, there may not be sufficient future cash payments against which to net. The applicable U.S. federal income tax rules are complex, and there can be no assurance that the IRS or a court will not disagree with our tax reporting positions. As a result, it is possible that we could make cash payments under the Tax Receivable Agreement that are substantially greater than our actual cash tax savings.
The Tax Receivable Agreement liability is recorded on the consolidated balance sheets as a contingent liability under ASC 450, “Liabilities,” and reflects management’s assessment that positive future taxable income and realization of cash tax savings are probable. Management’s assessment of whether payment of the Tax Receivable Agreement liability is probable is generally based on the determination as to whether it is more likely than not that the deferred tax assets will be realized. We evaluate the realizability of our deferred tax assets on a quarterly basis and establish valuation allowances when it is more likely than not that all or a portion of a deferred tax asset may not be realized. As of December 31, 2023, we established a full valuation allowance against our deferred tax assets and derecognized the remaining non-current liability under the Tax Receivable Agreement after concluding it was not probable that we would generate sufficient future taxable income to utilize deferred tax assets that would result in payments due under the Tax Receivable Agreement. If revised forecasts of our future taxable income or other relevant factors result in us releasing all or a portion of the valuation allowance recorded against the deferred tax assets applicable to the aforementioned tax attributes in a future period, the remaining Tax Receivable Agreement liability may be
considered probable at that time and recorded on the consolidated balance sheet and within earnings. It is impossible to predict when and to what extent, if at all, such valuation allowance will be released, and therefore whether we would again be required to recognize all or a portion of the Tax Receivable Agreement liability, which would adversely impact our future results of operations, possibly in a material manner.
Under the Tax Receivable Agreement, we are required to provide MLSH 1 and MLSH 2 with a schedule setting forth the calculation of payments that are due under the TRA with respect to each taxable year in which a payment obligation arises within ninety (90) days after the extended due date of our U.S. federal income tax return for such taxable year. This calculation will be based upon the advice of our tax advisors. The calculation will become final thirty (30) days after it is provided assuming that no objections are made. Payments under the Tax Receivable Agreement will generally be made within five (5) business days after this schedule becomes final pursuant to the procedures set forth in the Tax Receivable Agreement. Interest on such payments will begin to accrue at a rate of Intercontinental Exchange London Interbank Offer Rate (“LIBOR”) for a period of one month (or, if LIBOR ceases to be published, at a rate selected by us in good faith, with characteristics similar to LIBOR or consistent with market practices generally, any such rate, a “Replacement Rate”) plus 100 basis points from the due date (without extensions) of such tax return. Generally, any late payments that may be made under the Tax Receivable Agreement will continue to accrue interest at LIBOR (or a Replacement Rate, as applicable) plus 500 basis points until such payments are made, including any late payments that we may subsequently make because we did not have enough available cash to satisfy our payment obligations at the time at which they originally arose. Given the cessation of LIBOR, we have transitioned to the Secured Overnight Financing Rate (“SOFR”) as the applicable Replacement Rate as allowable under the Tax Receivable Agreement.
The amounts that we may be required to pay to MLSH 1 and MLSH 2 under the Tax Receivable Agreement may be accelerated in certain circumstances and may also significantly exceed the actual tax benefits that we ultimately realize.
The Tax Receivable Agreement provides that if (1) certain mergers, asset sales, other forms of business combination or other changes of control were to occur, (2) we breach any of our material obligations under the Tax Receivable Agreement or (3) at any time, we elect an early termination of the Tax Receivable Agreement, then the Tax Receivable Agreement will terminate and our obligations, or our successor’s obligations, to make payments under the Tax Receivable Agreement would accelerate and become immediately due and payable. The amount due and payable in that circumstance is based on certain assumptions, including an assumption that we would have sufficient taxable income to fully utilize all potential future tax benefits that are subject to the Tax Receivable Agreement. We may need to incur debt to finance payments under the Tax Receivable Agreement to the extent our cash resources are insufficient to meet our obligations under the Tax Receivable Agreement as a result of timing discrepancies or otherwise.
As a result of a change in control, material breach or our election to terminate the Tax Receivable Agreement early, (1) we could be required to make cash payments to MLSH 1 and MLSH 2 that are greater than the specified percentage of the actual benefits we ultimately realize in respect of the tax benefits that are subject to the Tax Receivable Agreement and (2) we would be required to make an immediate cash payment equal to the anticipated future tax benefits that are the subject of the Tax Receivable Agreement discounted in accordance with the Tax Receivable Agreement, which payment may be made significantly in advance of the actual realization, if any, of such future tax benefits. In these situations, our obligations under the Tax Receivable Agreement could have a substantial negative impact on our liquidity and could have the effect of delaying, deferring or preventing certain mergers, asset sales, other forms of business combination, or other changes of control. There can be no assurance that we will be able to finance our obligations under the Tax Receivable Agreement.
Our organizational structure, including the Tax Receivable Agreement, confers certain benefits upon MLSH 1 and MLSH 2 that will not benefit the other common shareholders to the same extent as they will benefit MLSH 1 and MLSH 2.
Our organizational structure, including the Tax Receivable Agreement, confers certain benefits upon MLSH 1, as the only other LLC Unitholder in Topco LLC, and MLSH 2 that will not benefit the other holders of our Class A common stock to the same extent. We have entered into a Tax Receivable Agreement with MLSH 1 and MLSH 2, which will provide for the payment by us to MLSH 1 and MLSH 2, collectively, of 85% of the amount of tax benefits, if any, that we actually realize, or in some circumstances are deemed to realize, as a result of (i) certain increases in the tax basis of assets of Topco LLC and its subsidiaries resulting from purchases or exchanges of LLC Units, (ii) certain tax attributes of certain of the entities through which GTCR and other existing members of MLSH 1 and MLSH 2 held their ownership interests in MLSH 1, Topco LLC and subsidiaries of Topco LLC that existed prior to our initial public offering and (iii) certain other tax benefits related to our entering into the Tax Receivable Agreement, including tax benefits attributable to payments that we make under the Tax Receivable Agreement. Due to the uncertainty of various factors, we cannot estimate the likely tax benefits we will realize as a result of purchases of LLC Units and LLC Unit exchanges, and the resulting amounts we are likely to pay out to MLSH 1 and MLSH 2 pursuant to the Tax Receivable Agreement; however, we estimate that such payments may be substantial. Although
we will retain 15% of the amount of such tax benefits, this and other aspects of our organizational structure may adversely impact the future trading market for the Class A common stock.
We may not be able to realize all or a portion of the tax benefits that are currently expected to result from the tax attributes covered by the Tax Receivable Agreement and from payments made under the Tax Receivable Agreement.
Our ability to realize the tax benefits that we currently expect to be available as a result of the attributes covered by the Tax Receivable Agreement, the payments made pursuant to the Tax Receivable Agreement, and the interest deductions imputed under the Tax Receivable Agreement all depend on a number of assumptions, including that we earn sufficient taxable income each year during the period over which such deductions are available and that there are no adverse changes in applicable law or regulations. Additionally, if our actual taxable income were insufficient or there were additional adverse changes in applicable law or regulations, we may be unable to realize all or a portion of the expected tax benefits and our cash flows and shareholders’ equity could be negatively affected.
In certain circumstances, Topco LLC will be required to make distributions to us and MLSH 1 and the distributions may be substantial.
Topco LLC is treated as a partnership for U.S. federal income tax purposes and, as such, is not subject to U.S. federal income tax. Instead, taxable income is allocated to its members, including us. We expect Topco LLC will continue to make tax distributions quarterly to the LLC Unitholders in Topco LLC (including us), in each case on a pro rata basis based on Topco LLC’s net taxable income and without regard to any applicable basis adjustment under Section 743(b) of the Code. Funds used by Topco LLC to satisfy its tax distribution obligations will not be available for reinvestment in our business. Moreover, these tax distributions may be substantial, and will likely exceed (as a percentage of Topco LLC’s income) the overall effective tax rate applicable to a similarly situated corporate taxpayer. As a result, it is possible that we will receive distributions significantly in excess of our tax liabilities and obligations to make payments under the Tax Receivable Agreement. While our Board may choose to distribute such cash balances as dividends on our Class A common stock, they will not be required to do so, and may in their sole discretion choose to use such excess cash for any purpose (including an investment of such cash into Topco LLC) depending upon the facts and circumstances at the time of determination. See “Dividend Policy.”
Unanticipated changes in effective tax rates or adverse outcomes resulting from examination of our income or other tax returns could adversely affect our operating results and financial condition.
We are subject to income taxes in the U.S. and certain foreign jurisdictions. Our tax liabilities will be subject to the allocation of expenses in differing jurisdictions. Our future effective tax rates could be subject to volatility or adversely affected by a number of factors, including:
•changes in the amount and realizability of our deferred tax assets and liabilities;
•changes in any tax valuation allowances;
•expiration of, or detrimental changes in, research and development tax credit laws; or
•changes in tax laws, regulations or interpretations thereof.
In addition, we may be subject to audits of our income, sales and other transaction taxes by U.S. federal, state and foreign authorities. Outcomes from these audits could have an adverse effect on our operating results and financial condition.
If we were deemed to be an investment company under the 1940 Act, applicable restrictions could make it impractical for us to continue our business as contemplated and could have a material adverse effect on our business, financial condition, results of operations, cash flows and prospects.
Under Sections 3(a)(1)(A) and (C) of the 1940 Act, a company generally will be deemed to be an “investment company” for purposes of the 1940 Act if it (1) is, or holds itself out as being, engaged primarily, or proposes to engage primarily, in the business of investing, reinvesting or trading in securities or (2) is engaged, or proposes to engage, in the business of investing, reinvesting, owning, holding or trading in securities and it owns or proposes to acquire investment securities having a value exceeding 40% of the value of its total assets (exclusive of U.S. government securities and cash items) on an unconsolidated basis. We do not believe that we are an “investment company,” as such term is defined in either of those sections of the 1940 Act.
As the sole managing member of Topco LLC, we will control and manage Topco LLC. On that basis, we believe that our interest in Topco LLC is not an “investment security” under the 1940 Act. Therefore, we have less than 40% of the value of our total assets (exclusive of U.S. government securities and cash items) in “investment securities.” However, if we were to lose the
right to manage and control Topco LLC, interests in Topco LLC could be deemed to be “investment securities” under the 1940 Act.
We intend to conduct our operations so that we will not be deemed to be an investment company. However, if we were deemed to be an investment company, restrictions imposed by the 1940 Act, including limitations on our capital structure and our ability to transact with affiliates, could make it impractical for us to continue our business as contemplated and could have a material adverse effect on our business, financial condition, results of operations, cash flows and prospects.
Risks Related to Being a Public Company
We are obligated to develop and maintain proper and effective internal control over financial reporting in order to comply with Section 404 of the Sarbanes-Oxley Act. We may not complete our analysis of our internal control over financial reporting in a timely manner, or to the extent these internal controls are determined by us or our auditors to not be operating effectively, investor confidence in us and the value of our Class A common stock could be adversely affected.
Our management is responsible for establishing and maintaining adequate internal control over financial reporting. Internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements in accordance with GAAP. Developing the system and processing documentation necessary to perform the evaluation needed to comply with Section 404 of the Sarbanes-Oxley Act is a costly and challenging process. If we are unable to assert that our internal control over financial reporting is effective, investors could lose confidence in the accuracy and completeness of our financial reports, which could cause the price of our Class A common stock to decline, and we may be subject to investigation or sanctions by the SEC.
We are required, pursuant to Section 404 of the Sarbanes-Oxley Act, to furnish a report by management on, among other things, the effectiveness of our internal control over financial reporting. This assessment must be made yearly and must include disclosure of any material weaknesses identified by our management in our internal control over financial reporting. We also must disclose changes made in our internal control and procedures on a quarterly basis. Further, our independent registered public accounting firm must report on the effectiveness of our internal control over financial reporting pursuant to Section 404 of the Sarbanes-Oxley Act. Our independent registered public accounting firm may issue a report that is adverse in the event it is not satisfied with the level at which our controls are documented, designed or operating, which could cause the price of our Class A common stock to decline, and we may be subject to investigation or sanctions by the SEC.
Additionally, the existence of a material weakness or significant deficiency would require management to devote significant time and incur significant expense to remediate any such material weaknesses or significant deficiencies and management may not be able to remediate any such material weaknesses or significant deficiencies in a timely manner. The existence of any material weakness in our internal control over financial reporting could also result in errors in our financial statements that could require us to restate our financial statements, cause us to fail to meet our reporting obligations and cause shareholders to lose confidence in our reported financial information, all of which could materially and adversely affect our business and stock price.
Risks Related to Our Class A Common Stock
GTCR controls us, and its interests may conflict with ours or yours in the future.
As of December 31, 2023, investment entities affiliated with GTCR collectively controlled approximately 56% of the voting power of our outstanding common stock and therefore GTCR controls the outcome of all matters submitted to a vote of our shareholders. This control enables GTCR to control the election of the members of the Board and all other corporate decisions. Even when GTCR ceases to control a majority of the total voting power, for so long as GTCR continues to own a significant percentage of our Class A common stock, GTCR will still be able to significantly influence the composition of our Board and the approval of actions requiring shareholder approval. Accordingly, for such period of time, GTCR will have significant influence with respect to our management, business plans and policies, including the appointment and removal of our officers, decisions on whether to raise future capital and amending our charter and bylaws, which govern the rights attached to our Class A common stock. In particular, for so long as GTCR continues to own a significant percentage of our Class A common stock, GTCR will be able to cause or prevent a change of control of us or a change in the composition of our Board and could preclude any unsolicited acquisition of us. The concentration of ownership could deprive you of an opportunity to receive a premium for your shares of Class A common stock as part of a sale of us and ultimately might affect the market price of our Class A common stock.
We entered into a Director Nomination Agreement with GTCR that provides GTCR the right to nominate to the Board a number of designees equal to at least: (i) 100% of the total number of directors comprising the Board, so long as GTCR
beneficially owns shares of Class A common stock and Class B common stock representing at least 40% of the total amount of shares of Class A common stock and Class B common stock it beneficially owned as of November 19, 2020, (ii) 40% of the total number of directors, in the event that GTCR beneficially owns shares of Class A common stock and Class B common stock representing at least 30% but less than 40% of the total amount of shares of Class A common stock and Class B common stock it owned as of November 19, 2020, (iii) 30% of the total number of directors, in the event that GTCR beneficially owns shares of Class A common stock and Class B common stock representing at least 20% but less than 30% of the total amount of shares of Class A common stock and Class B common stock it owned as of November 19, 2020, (iv) 20% of the total number of directors, in the event that GTCR beneficially owns shares of Class A common stock and Class B common stock representing at least 10% but less than 20% of the total amount of shares of Class A common stock and Class B common stock it owns as of November 19, 2020 and (v) one director, in the event that GTCR beneficially owns shares of Class A common stock and Class B common stock representing at least 5% of the total amount of shares of Class A common stock and Class B common stock it owned as of November 19, 2020. The Director Nomination Agreement provides that GTCR may assign such right to a GTCR affiliate. The Director Nomination Agreement prohibits us from increasing or decreasing the size of our Board without the prior written consent of GTCR.
GTCR and its affiliates engage in a broad spectrum of activities, including investments in our industry generally. In the ordinary course of their business activities, GTCR and its affiliates may engage in activities where their interests conflict with our interests or those of our other shareholders, such as investing in or advising businesses that directly or indirectly compete with certain portions of our business or are suppliers or customers of ours. Our certificate of incorporation provides that none of GTCR, any of its affiliates or any director who is not employed by us (including any non-employee director who serves as one of our officers in both his or her director and officer capacities) or its affiliates has any duty to refrain from engaging, directly or indirectly, in the same business activities or similar business activities or lines of business in which we operate. GTCR also may pursue acquisition opportunities that may be complementary to our business, and, as a result, those acquisition opportunities may not be available to us. In addition, GTCR may have an interest in pursuing acquisitions, divestitures and other transactions that, in its judgment, could enhance its investment, even though such transactions might involve risks to you or may not prove beneficial.
We are a “controlled company” within the meaning of the rules of NASDAQ and, as a result, we qualify for and rely on exemptions from certain corporate governance requirements. You will not have the same protections as those afforded to shareholders of companies that are subject to such governance requirements.
Currently, GTCR controls a majority of the voting power of our outstanding common stock. As a result, we are a “controlled company” within the meaning of the corporate governance requirements of NASDAQ. Under these rules, a company of which more than 50% of the voting power for the election of directors is held by an individual, group or another company is a “controlled company” and may elect not to comply with certain corporate governance requirements of NASDAQ, including:
•the requirement that a majority of our Board is composed of “independent directors” as defined under NASDAQ rules;
•the requirement that we have a nominations committee that is composed entirely of independent directors; and
•the requirement that we have a compensation committee that is composed entirely of independent directors.
From time to time, we may rely on these exceptions. Although a majority of our Board is currently composed of independent directors, neither our Compensation and Leadership Development Committee, nor our Nominating, Governance and Risk Committee, consists entirely of independent directors. Accordingly, you may not have the same protections afforded to shareholders of companies that are subject to all of the corporate governance requirements of NASDAQ.
Provisions of our corporate governance documents could make an acquisition of us more difficult and may prevent attempts by our shareholders to replace or remove our current management, even if beneficial to our shareholders.
Our certificate of incorporation and bylaws and the Delaware General Corporation Law (the “DGCL”) contain provisions that could make it more difficult for a third party to acquire us, even if doing so might be beneficial to our shareholders. Among other things:
•these provisions allow us to authorize the issuance of undesignated preferred stock, the terms of which may be established and the shares of which may be issued without shareholder approval, and which may include supermajority voting, special approval, dividend, or other rights or preferences superior to the rights of shareholders;
•these provisions provide for a classified board of directors with staggered three-year terms;
•these provisions provide that, at any time when GTCR controls, in the aggregate, less than 40% of the outstanding shares of our Class A common stock, directors may only be removed for cause, and only by the affirmative vote of
holders of at least 66 2⁄3% in voting power of all the then-outstanding shares of our stock entitled to vote thereon, voting together as a single class;
•these provisions prohibit shareholder action by written consent from and after the date on which GTCR controls, in the aggregate, less than 35% in voting power of our stock entitled to vote generally in the election of directors;
•these provisions provide that for as long as GTCR controls, in the aggregate, at least 50% in voting power of our stock entitled to vote generally in the election of directors, any amendment, alteration, rescission or repeal of our bylaws by our shareholders will require the affirmative vote of a majority in voting power of the outstanding shares of our capital stock and at any time when GTCR controls, in the aggregate, less than 50% in voting power of all outstanding shares of our stock entitled to vote generally in the election of directors, any amendment, alteration, rescission or repeal of our bylaws by our shareholders will require the affirmative vote of the holders of at least 66 2⁄3% in voting power of all the then-outstanding shares of our stock entitled to vote thereon, voting together as a single class; and
•these provisions establish advance notice requirements for nominations for elections to our Board or for proposing matters that can be acted upon by shareholders at shareholder meetings; provided, however, at any time when GTCR controls, in the aggregate, at least 10% in voting power of our stock entitled to vote generally in the election of directors, such advance notice procedure will not apply to GTCR.
We opted out of Section 203 of the DGCL, which generally prohibits a Delaware corporation from engaging in any of a broad range of business combinations with any interested shareholder for a period of three years following the date on which the shareholder became an interested shareholder. However, our certificate of incorporation contains a provision that provides us with protections similar to Section 203, and prevents us from engaging in a business combination with a person (excluding GTCR and any of its direct or indirect transferees and any group as to which such persons are a party) who acquires at least 85% of our Class A common stock for a period of three years from the date such person acquired such common stock, unless board or shareholder approval is obtained prior to the acquisition. These provisions could discourage, delay or prevent a transaction involving a change in control of our company. These provisions could also discourage proxy contests and make it more difficult for you and other shareholders to elect directors of your choosing and cause us to take other corporate actions you desire, including actions that you may deem advantageous, or negatively affect the trading price of our Class A common stock. In addition, because our Board is responsible for appointing the members of our management team, these provisions could in turn affect any attempt by our shareholders to replace current members of our management team.
These and other provisions in our certificate of incorporation, bylaws and Delaware law could make it more difficult for shareholders or potential acquirers to obtain control of our Board or initiate actions that are opposed by our then-current Board, including actions to delay or impede a merger, tender offer or proxy contest involving our company. The existence of these provisions could negatively affect the price of our Class A common stock and limit opportunities for you to realize value in a corporate transaction.
Our certificate of incorporation designates the Court of Chancery of the State of Delaware as the exclusive forum for certain litigation that may be initiated by our shareholders and the federal district courts of the United States as the exclusive forum for litigation arising under the Securities Act, which could limit our shareholders’ ability to obtain a favorable judicial forum for disputes with us.
Pursuant to our certificate of incorporation, unless we consent in writing to the selection of an alternative forum, the Court of Chancery of the State of Delaware is the sole and exclusive forum for any claims in state court for (1) any derivative action or proceeding brought on our behalf, (2) any action asserting a claim of breach of a fiduciary duty owed by any of our directors, officers or other employees to us or our shareholders, (3) any action asserting a claim against us arising pursuant to any provision of the DGCL, our certificate of incorporation or our bylaws or (4) any other action asserting a claim against us that is governed by the internal affairs doctrine; provided that for the avoidance of doubt, the forum selection provision that identifies the Court of Chancery of the State of Delaware as the exclusive forum for certain litigation, including any “derivative action,” will not apply to suits to enforce a duty or liability created by the Securities Act of 1933, as amended (the “Securities Act”), the Securities Exchange Act of 1934, as amended (the “Exchange Act”) or any other claim for which the federal courts have exclusive jurisdiction. Our certificate of incorporation also provides that, unless we consent in writing to the selection of an alternative forum, the federal district courts of the United States shall be the exclusive forum for the resolution of any complaint asserting a cause of action arising under the Securities Act. Our certificate of incorporation further provides that any person or entity purchasing or otherwise acquiring any interest in shares of our capital stock is deemed to have notice of and consented to the provisions of our certificate of incorporation described above. The forum selection provisions in our certificate of incorporation may have the effect of discouraging lawsuits against us or our directors and officers and may limit our shareholders’ ability to obtain a favorable judicial forum for disputes with us. If the enforceability of our forum selection provisions were to be challenged, we may incur additional costs associated with resolving such challenge. While we currently have no basis to expect any such challenge would be successful, if a court were to find our forum selection provisions to be
inapplicable or unenforceable with respect to one or more of these specified types of actions or proceedings, we may incur additional costs associated with having to litigate in other jurisdictions, which could have an adverse effect on our business, financial condition, results of operations, cash flows and prospects and result in a diversion of the time and resources of our employees, management and board of directors.
Our stock price may be volatile for reasons unrelated to our operating performance and financial condition.
Capital markets worldwide have experienced, and are likely to continue to experience, significant volatility, including as a result of the current challenging macroeconomic environment, fiscal and monetary policy uncertainty, as well as political instability and continued or worsening hostilities or military conflicts in certain regions. This market volatility, as well as other general economic, market or political conditions, could subject the trading price of our Class A common stock to wide price fluctuations regardless of our underlying operating performance and financial position.
If our existing investors sell a significant portion of our total outstanding shares of Class A common stock, the market price of our Class A common stock could drop significantly, even if our business is doing well.
Sales of a substantial number of shares of our Class A common stock in the public market could occur at any time. As of December 31, 2023, we had 132,227,632 outstanding shares of Class A common stock, 21,681,033 of which held by MLSH 2, and further, as of December 31, 2023, an additional 119,094,026 shares of Class A common stock are issuable upon the exchange by MLSH 1 of its interest in Topco. Because each of MLSH 1 and MLSH 2 is controlled by GTCR and is considered an “affiliate” of ours, the shares of Class A common stock held by MLSH 1 and MLSH 2 are subject to certain restrictions on resale imposed by U.S. federal securities laws. However, pursuant to a registration rights agreement, MLSH 1 and MLSH 2 have the right to request that we register these shares in which case the shares would be able to be freely sold in the public market without such restrictions. These sales, or the perception in the market that the holders of a large number of shares of Class A common stock intend to sell shares, could reduce the market price of our Class A common stock.
Because we have no current plans to pay regular cash dividends on our Class A common, you may not receive any return on investment unless you sell your Class A common stock for a price greater than that which you paid for it.
We do not anticipate paying any regular cash dividends on our Class A common stock. Any decision to declare and pay dividends in the future will be made at the discretion of our Board and will depend on, among other things, our results of operations, financial condition, cash requirements, contractual restrictions and other factors that our Board may deem relevant. In addition, our ability to pay dividends is, and may be, limited by covenants of existing and any future outstanding indebtedness we or our subsidiaries incur. Therefore, any return on investment in our Class A common stock is solely dependent upon the appreciation of the price of our Class A common stock on the open market, which may not occur.
We may issue shares of preferred stock in the future, which could make it difficult for another company to acquire us or could otherwise adversely affect holders of our Class A common stock, which could depress the price of our Class A common stock.
Our certificate of incorporation authorizes us to issue one or more series of preferred stock. Our Board has the authority to determine the preferences, limitations and relative rights of the shares of preferred stock and to fix the number of shares constituting any series and the designation of such series, without any further vote or action by our shareholders. Our preferred stock could be issued with voting, liquidation, dividend and other rights superior to the rights of our Class A common stock. The potential issuance of preferred stock may delay or prevent a change in control of us, discouraging bids for our Class A common stock at a premium to the market price, and materially adversely affect the market price and the voting and other rights of the holders of our Class A common stock.
Item 1B. Unresolved Staff Comments
None.
Item 1C. Cybersecurity
Risk Management and Strategy
Our cybersecurity risk management processes include technical security controls, policy enforcement mechanisms, monitoring systems, contractual arrangements, tools and related services, and management oversight to assess, identify and manage risks from cybersecurity threats. We implement risk-based controls to protect our information, information systems, business operations, and products and related services. We have adopted security-control principles based on the National Institute of
Standards and Technology Cybersecurity Framework (NIST), other global standards, and contractual requirements, as applicable. We also leverage government partnerships, industry and government associations, third-party benchmarking, audits, threat intelligence feeds, and other similar resources to inform our cybersecurity efforts and allocate resources.
We maintain an information security program that includes physical, administrative and technical safeguards, and we maintain plans and procedures whose objective is to help us prevent and timely and effectively respond to cybersecurity incidents. Through our cybersecurity risk management process, we continuously monitor cybersecurity vulnerabilities and potential attack vectors and evaluate the potential operational and financial effects of cybersecurity risk countermeasures made to defend against such threats. This process has been integrated into our Enterprise Risk Management program and our Compliance Risk Management program, both of which are overseen by our Board of Directors. In addition, we engage third-party consultants to assist us in assessing, enhancing, implementing, and monitoring our cybersecurity risk management programs, including conducting penetration testing, phishing campaigns, and vulnerability assessments, and responding to any incidents.
We also assess the risks from cybersecurity threats of our suppliers and third-party service providers. We also require our suppliers and third-party service providers to adopt security-control principles based on NIST or similar global standards.
We have experienced, and may in the future experience, whether directly or through our supply chain or other channels, cybersecurity incidents. While prior incidents have not had a material impact on us, future incidents could have a material impact on our business, operations, and reputation. Although our cybersecurity risk management processes are designed to help prevent, detect, respond to, and mitigate the impact of such incidents, there is no guarantee that they will be sufficient to prevent or mitigate the risk of a cyberattack or the potentially serious reputational, operational, legal or financial impacts that may result. See “Our internal computer systems, or those of our customers, collaborators or other contractors, have been and may in the future be subject to cyber-attacks or security breaches, which could result in a material disruption of our product development programs or otherwise adversely affect our business, financial condition, results of operations, cash flows and prospects” within Item 1A, “Risk Factors” in this Annual Report on Form 10-K.
Governance
Our Board has overall responsibility for risk oversight. Oversight of certain of the Company’s key risks is specifically allocated to Board committees based on their respective areas of expertise. The Nominating, Governance and Risk Committee assists the Board in overseeing risks specific to cybersecurity. Pursuant to its written charter, the Nominating, Governance and Risk Committee is charged with overseeing our management’s efforts to identify, evaluate and mitigate major risks related to cybersecurity, data protection controls, business continuity/disaster recovery systems and other information security matters, and periodically reviews our approach to the identification, evaluation and mitigation of such risks with the Board. Our Vice President, Information Technology (“VP of IT”), together with our General Counsel, briefs the Nominating, Governance and Risk Committee on cybersecurity risks at selected meetings. These briefings include assessments of the threat landscape, updates on incidents, and reports on our investments in cybersecurity risk mitigation and governance. To the extent that a significant cybersecurity event occurs, the Nominating, Governance and Risk Committee would also receive periodic updates from senior management, including the VP of IT, the General Counsel, and the relevant Company third-party consultants on any significant cybersecurity events. Such updates would include, as applicable and relevant, the nature, scope and timing of the event; the type and scale of information or data has been accessed, exfiltrated or encrypted; the systems involved; what is known about the threat actor, such as capabilities and demands, if any; management’s ongoing assessment of the impacts or likely impacts of the intrusion; the possibility of litigation or regulatory investigations or actions; and any other information that management finds relevant and that would aid in the assessment of the materiality of the impact of the intrusion.
Our Information Technology (IT) Department and Legal Department work together and are jointly responsible for developing and coordinating our enterprise-wide cybersecurity policy and strategy, including managing our cybersecurity risk management processes. The VP of IT and the General Counsel report to the Company’s senior leadership team on progress towards specific cybersecurity objectives.
Vijay Mani is our VP of IT. He is responsible for managing our information security, developing cybersecurity strategy, and implementing effective information and cybersecurity programs. Mr. Mani has 16 years of experience working in leadership roles in information technology, as well as relevant degrees and certifications, including an Advanced Computer Security Certificate from Stanford University. He reports directly to our Chief Financial Officer and meets periodically with the Nominating, Governance and Risk Committee.
Item 2. Properties
Our corporate headquarters and certain of our research and development operations are located in San Diego, California. The facilities serve as the principal hub of operations for our nucleic acid production business and were purpose built to expand the capacity of this business segment while adding specialized capabilities in the form of clean rooms, air handling, waste and
solvent handling, and GMP capabilities. Our facility leases expire at varying dates through 2038, not including renewals that are at our option.
All facilities are leased. A summary of our facilities is listed below.
| | | | | | | | | | | | | | |
| Location | | Approx. Square Footage | | Segment |
| San Diego, CA | | 237,000 | | Nucleic Acid Production |
| Sterling, VA | | 21,000 | | Nucleic Acid Production |
| Leland, NC | | 46,000 | | Biologics Safety Testing |
| Southport, NC | | 20,000 | | Biologics Safety Testing |
Jupiter, FL | | 17,000 | | Nucleic Acid Production |
Item 3. Legal Proceedings
From time to time, we may be involved in various legal proceedings and subject to claims that arise in the ordinary course of business. Although the results of litigation and claims are inherently unpredictable and uncertain, we are not currently a party to any legal proceedings the outcome of which, if determined adversely to us, are believed to, either individually or taken together, have a material adverse effect on our business, operating results, cash flows or financial condition. Regardless of the outcome, litigation has the potential to have an adverse impact on us because of defense and settlement costs, diversion of management resources, and other factors. See Item 1A. “Risk Factors—Risks Related to Our Intellectual Property—Intellectual property litigation and other proceedings could cause us to spend substantial resources and distract our personnel from their normal responsibilities” and “Risk Factors—Risks Related to Our Intellectual Property—If we are sued for infringing, misappropriating, or otherwise violating intellectual property rights of third parties, such litigation could be costly and time consuming and could prevent or delay us from developing or commercializing our current or future products.”
Item 4. Mine Safety Disclosures
Not applicable.
Part II.
Item 5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities
Market Information
Our Class A common stock trades on The Nasdaq Global Select Market under the symbol “MRVI.”
Our Class B common stock is not listed nor traded on any stock exchange.
Stock Performance Graph
The following graph shows the total stockholder’s return on an investment of $100 in cash at market close on November 20, 2020 (the first day of trading of our common stock), through December 31, 2023 for (i) our Class A common stock, (ii) the Nasdaq Composite Index and (iii) the Nasdaq Biotechnology Index. Pursuant to applicable Securities and Exchange Commission rules, all values assume reinvestment of pre-tax amount of all dividends; however, no dividends have been declared on our Class A common stock to date. The stockholder return shown in the graph below may not be indicative of future stock price performance, and we do not make or endorse any predictions as to future stockholder return. This graph shall not be deemed “soliciting material” or be deemed “filed” for purposes of Section 18 of the Exchange Act, or otherwise subject to the liabilities under that Section, and shall not be deemed to be incorporated by reference into any of our filings under the Securities Act whether made before or after the date hereof and irrespective of any general incorporation language in any such filing.
Holders of Common Stock
As of February 21, 2024, there were two holders of record of our Class A common stock. This number does not include a greater number of beneficial holders of our Class A common stock whose shares are held by clearing houses, banks, brokers and other financial institutions which are aggregated into a single holder of record.
As of February 21, 2024, there was one holder of record of our Class B common stock.
Dividend Policy
We currently intend to retain all available funds and any future earnings to fund the development and growth of our business and to repay indebtedness and, therefore, we do not anticipate paying any cash dividends in the foreseeable future. Additionally,
because we are a holding company, our ability to pay dividends on our Class A common stock may be limited by restrictions on the ability of our subsidiaries to pay dividends or make distributions to us. Any future determination to pay dividends will be at the discretion of our Board, subject to compliance with covenants in current and future agreements governing our and our subsidiaries’ indebtedness, including our Credit Agreement (the “Credit Agreement”) entered into in October 2020, and will depend on our results of operations, financial conditions, capital requirements and other factors that our Board deems relevant.
Equity Compensation Plan Information
The following table sets forth information as of December 31, 2023 regarding shares of our Class A common stock that may be issued under the Company’s equity compensation plans, consisting of our 2020 Omnibus Incentive Plan (the “2020 Plan”) and our 2020 Employee Stock Purchase Plan (the “ESPP”).
| | | | | | | | | | | | | | | | | | | | |
| Plan Category | | Number of securities to be issued upon exercise of outstanding options, warrants and rights (a) | | Weighted-average exercise price of outstanding options, warrants and rights | | Number of securities available for future issuance under equity compensation plans (excluding securities reflected in column (a)) |
Equity compensation plans approved by security holders (1) (2) | | 8,683,194 | | $ | 9.76 | | | 57,526,943 |
| Total | | 8,683,194 | | $ | 9.76 | | | 57,526,943 |
____________________
(1)Includes 10,482,025 shares that remain available for purchase under the 2020 Employee Stock Purchase Plan and 56,107,859 shares of common stock that remain available for grant under the 2020 Omnibus Incentive Plan. The 2020 Omnibus Incentive Plan provides for an automatic increase in the number of shares reserved for issuance thereunder on January 1 of each calendar year during the term of the Plan, equal to the lesser of (a) 4.0% of the aggregate number of shares and shares of Class B common stock outstanding on the final day of the immediately preceding calendar year and (b) such smaller number of shares as determined by the Board. The 2020 Employee Stock Purchase Plan also provides for an automatic increase in the number of shares reserved for issuance thereunder on January 1 of each calendar year during the term of the plan, equal to the lesser of (a) 1.25% of the aggregate number of shares and shares of Class B common stock outstanding on the final day of the immediately preceding calendar year and (b) such smaller number of shares as is determined by the Board, provided that the shares reserved under the ESPP shall not exceed an aggregate of 10,948,877 shares.
(2)The weighted average exercise price includes restricted stock unit and performance stock unit awards that can be exercised for no consideration. The weighted average exercise price excluding these restricted stock units and performance stock units is $20.55.
Item 6. Reserved
Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations
You should read the following discussion and analysis of financial condition and results of operations together with our audited consolidated financial statements and related notes included elsewhere in this Annual Report on Form 10-K. This discussion and analysis reflects our historical consolidated results of operations and financial position, and contain forward-looking statements that involve risks and uncertainties. Our actual results could differ materially from those discussed in or implied by these forward-looking statements. Factors that could cause or contribute to such differences include, but are not limited to, those discussed in the section titled “Risk Factors.” Please also see the section titled “Forward Looking Statements.” We were incorporated in August 2020 and, pursuant to the organizational transactions described in Note 1 to our consolidated financial statements, became a holding company whose principal asset is a controlling equity interest in Topco LLC. As the sole managing member of Topco LLC, we operate and control the business and affairs of Topco LLC and its subsidiaries. Accordingly, we consolidate Topco LLC in our consolidated financial statements and report a non-controlling interest related to the portion of Topco LLC not owned by us. Because the organizational transactions were considered transactions between entities under common control, the consolidated financial statements for periods prior to the organizational transactions and the initial public offering have been adjusted to combine the previously separate entities for presentation purposes. Unless otherwise noted or the context otherwise requires, references in this Annual Report on Form 10-K to “we,” “us” or “our” refer to Maravai LifeSciences Holdings, Inc. and its subsidiaries.
This discussion and analysis generally addresses 2023 and 2022 items and year-over-year comparisons between 2023 and 2022. Discussions of 2021 items and year-over-year comparisons between 2022 and 2021 that are not included in this Annual Report on Form 10-K can be found in Part II, Item 7 of our 2022 Annual Report on Form 10-K filed with the SEC on February 28, 2023.
Overview
We are a leading life sciences company providing critical products to enable the development of drug therapies, diagnostics, novel vaccines and support research on human diseases. Our customers include the top global biopharmaceutical companies ranked by research and development expenditures according to industry consultants, and many other emerging biopharmaceutical and life sciences research companies, as well as leading academic research institutions and in vitro diagnostics companies. Our products address the key phases of biopharmaceutical development and include complex nucleic acids for diagnostic and therapeutic applications, antibody-based products to detect impurities during the production of biopharmaceutical products, and products to detect the expression of proteins in tissues of various species.
We have and will continue to build a transformative life sciences products company by acquiring businesses and accelerating their growth through capital infusions and industry expertise. Biomedical innovation is dependent on a reliable supply of reagents in the fields of nucleic acid production, biologics safety testing and protein labeling. From inventive startups to the world’s leading biopharmaceutical, vaccine, diagnostics and gene and cell therapy companies, these customers turn to us to solve their complex discovery challenges and help them streamline and scale their supply chain needs beginning from research and development through clinical trials to commercialization.
Our primary customers are biopharmaceutical companies who are pursuing novel research and product development programs. Our customers also include a range of government, academic and biotechnology institutions.
As of December 31, 2023, we employed a team of over 650 employees, approximately 24% of whom have advanced degrees. As of January 5, 2024, following the completion of a reduction in force (as described under “Restructuring” below), we had approximately 570 employees, approximately 25% of whom have advanced degrees.
We primarily utilize a direct sales model for our sales to our customers in North America. Our international sales, primarily in Europe and Asia Pacific, are effected through a combination of third-party distributors as well as via a direct sales model. The percentage of our total revenue derived from customers in North America was 48.8% and 38.4% for the years ended December 31, 2023 and 2022, respectively.
We generated revenue of $288.9 million and $883.0 million for the years ended December 31, 2023 and 2022, respectively.
Total revenue by segment was $224.8 million in Nucleic Acid Production and $64.2 million in Biologics Safety Testing for the year ended December 31, 2023. Total revenue by segment was $813.1 million in Nucleic Acid Production and $69.9 million in Biologics Safety Testing for the year ended December 31, 2022.
We focus a substantial portion of our resources supporting our core business segments. We are actively pursuing opportunities to expand our customer base both domestically and internationally by fostering strong relationships with both existing and new customers and distributors. Our management team has experience working with biopharmaceutical, vaccine, diagnostics and gene and cell therapy companies as well as academic and research scientists. We also intend to continue making investments in our overall infrastructure and business segments to support our growth. We incurred aggregate selling, general, and administrative expenses of $151.4 million and $129.3 million for the years ended December 31, 2023 and 2022, respectively.
Our research and development efforts are geared towards meeting our customers’ needs. We incurred research and development expenses of $17.3 million and $18.4 million for the years ended December 31, 2023 and 2022, respectively. We intend to continue to invest in research and development and new products and technologies to support our customers’ needs for the foreseeable future.
2023 and Recent Developments
Acquisition
In January 2023, we completed the acquisition of Alphazyme, LLC (“Alphazyme”), a privately-held original equipment manufacturer (“OEM”) and provider of custom, scalable, molecular biology enzymes to customers in the genetic analysis and nucleic acid synthesis markets, for a total purchase consideration of $75.3 million. As a result of the acquisition, we own all the outstanding equity interest in Alphazyme. Our consolidated results of operations for the year ended December 31, 2023 include the operating results of Alphazyme from the acquisition date. See Note 2 to our consolidated financial statements for additional information.
Restructuring
In November 2023, the Company implemented a cost realignment plan (the “Cost Realignment Plan”) that included the termination of approximately 15% of the Company’s workforce, the termination of certain leases, and other actions to reduce expenses, all as part of a plan to optimize business operations and match them to current market conditions. The reduction in
force was completed on January 5, 2024, following the end of the sixty-day notification period required by the Worker Adjustment and Retraining Notification Act. The Company expects the remaining actions under the Cost Realignment Plan to be substantially complete during the first quarter of 2024.
During the year ended December 31, 2023, we incurred restructuring costs of $6.5 million, primarily related to severance and other employee-related costs, facility and other exit costs, professional fees and other restructuring costs. We do not expect to incur any additional restructuring costs, however, we expect to recognize a benefit of $1.2 million during the first quarter of 2024 relating to equity award forfeitures associated with employee terminations. We expect the Cost Realignment Plan to yield certain cost savings of approximately $30.0 million annually, of which $23.0 million relates to labor costs. See Note 3 to our consolidated financial statements for additional information.
Trends and Uncertainties
COVID-19 Related Revenue Trends and Uncertainties
Our results of operations and cash flows during each of the years ended December 31, 2022, 2021 and 2020 substantially benefited from the demand for COVID-19 related products and services, including our proprietary CleanCap® analogs and highly modified RNA products, particularly mRNA, which are used by our customers in the production of COVID-19 vaccines. As a result of the general decrease in market demand for COVID-19 related products and services, including the supply and manufacture of COVID-19 vaccines, and in particular, following the end of U.S. federal public health emergency declaration and World Health Organization declaration of the end of the pandemic in early May 2022, we expect to experience further declines in COVID-19 related revenue, as discussed in further detail below.
We estimate that revenue from COVID-19 related products and services represented approximately 21.0% and 67.9% of our total revenues for the years ended December 31, 2023 and 2022, respectively. We believe that the second quarter of 2022 will have represented the highest revenue quarter for revenue attributable to our COVID-19 related products and services, and have experienced substantial declines in COVID-19 related revenue since such quarter as a result of the general market trend of reduced demand for COVID-19 related products and services as the pandemic subsides, including the supply and manufacture of COVID-19 related vaccines, and the World Health Organization declaring an end to the COVID-19 pandemic. We expect further declines in COVID-19 related revenue for these reasons, as well as a result of unused inventory of our products that our customers have on hand. We are currently unable to fully estimate the impact of this unused inventory on our future COVID-19 related revenue, nor are we able to predict when our customers will resume purchasing COVID-19 related products given that our customers generally have not provided us with detailed inventory data. Our longer-term revenue prospects for COVID-19 related products are highly uncertain but are expected to be substantially less than pandemic highs. The factors that could influence longer-term COVID-19 related revenue include: the emergence, duration and intensity of new virus variants; regional resurgences of the virus globally; competition faced by our customers from other COVID-19 vaccine manufacturers and the development and availability of antiviral therapeutic alternatives; the availability and administration of pediatric and booster vaccinations, vaccine supply constraints, vaccine hesitancy and the effectiveness of vaccines against new virus strains; and the U.S. economy and global economy, including impacts resulting from supply chain constraints, labor market shortages and inflationary pressures. This contraction in COVID-19 related demand will significantly decrease our revenue and cash flow, which in turn could have a material adverse impact on our operating results and financial condition in the future.
Other Trends and Uncertainties
While we believe that the long-term trend of biopharmaceutical customers relying on outside parties to provide important inputs and services for their clinical research and manufacturing remains a long-term growth driver for us, we believe that recent industry trends and uncertainties, including changes in our customers’ spending priorities and budgetary policies and practices, which negatively impacted our revenue and operating results in the year ended December 31, 2023, may continue and result in slower growth and/or cause a further decline in our revenues during the year ending December 31, 2024. These trends and uncertainties, which we primarily attribute to lower levels of investment in the research and development funding of early-stage biotechnology companies and declines and uncertainties in the capital markets amidst ongoing negative macroeconomic challenges, has and may continue to cause those companies to take action to conserve capital, resulting in a potential reduction in research and development spending across the markets in which we participate.
Our businesses also continue to see headwinds from a general contraction in economic activity in Asia, particularly in China, which may negatively impact our revenue derived from those markets. See more information under Part I, Item 1. Business.
How We Assess Our Business
We consider a variety of financial and operating measures in assessing the performance of our business. The key measures we use to determine how our business is performing are revenue and Adjusted EBITDA.
Adjusted EBITDA is a non-GAAP financial measure that we define as net (loss) income adjusted for interest, provision for income taxes, depreciation, amortization and equity-based compensation expenses. Adjusted EBITDA reflects further adjustments to eliminate the impact of certain items, including certain non-cash and other items, that we do not consider representative of our ongoing operating performance. We also present Adjusted Free Cash Flow, which is a non-GAAP measure that we define as Adjusted EBITDA less capital expenditures.
Management uses Adjusted EBITDA to evaluate the financial performance of our business and the effectiveness of our business strategies. We present Adjusted EBITDA and Adjusted Free Cash Flow because we believe they are frequently used by analysts, investors and other interested parties to evaluate companies in our industry, and they facilitate comparisons on a consistent basis across reporting periods. Further, we believe they are helpful in highlighting trends in our operating results because they exclude items that are not indicative of our core operating performance. Adjusted EBITDA is also a component of the financial covenant under our credit agreement that governs our ability to access more than $63.0 million in aggregate letters of credit and available borrowings under our revolving credit facility. In addition, if we borrow more than $63.0 million, we are required to maintain a specified net leverage ratio. See “Liquidity and Capital Resources—Sources of Liquidity—Debt Covenants” for a discussion of this financial covenant.
Adjusted EBITDA and Adjusted Free Cash Flow have limitations as analytical tools and you should not consider them in isolation, or as substitutes for analysis of our results as reported under GAAP. We may in the future incur expenses similar to the adjustments in the presentation of Adjusted EBITDA. In particular, we expect to incur meaningful share-based compensation expense in the future. Other limitations include that Adjusted EBITDA and Adjusted Free Cash Flow do not reflect:
•all expenditures or future requirements for capital expenditures or contractual commitments;
•changes in our working capital needs;
•provision for income taxes, which may be a necessary element of our costs and ability to operate;
•the costs of replacing the assets being depreciated, which will often have to be replaced in the future;
•the non-cash component of employee compensation expense; and
•the impact of earnings or charges resulting from matters we consider not to be reflective, on a recurring basis, of our ongoing operations.
In addition, Adjusted EBITDA and Adjusted Free Cash Flow may not be comparable to similarly titled measures used by other companies in our industry or across different industries.
Components of Results of Operations
Revenue
Our revenue consists primarily of product revenue and, to a much lesser extent, service revenue. We generated total consolidated revenue of $288.9 million and $883.0 million for the years ended December 31, 2023 and 2022, respectively, through the following segments: (i) Nucleic Acid Production and (ii) Biologics Safety Testing.
Nucleic Acid Production Segment
Our Nucleic Acid Production segment focuses on the manufacturing and sale of highly modified nucleic acids products to support the needs of customers’ research, therapeutic and vaccine programs. This segment also provides research products for labeling and detecting proteins in cells and tissue samples.
Biologics Safety Testing Segment
Our Biologics Safety Testing segment focuses on manufacturing and selling biologics safety and impurity tests and assay development services that are utilized by our customers in their biologic drug manufacturing activities.
Cost of Revenue
Cost of revenue associated with our products primarily consists of manufacturing related costs incurred in the production process, including personnel and related costs, equity-based compensation expense, inventory write-downs, costs of materials, labor and overhead, packaging and delivery costs and allocated costs, including facilities, information technology, depreciation, and amortization of intangibles. Cost of revenue also includes adjustments for excess, obsolete or expired inventory, and idle capacity. Cost of revenue associated with our services primarily consists of personnel and related costs, equity-based compensation expense, cost of materials and allocated costs, including facilities and information technology costs. Costs of services were not material for the years ended December 31, 2023 and 2022.
Operating Expenses
Selling, General and Administrative
Our selling, general and administrative expenses primarily consist of salaries, benefits and equity-based compensation expense for our employees in our commercial sales functions, marketing, executive, accounting and finance, legal and human resource functions as well as travel expenses, professional services fees, such as consulting, audit, tax and legal fees, general corporate costs and allocated costs, including facilities, information technology and amortization of intangibles.
We expect that our selling, general and administrative expenses will gradually increase in future periods, primarily due to expanding facilities footprint to support anticipated long-term growth in the business, costs incurred in increasing our presence globally, and increases in marketing activities to drive awareness and adoption of our products and services.
Research and Development
Research and development costs primarily consist of salaries, benefits, equity-based compensation expense, outside contracted services, cost of supplies, in-process research and development costs from asset acquisitions and allocated facilities costs for employees engaged in research and development of products and services. We expense all research and development costs in the period in which they are incurred. Payment made prior to the receipt of goods or services to be used in research and development are recognized as prepaid assets until the goods are received or services are rendered.
We expect our research and development costs to increase to support our research and development efforts, including meeting our customers’ needs.
Change in Estimated Fair Value of Contingent Consideration
Change in estimated fair value of contingent consideration consists of fair value adjustments to contingent consideration liabilities associated with completed acquisitions. These adjustments are based on our assessment of the probability of achieving certain revenue thresholds and other probability factors.
Restructuring
Restructuring costs primarily consist of severance and other employee-related costs, facility and other exit costs, professional fees and other restructuring costs resulting from the Cost Realignment Plan.
Other Income (Expense)
Interest Expense
Interest expense consist of interest costs and the related amortization of the debt discount and deferred issuance costs on our outstanding debt. Interest expense also consists of changes in the fair value of our interest rate cap agreement.
Interest Income
Interest income consists of interest earned on our cash balances and short-term investments in money market funds held at financial institutions.
Change in Payable to Related Parties Pursuant to the Tax Receivable Agreement
During the year ended December 31, 2023, we determined that making a payment under the Tax Receivable Agreement for subsequent years was not probable under Accounting Standards Codification 450 - Contingencies as a result of a valuation allowance having been recorded against our deferred tax assets, and therefore, that it is more likely than not that we will not
generate sufficient future taxable income to utilize related tax benefits that would result in a payment under the Tax Receivable Agreement. As a result, we remeasured the non-current portion of the liability due under the Tax Receivable Agreement to zero, as of December 31, 2023, and recorded a corresponding gain on Tax Receivable Agreement liability remeasurement.
Income Tax Expense
As a result of our ownership of LLC Units in Topco LLC, we are subject to U.S. federal, state and local income taxes with respect to our allocable share of any taxable income of Topco LLC and will be taxed at the prevailing corporate tax rates. In addition, we evaluate the realizability of our deferred tax assets on a quarterly basis and establish valuation allowances when it is more likely than not that all or a portion of a deferred tax asset may not be realized. During the year ended December 31, 2023, we recognized a full valuation allowance against our deferred tax assets and recorded a corresponding income tax expense.
Non-Controlling Interests
Non-controlling interests represent the portion of profit or loss, net assets and comprehensive income or loss of our consolidated subsidiaries that is not allocable to the Company based on our percentage of ownership of such entities. Income or loss attributed to the non-controlling interests is based on the LLC Units outstanding during the period and is presented on the consolidated statements of operations. As of December 31, 2023, we held approximately 52.6% of the outstanding LLC Units of Topco LLC, and MLSH 1 held approximately 47.4% of the outstanding LLC Units of Topco LLC.
Results of Operations
The results of operations presented below should be reviewed in conjunction with the consolidated financial statements and notes included elsewhere in this Annual Report on Form 10-K.
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 | | Change |
| (in thousands, except per share data) | | |
| Revenue | $ | 288,945 | | | $ | 883,001 | | | (67.3) | % |
| Operating expenses: | | | | | |
Cost of revenue (1) | 148,743 | | | 168,957 | | | (12.0) | % |
Selling, general and administrative (1) | 151,390 | | | 129,259 | | | 17.1 | % |
Research and development (1) | 17,280 | | | 18,369 | | | (5.9) | % |
| Change in estimated fair value of contingent consideration | (3,286) | | | (7,800) | | | (57.9) | % |
| | | | | |
Restructuring (1) | 6,466 | | | — | | | * |
| Total operating expenses | 320,593 | | | 308,785 | | | 3.8 | % |
(Loss) income from operations | (31,648) | | | 574,216 | | | (105.5) | % |
| Other income (expense), net | 649,384 | | | (22,744) | | | (2955.2) | % |
Income before income taxes | 617,736 | | | 551,472 | | | 12.0 | % |
| Income tax expense | 756,111 | | | 60,809 | | | 1143.4 | % |
| Net (loss) income | $ | (138,375) | | | $ | 490,663 | | | (128.2) | % |
| Net (loss) income attributable to non-controlling interests | (19,346) | | | 270,458 | | | (107.2) | % |
| Net (loss) income attributable to Maravai LifeSciences Holdings, Inc. | $ | (119,029) | | | $ | 220,205 | | | (154.1) | % |
| | | | | |
| Net (loss) income per Class A common share attributable to Maravai LifeSciences Holdings, Inc.: | | | | | |
| Basic | $ | (0.90) | | | $ | 1.67 | | | |
| Diluted | $ | (0.90) | | | $ | 1.67 | | | |
| Weighted average number of Class A common shares outstanding: | | | | | |
| Basic | 131,919 | | | 131,545 | | | |
| Diluted | 131,919 | | | 255,323 | | | |
| Non-GAAP measures: | | | | | |
| Adjusted EBITDA | $ | 65,309 | | | $ | 637,800 | | | |
| Adjusted Free Cash Flow | $ | 12,621 | | | $ | 586,052 | | | |
____________________
*Not meaningful
(1)Includes equity-based compensation expense as follows (in thousands, except percentages):
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 | | Change |
| Cost of revenue | $ | 7,324 | | | $ | 4,192 | | | 74.7 | % |
| Selling, general and administrative | 24,650 | | | 13,349 | | | 84.7 | % |
| Research and development | 2,715 | | | 1,129 | | | 140.5 | % |
Restructuring | (101) | | | — | | | * |
| Total equity-based compensation expense | $ | 34,588 | | | $ | 18,670 | | | 85.3 | % |
Revenue
Consolidated revenue by segment was as follows for the periods presented (in thousands, except percentages):
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Year Ended December 31, | | Percentage of Revenue |
| 2023 | | 2022 | | Change | | 2023 | | 2022 |
| Nucleic Acid Production | $ | 224,769 | | | $ | 813,069 | | | (72.4) | % | | 77.8 | % | | 92.1 | % |
| Biologics Safety Testing | 64,176 | | | 69,932 | | | (8.2) | % | | 22.2 | % | | 7.9 | % |
| Total revenue | $ | 288,945 | | | $ | 883,001 | | | (67.3) | % | | 100.0 | % | | 100.0 | % |
Total revenue was $288.9 million for the year ended December 31, 2023 compared to $883.0 million for the year ended December 31, 2022, representing a decrease of $594.1 million, or 67.3%.
Nucleic Acid Production revenue decreased from $813.1 million for the year ended December 31, 2022 to $224.8 million for the year ended December 31, 2023, representing a decrease of $588.3 million, or 72.4%. The decrease in Nucleic Acid Production was primarily driven by decreased revenue from our proprietary CleanCap analogs as demand decreased from COVID-19 vaccine manufacturers. For the year ended December 31, 2023, we estimate that approximately $60.8 million, or 54.7%, of our $111.1 million CleanCap revenue was a result of customer demand attributable to COVID-19 vaccines or other COVID-19 related commercial products or developmental programs. For the year ended December 31, 2022, we estimate that approximately $599.8 million, or 90.8%, of our $660.5 million CleanCap revenue was a result of customer demand attributable to COVID-19 vaccines or other COVID-19 related commercial products or developmental programs.
Biologics Safety Testing revenue decreased from $69.9 million for the year ended December 31, 2022 to $64.2 million for the year ended December 31, 2023, representing a decrease of $5.8 million, or 8.2%. The decrease was primarily due to an industry-wide weak demand environment and slowdowns in biologics manufacturing, which continued to impact demand for our HCP ELISA kits.
Segment Information
Management has determined that adjusted earnings before interest, tax, depreciation and amortization is the profit or loss measure used to make resource allocation decisions and evaluate segment performance. Adjusted EBITDA assists management in comparing the segment performance on a consistent basis for purposes of business decision-making by removing the impact of certain items that management believes do not directly reflect the core operations and, therefore, are not included in measuring segment performance. We define Adjusted EBITDA as net (loss) income before interest, taxes, depreciation and amortization, certain non-cash items and other adjustments that we do not consider in our evaluation of ongoing operating performance from period to period. Corporate costs, net of eliminations, are managed on a standalone basis and are not allocated to segments.
We do not allocate assets to our reportable segments as they are not included in the review performed by our Chief Operating Decision Maker for purposes of assessing segment performance and allocating resources.
As of December 31, 2023, all of our long-lived assets were located within the United States.
The following schedule includes revenue and adjusted EBITDA for each of our reportable operating segments (in thousands):
| | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 |
| | | |
| Revenue: | | | |
| Nucleic Acid Production | $ | 224,769 | | | $ | 813,076 | |
| Biologics Safety Testing | 64,179 | | | 69,932 | |
| Total reportable segments’ revenue | 288,948 | | | 883,008 | |
| Intersegment eliminations | (3) | | | (7) | |
| Total | $ | 288,945 | | | $ | 883,001 | |
| | | |
| Segment adjusted EBITDA: | | | |
| Nucleic Acid Production | $ | 82,658 | | | $ | 638,337 | |
| Biologics Safety Testing | 46,908 | | | 54,841 | |
| Total reportable segments’ adjusted EBITDA | 129,566 | | | 693,178 | |
| Reconciliation of total reportable segments’ adjusted EBITDA to income before income taxes | | | |
| Amortization | (27,356) | | | (24,269) | |
| Depreciation | (12,898) | | | (7,566) | |
| Interest expense | (45,892) | | | (20,414) | |
| Interest income | 27,727 | | | 2,338 | |
| Corporate costs, net of eliminations | (64,257) | | | (55,378) | |
| Other adjustments: | | | |
| Acquisition contingent consideration | 3,286 | | | 7,800 | |
| Acquisition integration costs | (12,695) | | | (13,362) | |
| | | |
| Equity-based compensation | (34,588) | | | (18,670) | |
| | | |
| | | |
| Merger and acquisition related expenses | (4,392) | | | (2,416) | |
| Financing costs | — | | | (1,078) | |
| Acquisition related tax adjustment | (1,293) | | | (349) | |
| Tax Receivable Agreement liability adjustment | 668,886 | | | (4,102) | |
| Chief Executive Officer transition costs | (28) | | | (2,426) | |
Restructuring costs (1) | (6,567) | | | — | |
| Other | (1,763) | | | (1,814) | |
| Income before income taxes | 617,736 | | | 551,472 | |
| Income tax expense | (756,111) | | | (60,809) | |
Net (loss) income | $ | (138,375) | | | $ | 490,663 | |
___________________
(1)Equity-based compensation benefit of $0.1 million related to forfeited equity awards in connection with the restructuring is included on the equity-based compensation line item.
During the year ended December 31, 2023 and 2022, intersegment revenue was immaterial between the Nucleic Acid Production and Biologics Safety Testing segments. The intersegment sales and the related gross margin on inventory recorded at the end of the period are eliminated for consolidation purposes. Internal selling prices for intersegment sales are consistent with the segment’s normal retail price offered to external parties. There was no commission expense recognized for intersegment sales for the years ended December 31, 2023 and 2022.
Non-GAAP Financial Measures
Adjusted EBITDA
A reconciliation of net (loss) income to Adjusted EBITDA, which is a non-GAAP measure, is set forth below (in thousands):
| | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 |
Net (loss) income | $ | (138,375) | | | $ | 490,663 | |
| Add: | | | |
| Amortization | 27,356 | | | 24,269 | |
| Depreciation | 12,898 | | | 7,566 | |
| Interest expense | 45,892 | | | 20,414 | |
| Interest income | (27,727) | | | (2,338) | |
| Income tax expense | 756,111 | | | 60,809 | |
| EBITDA | 676,155 | | | 601,383 | |
Acquisition contingent consideration (1) | (3,286) | | | (7,800) | |
Acquisition integration costs (2) | 12,695 | | | 13,362 | |
| | | |
Equity-based compensation (3) | 34,588 | | | 18,670 | |
| | | |
| | | |
Merger and acquisition related expenses (4) | 4,392 | | | 2,416 | |
Financing costs (5) | — | | | 1,078 | |
Acquisition related tax adjustment (6) | 1,293 | | | 349 | |
Tax Receivable Agreement liability adjustment (7) | (668,886) | | | 4,102 | |
Chief Executive Officer transition costs (8) | 28 | | | 2,426 | |
Restructuring costs (9) | 6,567 | | | — | |
Other (10) | 1,763 | | | 1,814 | |
| Adjusted EBITDA | $ | 65,309 | | | $ | 637,800 | |
____________________
(1)Refers to the change in the estimated fair value of contingent consideration related to completed acquisitions.
(2)Refers to incremental costs incurred to execute and integrate completed acquisitions, and retention payments in connection with these acquisitions.
(3)Refers to non-cash expense associated with equity-based compensation.
(4)Refers to diligence, legal, accounting, tax and consulting fees incurred associated with acquisitions that were pursued but not consummated.
(5)Refers to transaction costs related to the refinancing of our long-term debt that are not capitalizable.
(6)Refers to non-cash expense associated with adjustments to the carrying value of the indemnification asset recorded in connection with the acquisition of MyChem, LLC (“MyChem”), which was completed in January 2022.
(7)For the year ended December 31, 2023, refers to the adjustment of our Tax Receivable Agreement liability primarily due to remeasuring the non-current portion of the liability to zero as we no longer consider the payments under the agreement to be probable. For the year ended December 31, 2022, refers to the adjustment of our Tax Receivable Agreement liability primarily due to changes in our estimated state apportionment and the corresponding change of our estimated state tax rate.
(8)Refers to legal fees and other costs associated with the Chief Executive Officer leadership transition that occurred during July 2023.
(9)Refers to restructuring costs associated with the Cost Realignment Plan, which was implemented in November 2023. Equity-based compensation benefit of $0.1 million related to forfeited equity awards in connection with the restructuring is included on the equity-based compensation line item.
(10)For the year ended December 31, 2023, refers to severance payments, legal settlement amounts, inventory step-up charges in connection with the acquisition of Alphazyme, certain working capital and other adjustments related to the acquisition of MyChem, and other non-recurring costs. For the year ended December 31, 2022, refers to the loss recognized during the period associated with certain working capital and other adjustments related to the sale of Vector Laboratories, Inc., which was completed in September 2021, and the loss incurred on extinguishment of debt.
Adjusted Free Cash Flow
A reconciliation of Adjusted Free Cash Flow, which is a non-GAAP measure that we define as Adjusted EBITDA less capital expenditures, is set forth below (in thousands):
| | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 |
| Adjusted EBITDA | $ | 65,309 | | | $ | 637,800 | |
Capital expenditures (1) | (52,688) | | | (51,748) | |
| Adjusted Free Cash Flow | $ | 12,621 | | | $ | 586,052 | |
____________________
(1)We define capital expenditures as: (i) purchases of property and equipment which are included in cash flows from investing activities, offset by government funding received; and (ii) construction costs determined to be lessor improvements recorded as prepaid lease payments and right-of-use assets, offset by government funding received. We revised our capital expenditures definition in the quarter ended March 31, 2023 to exclude the portions in accounts payable and accrued expenses.
Operating Expenses
Operating expenses include the following for the periods presented (in thousands, except percentages):
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Year Ended December 31, | | Percentage of Revenue |
| 2023 | | 2022 | | Change | | 2023 | | 2022 |
| Cost of revenue | $ | 148,743 | | | $ | 168,957 | | | (12.0) | % | | 51.5 | % | | 19.1 | % |
| Selling, general and administrative | 151,390 | | | 129,259 | | | 17.1 | % | | 52.4 | % | | 14.7 | % |
| Research and development | 17,280 | | | 18,369 | | | (5.9) | % | | 6.0 | % | | 2.1 | % |
| Change in estimated fair value of contingent consideration | (3,286) | | | (7,800) | | | (57.9) | % | | (1.1) | % | | (0.9) | % |
| | | | | | | | | |
| Restructuring | 6,466 | | | — | | | * | | 2.2 | % | | — | % |
| Total operating expenses | $ | 320,593 | | | $ | 308,785 | | | 3.8 | % | | 111.0 | % | | 35.0 | % |
____________________
*Not meaningful
Cost of Revenue
Cost of revenue decreased by $20.2 million from $169.0 million for the year ended December 31, 2022 to $148.7 million for the year ended December 31, 2023, or 12.0%. The decrease in cost of revenue compared to the prior period was primarily attributable to a decrease of $42.2 million in direct product costs driven by overall decreased revenues. This was partially offset by an increase of $14.3 million in personnel costs primarily driven by retention payment accruals associated with the acquisition of MyChem, additional headcount to support expanded manufacturing capacity and additional headcount related to the acquisition of Alphazyme, an increase of $6.0 million in depreciation and amortization expense primarily due to new equipment and newly acquired intangible assets, and an increase of $1.6 million in facilities costs driven by new facilities.
Gross profit decreased by $573.8 million from $714.0 million for the year ended December 31, 2022 to $140.2 million for the year ended December 31, 2023. The decrease in gross profit margin as a percentage of sales was primarily attributable to a decrease in volume, unfavorable product mix shift, an overall increase in the cost of revenue as a percentage of sales as the result of higher labor and facility costs, and depreciation and amortization.
Selling, General and Administrative
Selling, general and administrative expenses increased by $22.1 million from $129.3 million for the year ended December 31, 2022 to $151.4 million for the year ended December 31, 2023, or 17.1%. The increase was primarily driven by an increase of $22.7 million in personnel costs largely due to additional headcount from the acquisition of Alphazyme and incremental equity-based compensation expense.
Research and Development
Research and development expenses decreased by $1.1 million from $18.4 million for the year ended December 31, 2022 to $17.3 million for the year ended December 31, 2023, or 5.9%. The decrease in expenses compared to the prior period was primarily driven by a decrease of $1.8 million in personnel costs largely due to certain retention payment accruals now being included in cost of revenue. This is partially offset by an increase of $0.6 million in facilities costs driven by new facilities.
Change in Estimated Fair Value of Contingent Consideration
The change in estimated fair value of contingent consideration changed by $4.5 million from $7.8 million for the year ended December 31, 2022 to $3.3 million for the year ended December 31, 2023, or 57.9%. The changes were due to the decreases in estimated fair value of the liability for the contingent payments associated with the acquisitions of Alphazyme and MyChem. These were due to changes in estimates associated with revenue projections relative to defined revenue targets or thresholds that would trigger contingent payments per the Securities Purchase Agreement between the Company and sellers of Alphazyme and the Securities Purchase Agreement between the Company and the sellers of MyChem. See Notes 2 and 5 to our consolidated financial statements for additional information.
Restructuring
Restructuring costs for the year ended December 31, 2023 relate to the Cost Realignment Plan, which was implemented in November 2023. These include severance and other employee-related costs of $4.3 million, offset by a $0.1 million equity-based compensation benefit, facility and other exit costs of $2.0 million, and professional fees and other associated costs of $0.3 million. See Note 3 to our consolidated financial statements for additional information.
Other Income (Expense)
Other income (expense) includes the following for the periods presented (in thousands, except percentages):
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Year Ended December 31, | | Percentage of Revenue |
| 2023 | | 2022 | | Change | | 2023 | | 2022 |
| Interest expense | $ | (45,892) | | | $ | (20,414) | | | 124.8 | % | | (15.9) | % | | (2.3) | % |
| Interest income | 27,727 | | | 2,338 | | | 1085.9 | % | | 9.6 | % | | 0.2 | % |
| Loss on extinguishment of debt | — | | | (208) | | | * | | — | % | | 0.0 | % |
| Change in payable to related parties pursuant to the Tax Receivable Agreement | 668,886 | | | (4,102) | | | * | | 231.5 | % | | (0.5) | % |
Other expense | (1,337) | | | (358) | | | 273.5 | % | | (0.5) | % | | 0.0 | % |
| Total other income (expense), net | $ | 649,384 | | | $ | (22,744) | | | * | | 224.7 | % | | (2.6) | % |
____________________
*Not meaningful
Other expense was $22.7 million for the year ended December 31, 2022 compared to Other income of $649.4 million for the year ended December 31, 2023, representing a change of $672.1 million. The overall change in Other income (expense) was primarily attributable to a $668.9 million gain related to the payable to related parties pursuant to the Tax Receivable Agreement as we concluded that it was not probable that we will be able to realize the remaining tax benefits based on estimates of future taxable income. This was partially offset by a $0.9 million increase in Other expense relating to the indemnification asset recorded in connection with the acquisition of MyChem.
Relationship with GTCR, LLC (“GTCR”)
As of December 31, 2023, investment entities affiliated with GTCR collectively controlled approximately 56% of the voting power of our common stock, which enables GTCR to control the vote of all matters submitted to a vote of our shareholders and to control the election of members of the Board and all other corporate decisions.
During the years ended December 31, 2023 and 2022, the Company made distributions of $9.6 million and $150.2 million, respectively, for tax liabilities to MLSH 1.
We are also a party to a Tax Receivable Agreement, or TRA, with MLSH 1, who is primarily owned by GTCR, and MLSH 2 (see Note 14 to our consolidated financial statements). The TRA provides for the payment by us to MLSH 1 and MLSH 2, collectively, of 85% of the amount of tax benefits, if any, that we actually realize, or in some circumstances are deemed to
realize, from exchanges of LLC Units (together with the corresponding shares of Class B common stock) for Class A common stock, as a result of (i) certain increases in the tax basis of assets of Topco LLC and its subsidiaries resulting from purchases or exchanges of LLC Units, (ii) certain tax attributes of the entities acquired from MLSH 1 and MLSH 2 in connection with the Organizational Transactions, Topco LLC and subsidiaries of Topco LLC that existed prior to the IPO, and (iii) certain other tax benefits related to our entering into the TRA, including tax benefits attributable to payments that we make under the TRA (collectively, the “Tax Attributes”). Payment obligations under the TRA are not conditioned upon any Topco LLC unitholders maintaining a continued ownership interest in us or Topco LLC, and the rights of MLSH 1 and MLSH 2 under the TRA are assignable. There is no stated term for the TRA, and the TRA will continue until all tax benefits have been utilized or expired unless we exercise our right to terminate the TRA for an agreed-upon amount.
We recognize the amount of TRA payments expected to be paid within the next 12 months and classify this amount as current. This determination was based on our estimate of taxable income for the year ended December 31, 2023. As of December 31, 2023, our current liability under the TRA was $7.1 million.
As of December 31, 2023, the Company has derecognized the remaining non-current liability under the TRA after concluding it was not probable that the Company will be able to realize the remaining tax benefits based on estimates of future taxable income. The estimation of liability under the TRA is by its nature imprecise and subject to significant assumptions regarding the amount, character, and timing of the taxable income in the future. If the valuation allowance recorded against the deferred tax assets applicable to the tax attributes referenced above is released in a future period, the remaining TRA liability may be considered probable at that time and recorded on the consolidated balance sheet and within earnings.
We made payments of $42.6 million to MLSH 1 and MLSH 2 pursuant to the TRA during the year ended December 31, 2023, of which $0.4 million is related to interest. This determination was based on our taxable income for the year ended December 31, 2022. We made payments of $35.3 million to MLSH 1 and MLSH 2 pursuant to the TRA during the year ended December 31, 2022, of which $1.1 million is related to interest. This determination was based on our taxable income for the year ended December 31, 2021.
Liquidity and Capital Resources
Overview
We have financed our operations primarily from cash flow from operations, borrowings under long-term debt agreements and, to a lesser extent, the sale of our Class A common stock.
As of December 31, 2023, we had cash and cash equivalents of $575.0 million and retained earnings of $285.7 million. We had a net loss of $138.4 million for the fiscal year ended December 31, 2023. We also had positive cash flow from operations of $126.2 million.
We have relied on revenue derived from product and services sales, and equity and debt financings to fund our operations to date.
Our principal uses of cash have been to fund operations, acquisitions and capital expenditures, as well as make tax distributions to MLSH 1, make TRA payments to MLSH 1 and MLSH 2 and make interest payments and mandatory principal payments on our long-term debt.
We plan to utilize our existing cash on hand, together with cash generated from operations, primarily to fund our commercial and marketing activities associated with our products and services, continued research and development initiatives, and ongoing investments into our manufacturing facilities to create efficiencies and build capacity. We believe our cash on hand, cash generated from operations and continued access to our credit facilities, will be sufficient to satisfy our cash requirements over the next 12 months and beyond.
We expect to spend approximately $2.8 million in restructuring costs primarily during the first quarter of 2024 associated with the Cost Realignment Plan using existing cash on hand.
As a result of our ownership of LLC Units in Topco LLC, the Company is subject to U.S. federal, state and local income taxes with respect to its allocable share of any taxable income of Topco LLC and is taxed at the prevailing corporate tax rates. In addition to tax expenses, we also will incur expenses related to our operations and we may be required to make payments under the TRA with MLSH 1 and MLSH 2. Due to the uncertainty of various factors, we cannot precisely quantify the likely tax benefits we will realize as a result of LLC Unit exchanges and the resulting amounts we are likely to pay out to LLC Unitholders of Topco LLC pursuant to the TRA. Assuming no changes in the relevant tax law, we expect that probable future payments under the TRA relating to the purchase by the Company of LLC Units from MLSH 1 and the corresponding tax attributes to be approximately $7.1 million. This determination is based on our estimate of taxable income for the year ended December 31, 2023. During the year ended December 31, 2023, we determined that making a payment under the non-current
portion of the TRA was not probable under Accounting Standards Codification 450 - Contingencies since a valuation allowance has been recorded against our deferred tax assets and we do not believe we will generate sufficient future taxable income to utilize related tax benefits and result in a payment under the TRA. If we had determined that making a payment under the TRA and generating sufficient future taxable income was probable, we would have also recorded a liability pursuant to the TRA, net of current portion, of approximately $665.3 million in the consolidated balance sheet. Future payments in respect of subsequent exchanges or financings and tax attributes relating to the purchase by the Company of LLC Units from MLSH 1 would be in addition to this amount and may be substantial. The foregoing numbers are estimates and the actual payments could differ materially. We expect to fund these payments using cash on hand and cash generated from operations.
As a result of a change of control, material breach, or our election to terminate the TRA early, (1) we could be required to make cash payments to MLSH 1 and MLSH 2 that are greater than the specified percentage of the actual benefits we ultimately realize in respect of the tax benefits that are subject to the TRA, and (2) we will be required to make an immediate cash payment equal to the present value of the anticipated future tax benefits that are the subject of the TRA, which payment may be made significantly in advance of the actual realization, if any, of such future tax benefits. In these situations, our obligations under the TRA could have a material adverse effect on our liquidity and could have the effect of delaying, deferring or preventing certain mergers, asset sales, other forms of business combination, or other changes of control. There can be no assurance that we will be able to finance our obligations under the TRA.
In addition to payments to be made under the TRA, we are also required to make tax distributions to MLSH 1 pursuant to the LLC Operating Agreement for the portion of income passing through to them from Topco LLC. During the years ended December 31, 2023 and 2022, the Company made distributions of $9.6 million and $150.2 million, respectively, for tax liabilities to MLSH 1 under this agreement.
Credit Agreement
The Credit Agreement among Intermediate, Cygnus and TriLink, as the borrowers, Topco LLC, as holdings, the lenders from time-to-time party thereto and Morgan Stanley Senior Funding, Inc., as administrative and collateral agent (as amended, supplemented or otherwise modified, the “Credit Agreement”), provides us with a term-loan facility (the “Term Loan”) totaling $600.0 million and a revolving credit facility (the “Revolving Credit Facility”) of $180.0 million for letters of credit and loans to be used for working capital and other general corporate financing purposes. Borrowings under the Credit Agreement are unconditionally guaranteed by Topco LLC, along with the existing and future material domestic subsidiaries of Topco LLC (subject to certain exceptions) as specified in the respective guaranty agreements, and are secured by a lien and security interest in substantially all of the assets of existing and future material domestic subsidiaries of Topco LLC that are loan parties.
In January 2022, the Company entered into an amendment (the “Amendment”) to refinance the term loan and to replace London Interbank Offered Rate (“LIBOR”) with a Term Secured Overnight Financing Rate (“SOFR”) based rate.
Commencing with the fiscal year ended December 31, 2021, and each fiscal year thereafter, the Credit Agreement requires that we make mandatory prepayments on the Term Loan principal out of certain excess cash flow, subject to certain step-downs based on the Company’s first lien net leverage ratio. The mandatory prepayment shall be reduced to 25% or 0% of the calculated excess cash flow if the Company’s first lien net leverage ratio was equal to or less than 4.75:1.00 or 4.25:1.00, respectively; however, no prepayment is required to the extent excess cash flow calculated for the respective period is equal to or less than $10.0 million. As of December 31, 2023, our first lien net leverage ratio was less than 4.25:1.00. Thus, a mandatory prepayment on the Term Loan out of excess cash flow was not required.
The Term Loan became repayable in quarterly payments of $1.4 million beginning in March 2022, with all remaining outstanding principal due in October 2027. The Term Loan includes prepayment provisions that allow us, at our option, to repay all or a portion of the principal amount at any time. The Revolving Credit Facility allows us to repay and borrow from time to time until October 2025, at which time all amounts borrowed must be repaid. Subject to certain exceptions and limitations, we are required to repay borrowings under the Term Loan and Revolving Credit Facility with the proceeds of certain occurrences, such as the incurrence of debt, certain equity contributions and certain asset sales or dispositions.
Accrued interest under the Credit Agreement is payable by us (a) quarterly in arrears with respect to Base Rate loans, (b) at the end of each interest rate period (or at each three-month interval in the case of loans with interest periods greater than three months) with respect to Term SOFR Rate loans, (c) on the date of any repayment or prepayment and (d) at maturity (whether by acceleration or otherwise). An annual commitment fee is applied to the daily unutilized amount under the Revolving Credit Facility at 0.375% per annum, with one stepdown to 0.25% per annum based on Intermediate’s first lien net leverage ratio.
Debt Covenants
The Credit Agreement includes financial covenants. One financial covenant is a consolidated first lien coverage ratio measured as of the last day of each fiscal quarter. Another requires that, if as of the end of any fiscal quarter the aggregate amount of
letters of credit obligations and borrowings under the Revolving Credit Facility outstanding as of the end of such fiscal quarter (excluding cash collateralized letters of credit obligations and letter of credit obligations in an aggregate amount not in excess of $5.0 million at any time outstanding and for the first four fiscal quarters ending after October 2020, borrowings of revolving credit loans made before October 2020) exceeds 35% of the aggregate amount of all Revolving Credit Commitments in effect as of such date, then the net leverage ratio of Intermediate may not be greater than 8.00 to 1.00. For purposes of this covenant, the net leverage ratio is calculated by dividing outstanding first lien indebtedness (net of cash) by Adjusted EBITDA over the preceding four fiscal quarters.
The Credit Agreement also contains negative and affirmative covenants in addition to the financial covenant, including covenants that restrict our ability to, among other things, incur or prepay certain indebtedness, pay dividends or distributions, dispose of assets, engage in mergers and consolidations, make acquisitions or other investments, and make changes in the nature of the business. The Credit Agreement contains certain events of default, including, without limitation, nonpayment of principal, interest or other obligations, violation of the covenants, insolvency, court ordered judgments and certain changes of control. The Credit Agreement also requires the Company to provide audited consolidated financial statements to the lenders no later than 120 days after year-end.
As of December 31, 2023, we were in compliance with these covenants.
As of December 31, 2023, interest rate on the Term Loan was 8.40%.
Tax Receivable Agreement
We are a party to the TRA with MLSH 1 and MLSH 2. The TRA provides for the payment by us to MLSH 1 and MLSH 2, collectively, of 85% of the amount of certain tax benefits, if any, that we actually realize, or in some circumstances are deemed to realize, as a result of the Organizational Transactions, IPO and any subsequent purchases or exchanges of LLC Units of Topco LLC.
As of December 31, 2023, our current liability under the TRA was $7.1 million, representing 85% of the calculated tax savings we expect to utilize for the year ended December 31, 2023. During the year ended December 31, 2023, we recognized a gain of $668.9 million, in connection with a TRA liability adjustment due to remeasuring the non-current portion of the liability to zero as we no longer consider the payments under the agreement to be probable. We may record additional liabilities under the TRA when LLC Units are exchanged in the future and as our estimates of the future utilization of the Tax Attributes, net operating losses and other tax benefits change. We expect to make payments under the TRA, to the extent they are required, within 125 days after the extended due date of our U.S. federal income tax return for such taxable year. Interest on such payments will begin to accrue from the due date (without extensions) of such tax return at a rate of LIBOR (or, if LIBOR ceases to be published, a Replacement Rate) plus 100 basis points. Generally, any late payments will continue to accrue interest at LIBOR (or a Replacement Rate, as applicable) plus 500 basis points until such payments are made. Given the cessation of LIBOR, we have transitioned to the Secured Overnight Financing Rate (“SOFR”) as the applicable Replacement Rate as allowable under the Tax Receivable Agreement.
The payment obligations under the TRA are obligations of Maravai LifeSciences Holdings, Inc. and not of Topco LLC. Although the actual timing and amount of any payments that may be made under the TRA will vary, the aggregate payments that we will be required to make to MLSH 1 and MLSH 2 may be substantial. Any payments made by us under the TRA will generally reduce the amount of overall cash flow that might have otherwise been available to us or to Topco LLC and, to the extent that we are unable to make payments under the TRA for any reason, the unpaid amounts will be deferred and will accrue interest until paid by us. We anticipate funding ordinary course payments under the TRA from cash flow from operations of Topco LLC and its subsidiaries, available cash and/or available borrowings under the Credit Agreement.
During the year ended December 31, 2023, we determined that making a payment under the non-current portion of the TRA was not probable under Accounting Standards Codification 450 - Contingencies as a result of a valuation allowance having been recorded against our deferred tax assets, and therefore, that it is more likely than not that we will not generate sufficient future taxable income to utilize related tax benefits that would result in a payment under the TRA. If we had determined that making a payment under the TRA and generating sufficient future taxable income was probable, we would have also recorded a liability pursuant to the TRA, net of current portion, of approximately $665.3 million in the consolidated balance sheet.
Cash Flows
The following table summarizes our cash flows for the periods presented (in thousands):
| | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 |
| Net cash provided by (used in): | | | |
| Operating activities | $ | 126,224 | | | $ | 535,977 | |
| Investing activities | (122,310) | | | (267,612) | |
| Financing activities | (61,090) | | | (187,499) | |
| | | |
Net (decrease) increase in cash and cash equivalents | $ | (57,176) | | | $ | 80,866 | |
Operating Activities
Net cash provided by operating activities for the year ended December 31, 2023 was $126.2 million, which was primarily attributable to a net cash inflow from the change in our operating assets and liabilities of $97.8 million, non-cash depreciation and amortization of $40.3 million, non-cash amortization of operating lease right-of-use assets of $8.5 million, non-cash amortization of deferred financing costs of $2.9 million, non-cash equity-based compensation of $34.6 million, and non-cash deferred income taxes of $754.9 million. These were partially offset by a net loss of $138.4 million, non-cash gain on the change in estimated fair value of contingent consideration of $3.3 million, and non-cash gain on the revaluation of liabilities under the TRA of $668.9 million.
Net cash provided by operating activities for the year ended December 31, 2022 was $536.0 million, which was primarily attributable to a net income of $490.7 million, non-cash depreciation and amortization of $31.8 million, non-cash amortization of operating lease right-of-use assets of $6.3 million, non-cash amortization of deferred financing costs of $2.8 million, non-cash equity-based compensation of $18.7 million, non-cash deferred income taxes of $42.3 million, and non-cash loss on the revaluation of liabilities under the TRA of $4.1 million. These were partially offset by a non-cash loss on the change in estimated fair value of contingent consideration $7.8 million, and a net cash outflow from the change in our operating assets and liabilities of $45.1 million, which is net of government funding of $17.0 million. The net cash outflow from the change in our operating assets includes $13.4 million relating to an increase in prepaid lease payments for Flanders I (as defined in Note 7 to our consolidated financial statements).
Investing Activities
Net cash used in investing activities for the year ended December 31, 2023 was $122.3 million, which was primarily comprised of $69.8 million for the net cash consideration paid for the acquisition of Alphazyme and cash outflows of $65.6 million for property and equipment purchases. These were partially offset by proceeds from government assistance allocated to property and equipment of $12.9 million.
Net cash used in investing activities for the year ended December 31, 2022 was $267.6 million, which was primarily comprised of $239.0 million for the net cash consideration paid for the acquisition of MyChem, net cash outflows of $17.1 million for property and equipment purchases, and $13.3 million of prepaid lease payments for Flanders II (as defined in Note 7 to our consolidated financial statements).
Financing Activities
Net cash used in financing activities for the year ended December 31, 2023 was $61.1 million, which was primarily attributable to $42.2 million of payments to MLSH 1 and MLSH 2 pursuant to the TRA, a $9.7 million payment of acquisition consideration holdback relating to the acquisition of MyChem, $9.6 million of distributions for tax liabilities to non-controlling interest holders, required pursuant to the terms of the LLC Operating Agreement, and $5.4 million of principal repayments of long-term debt. This was partially offset by proceeds from derivative instruments of $6.2 million.
Net cash used in financing activities for the year ended December 31, 2022 was $187.5 million, which was primarily attributable to $150.2 million of distributions for tax liabilities to non-controlling interest holders, required pursuant to the terms of the LLC Operating Agreement, $34.2 million of payments to MLSH 1 and MLSH 2 pursuant to the TRA, and $13.9 million of principal repayments of long-term debt. This was partially offset by proceeds from borrowings of long-term debt of $8.5 million.
Capital Expenditures
Capital expenditures for the year ended December 31, 2023 totaled $52.7 million, which is net of government funding of $12.9 million. Capital expenditures for the year ending December 31, 2024 are projected to be in the range of $30.0 million to $35.0 million, which is net of anticipated government funding recognized. This includes leasehold improvements and equipment primarily for the Flanders San Diego Facility.
Contractual Obligations and Commitments
The following table summarizes our contractual obligations and commitments as of December 31, 2023 (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Payments due by period |
| Total | | 1 year | | 2 - 3 years | | 4 - 5 years | | 5+ years |
Operating leases (1) | $ | 65,533 | | | $ | 10,224 | | | $ | 20,431 | | | $ | 17,228 | | | $ | 17,650 | |
Finance leases (2) | 34,517 | | | 3,327 | | | 6,957 | | | 7,380 | | | 16,853 | |
Debt obligations (3) | 533,120 | | | 5,440 | | | 10,880 | | | 516,800 | | | — | |
TRA payments (4) | 7,069 | | | 7,069 | | | — | | | — | | | — | |
Unconditional purchase obligations (5) | 3,300 | | | 3,300 | | | — | | | — | | | — | |
MyChem retention payments (6) | 20,000 | | | 20,000 | | | — | | | — | | | — | |
| Total | $ | 663,539 | | | $ | 49,360 | | | $ | 38,268 | | | $ | 541,408 | | | $ | 34,503 | |
_______________
(1)Represents operating lease payment obligations, excluding any renewal options we are reasonably certain to execute and have recognized as lease liabilities. See Note 8 to our consolidated financial statements for additional information.
(2)Represents finance lease payment obligations, excluding any renewal options we are reasonably certain to execute and have recognized as lease liabilities. See Note 8 to our consolidated financial statements for additional information.
(3)Represents long-term debt principal maturities, excluding interest. See Note 10 to our consolidated financial statements for additional information.
(4)Reflects the estimated timing of the current TRA liability payment as of December 31, 2023. See "Risk Factors-Risks Related to Our Organizational Structure" and Note 14 to our consolidated financial statements for additional information regarding our liability under the TRA.
(5)Represents firm purchase commitments to our suppliers. See Note 9 to our consolidated financial statements for additional information.
(6)Represents certain payments to the sellers of MyChem as of the second anniversary of the closing of the acquisition date as long as they continue to be employed by TriLink. See Note 2 to our consolidated financial statements for additional information.
Tax distributions are required under the terms of the Topco LLC Agreement. As of December 31, 2023, we have made tax distributions equal to the estimated obligation due for 2023. See Note 14 to our consolidated financial statements for additional information regarding tax distributions.
Commencing with the fiscal year ended December 31, 2021, and each fiscal year thereafter, the Credit Agreement requires that we make mandatory prepayments of the Term Loan principal upon certain excess cash flow, subject to certain step-downs based on our first lien net leverage ratio. The mandatory prepayment shall be reduced to 25% or 0% of the calculated excess cash flow if the first lien net leverage ratio was equal to or less than 4.75:1.00 or 4.25:1.00, respectively; however, no prepayment shall be required to the extent excess cash flow calculated for the respective period is equal to or less than $10.0 million. As of December 31, 2023, our first lien net leverage ratio was less than 4.25:1.00.
In connection with our acquisition of Alphazyme, we may be required to make additional payments of up to $75.0 million to the sellers of Alphazyme dependent upon meeting or exceeding defined revenue targets during fiscal years 2023 through 2025. We may also be required to make certain payments of $9.3 million to its sellers and certain employees as of various dates but primarily through December 31, 2025 as long as these individuals continue to be employed by the Company. We cannot, at this time, determine when or if the related targets will be achieved or whether the events triggering the commencement of payment obligations will occur. Therefore, such payments were not included in the table above. See Notes 2 and 5 to our consolidated financial statements for additional details.
Critical Accounting Estimates
We have prepared our consolidated financial statements in accordance with GAAP. Our preparation of these consolidated financial statements requires us to make estimates and assumptions that affect the reported amounts of assets, liabilities, revenue, expenses and related disclosures in the consolidated financial statements. Our estimates are based on historical
experience and on various other assumptions that we believe are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying value of assets and liabilities that are not readily apparent from other sources. Actual results could differ from these estimates under different assumptions or conditions and any such difference may be material.
Our significant accounting policies are described in more detail in Note 1 to our consolidated financial statements. We believe the following discussion addresses our most critical accounting estimates used in the preparation of our consolidated financial statements, which require subjective and complex judgments.
Income Taxes
We are subject to U.S. federal and state income taxes. We are the controlling member of Topco LLC, which has been, and will continue to be, treated as a partnership for U.S. federal and state income tax purposes. Topco LLC’s previously wholly-owned U.S. subsidiary, Maravai Life Sciences, Inc. (“Maravai Inc.”) and its subsidiaries, were taxpaying entities in the U.S., Canada, and the U.K. Maravai Inc.’s subsidiaries were sold and Maravai Inc. ceased to be a regarded entity and was deemed liquidated for U.S. tax purposes during the year ended December 31, 2021. Topco LLC’s wholly-owned subsidiary, Maravai LifeSciences International Holdings, Inc., is a taxpaying entity for U.S. and foreign jurisdictions and had limited activity subject to a transfer pricing arrangement during the year ended December 31, 2023. Topco LLC’s other subsidiaries are treated as pass-through entities for federal and state income tax purposes. The income or loss generated by these entities is not taxed at the LLC level. As required by U.S. tax law, income or loss generated by these LLCs passes through to their owners. As such, our tax provision consists solely of the activities of Maravai Inc. and its subsidiaries, prior to their disposal, and Maravai LifeSciences International Holdings, Inc., as well as our share of income or loss generated by Topco LLC. We anticipate this structure to remain in existence for the foreseeable future.
We account for income taxes under the asset and liability method of accounting. We recognize deferred tax assets and liabilities for the future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities and their respective tax bases, as well as for operating loss and tax credit carryforwards. We measure deferred tax assets and liabilities using enacted tax rates expected to apply to taxable income in the years in which we expect to recover or settle those temporary differences. We recognize the effect of a change in tax rates on deferred tax assets and liabilities in the results of operations in the period that includes the enactment date. We reduce the measurement of a deferred tax asset, if necessary, by a valuation allowance if it is more likely than not that we will not realize some or all of the deferred tax asset.
The realizability of the Company’s deferred tax asset related to its investment in Topco LLC depends on the Company receiving allocations of tax deductions for its tax basis in the investment and on the Company generating sufficient taxable income to fully offset such deductions. We believe it is more likely than not that the Company will not generate sufficient taxable income in the future to fully realize any deductions allocated to it from Topco LLC associated with the reversal of its tax basis as of December 31, 2023. In addition, a portion of the deferred tax asset may only be realizable through the sale or liquidation of the investment and our ability to generate sufficient capital gains. As such, a valuation allowance of $642.2 million has been recorded as of December 31, 2023 to reflect the deferred tax assets that are more likely than not to not be realized.
We account for uncertain tax positions by recognizing the financial statement effects of a tax position only when, based upon technical merits, it is more likely than not that the position will be sustained upon examination.
Significant judgment is required in determining the accounting for income taxes. In the ordinary course of business, many transactions and calculations arise where the ultimate tax outcome is uncertain. Our judgments, assumptions and estimates relative to the accounting for income taxes take into account current tax laws, our interpretation of current tax laws, and possible outcomes of future audits conducted by foreign and domestic tax authorities. Although we believe that our estimates are reasonable, the final tax outcome of matters could be different from our assumptions and estimates used when determining the accounting for income taxes. Such differences, if identified in future periods, could have a material effect on the amounts recorded in our consolidated financial statements.
Payable to Related Parties Pursuant to the Tax Receivable Agreement
In November 2020, we entered into a TRA with MLSH 1 and MLSH 2. The TRA provides for the payment by us to MLSH 1 and MLSH 2, collectively, of 85% of the amount of tax benefits, if any, that we actually realize, or in some circumstances are deemed to realize from exchanges of LLC Units (together with the corresponding share of Class B Common stock), as a result of (i) certain increases in the tax basis of assets of Topco LLC and its subsidiaries resulting from purchases or exchanges of LLC Units, (ii) increase in the tax basis of assets of Topco LLC received form LLC Units held by entities acquired from MLSH 1 and MLSH 2 in connection with the Organizational Transactions, Topco LLC and subsidiaries of Topco LLC that existed prior to this offering and (iii) certain other tax benefits related to our entering into the TRA, including tax benefits attributable to payments that we make under the TRA (collectively, the “Tax Attributes”). The payment obligations under the TRA are not
conditioned upon any LLC Unitholder maintaining a continued ownership interest in us or Topco LLC and the rights of MLSH 1 and MLSH 2 under the TRA are assignable. We expect to benefit from the remaining 15% of the tax benefits, if any, that we may actually realize.
We accrue a liability for the payable to related parties for the TRA and a reduction to stockholders’ equity, when it is deemed probable that the Tax Attributes will be used to reduce our taxable income, as the contractual percentage of the benefit of Tax Attributes that we expected to receive over a period of time. The current portion, if any, of the liability is the amount estimated to be paid within one year of the balance sheet date. For purposes of estimating the value of the payable to related parties for the TRA, the tax benefit deemed realized by us and payable to MLSH 1 and MLSH 2 is computed by taking 85% of the difference between undiscounted forecasted cash income tax liability over the term of benefit of the Tax Attributes and the forecasted amount of such taxes that we would have been required to pay had there been no Tax Attributes (i.e. a with-and-without analysis); provided that, for purposes of determining the tax benefit with respect to state and local income taxes, use simplifying assumptions. The TRA will generally apply to each of our taxable years, beginning with the taxable year that the TRA is entered into. There is no maximum term for the TRA and the TRA will continue until all such tax benefits have been utilized or expired unless we exercise our right to terminate the TRA for an agreed-upon amount equal to the estimated present value of the remaining payments to be made under the agreement (calculated with certain assumptions, including as to utilization of the Tax Attributes). We may record additional liabilities under the TRA when LLC Units of Topco LLC are exchanged in the future and as our estimates of the future utilization of the tax benefits change. If, due to a change in facts, these tax attributes are not utilized in future years, it is reasonably possible no amounts would be paid under the TRA. In this scenario, the reduction of the liability under the TRA would result in a benefit to our consolidated statements of operations. Subsequent adjustments to the payable to related parties for the TRA based on changes in anticipated future taxable income, which could include changes in estimated income allocated to the partners of Topco LLC or apportionment of state income taxes, are recorded in our consolidated statements of operations.
The actual Tax Attributes, as well as any amounts paid to MLSH 1 and MLSH 2 under the TRA, will vary depending on a number of factors, including:
•the timing of any future exchanges—for instance, the increase in any tax deductions will vary depending on the fair value, which may fluctuate over time, of the depreciable or amortizable assets of Topco LLC and its flow-through subsidiaries at the time of each exchange;
•the price of shares of our Class A common stock at the time of any future exchanges—the increases and adjustments in our proportionate share of the existing tax basis of the assets of Topco LLC and its flow-through subsidiaries that are directly related to the price of shares of our Class A common stock at the time of future exchanges;
•the extent to which such exchanges are taxable—if an exchange is not taxable for any reason, increased tax deductions as a result of legacy IRC Section 754 election in place at Topco LLC will not be available to generate payments under the TRA;
•the amount and timing of our income—the TRA generally will require us to pay 85% of the tax benefits as and when those benefits are treated as realized by us under the terms of the TRA. If we do not have taxable income in a particular taxable year, we generally will not be required (absent a change of control or other circumstances requiring an early termination payment) to make payments under the TRA for that taxable year because no tax benefits will have been actually realized. Nevertheless, any tax benefits that do not result in realized tax benefits in a given taxable year will likely generate tax attributes that may be utilized to generate tax benefits in future (and possibly previous) taxable years. The utilization of any such tax attributes will result in payments under the TRA; and
•applicable tax rates—the tax rates in effect at the time a tax benefit is recognized.
The payment obligations under the TRA are obligations of Maravai LifeSciences Holdings, Inc. and not of Topco LLC. Although the actual timing and amount of any payments that may be made under the TRA will vary, we expect that the aggregate payments that we will be required to make to MLSH 1 and MLSH 2 may be substantial. Any payments made by us under the TRA will generally reduce the amount of overall cash flow that might have otherwise been available to us or to Topco LLC and, to the extent that we are unable to make payments under the TRA for any reason, the unpaid amounts will be deferred and will accrue interest until paid by us. We anticipate funding ordinary course payments under the TRA from cash flow from operations of Topco LLC and its subsidiaries, available cash and/or available borrowings under the Credit Agreement.
Assuming no changes in the relevant tax law, we expect that probable future payments under the TRA relating to the purchase by the Company of LLC Units from MLSH 1 and the tax attributes to be approximately $7.1 million. This determination is based on our estimate of taxable income for the year ended December 31, 2023. To the extent there is a change in the determination of the realizability of our deferred tax assets, this could impact the expected probable future payments under the TRA and the amount recorded in the consolidated balance sheet. Future payments in respect of subsequent exchanges or
financings and tax attributes relating to the purchase by the Company of LLC Units from MLSH 1 would be in addition to this amount and may be substantial. The foregoing numbers are estimates and the actual payments could differ materially. It is possible that future transactions or events could increase or decrease the actual tax benefits realized and the corresponding TRA payments. There may be a material negative effect on our liquidity if, as a result of timing discrepancies or otherwise, the payments under the TRA exceed the actual benefits we realize in respect of the tax attributes subject to the TRA and/or distributions to us by Topco LLC are not sufficient to permit us to make payments under the TRA after we have paid taxes.
The term of the TRA commenced upon the completion of our IPO and will continue until all such tax benefits have been utilized or expire, unless we exercise our rights to terminate the agreements or payments under the agreements are accelerated in the event we materially breach any of our material obligations under the agreements.
Goodwill
We evaluate goodwill at the reporting unit level on an annual basis and between annual tests if events and circumstances indicate it is more likely than not that the fair value of a reporting unit is less than its carrying value. Such indicators could include, but are not limited to, current economic and market conditions, including a decline in market capitalization, a significant adverse change in legal factors, business climate, operational performance of the business or key personnel. We perform our annual impairment test in the fourth quarter.
We first assess qualitative factors to determine whether it is more likely than not that the fair value of a reporting unit is less than its carrying value, including goodwill. If management concludes that it is more likely than not that the fair value of a reporting unit is less than its carrying value, management performs a quantitative goodwill impairment test. In performing the quantitative impairment test, management considers a number of factors to determine the fair value of a reporting unit, including an independent valuation to conduct this test. The valuation is based upon expected future discounted operating cash flows of a reporting unit as well as analysis of recent sales and ratio comparisons of similar companies. We base the discount rate on the weighted average cost of capital, or WACC, or market participants. If the carrying value of a reporting unit exceeds its estimated fair value, an impairment loss will be recognized for the amount in which the carrying amount exceeds the reporting unit’s fair value.
Due to the sustained decline in our stock price and the announcement of the Cost Realignment Plan in November 2023, we performed a quantitative analysis and compared our reporting units’ fair values to their respective carrying values to determine whether goodwill was impaired. We determined the fair values of our reporting units using a combination of the income approach using discounted cash flows and the market approach utilizing data from comparable public companies. The assumptions and estimates, including management’s estimated future revenue growth rates, estimated future margins and discount rates, used in the quantitative analysis were based on management’s best estimate about current and future conditions. The result of the quantitative analysis showed that the reporting units’ fair values exceeded their carrying values and there was no impairment of the recorded goodwill as of December 31, 2023. However, to the extent we continue to experience declines in our stock price or experience other impairment indicators, such as industry and market considerations or a decline in financial performance, or that the fair values of our reporting units are less than their carrying values, there could be a risk of goodwill impairment of our reporting units in future periods.
Recoverability and Impairment of Long-Lived Assets
We review the recoverability of our long-lived assets (including definite-lived intangible assets) if events or circumstances indicate the assets may be impaired. We measure recoverability of assets by comparing the respective carrying value of the assets to the current and expected future cash flows, on an undiscounted basis, to be generated from such assets. If our analysis indicates that the carrying value of these assets is not recoverable, we measure an impairment based on the amount by which the net carrying amount of the assets exceeds the fair values of the assets.
In conjunction with the goodwill impairment test performed during the fourth quarter of 2023, we also performed a recoverability assessment of our long-lived assets. The results of the analysis did not result in an impairment charge.
Recognition of Intangible Assets as Part of a Business Combination
We account for our business combinations using the acquisition method of accounting which requires that the assets acquired and liabilities assumed of acquired businesses be recorded at their respective fair values at the date of acquisition. The purchase price, which includes the fair value of consideration transferred, is attributed to the fair value of the assets acquired and liabilities assumed. The excess of the purchase price of the acquisition over the fair value of the identifiable net assets of the acquiree is recorded as goodwill.
Determining the fair value of intangible assets acquired requires management to use significant judgment and estimates, including the selection of valuation methodologies, assumptions about future net cash flows, discount rates and market
participants. Each of these factors can significantly affect the value attributed to the identifiable intangible asset acquired in a business combination.
We generally utilize a discounted cash flow method under the income approach to estimate the fair value of identifiable intangible assets acquired in a business combination. For the acquisitions of Alphazyme, LLC and MyChem, LLC, the estimated fair values of the developed technology intangible assets were based on the multi-period excess earnings method. The estimated fair values were developed by discounting future net cash flows to their present value at market-based rates of return. We selected the assumptions used in the financial forecasts using historical data, supplemented by current and anticipated market conditions, estimated revenue growth rates, management’s plans, and guideline companies. Some of the more significant assumptions inherent in estimating the fair value of these intangible assets included revenue growth rates, discount rates and assumed technical obsolescent curves.
The use of alternative estimates and assumptions could increase or decrease the estimated fair value and amounts allocated to identifiable intangible assets acquired and future amortization expense as well as goodwill.
Recent Accounting Pronouncements
See Note 1 to our consolidated financial statements for a discussion of recent accounting standards and pronouncements.
Item 7A. Quantitative and Qualitative Disclosures About Market Risk
Interest Rate Risk
As of December 31, 2023, our primary exposure to interest rate risk was associated with our variable rate long-term debt. Borrowings under our Credit Agreement bear interest at a rate equal to the Base Rate plus a margin of 2.00%, with respect to each Base Rate-based loan, or the Term SOFR (Secured Overnight Financing Rate) plus a margin of 3.00% with respect to each Term SOFR-based loan, subject in each case to an applicable Base Rate or Term SOFR floor (see Note 10 to our consolidated financial statements). Interest rates can fluctuate for a number of reasons, including changes in the fiscal and monetary policies or geopolitical events or changes in general economic conditions. This could adversely affect our cash flows.
As of December 31, 2023, we have an interest rate cap agreement in place to hedge a portion of our variable interest rate risk on our outstanding long-term debt. The agreement has a contract notional amount of $500.0 million and entitles us to receive from the counterparty at each calendar quarter end the amount, if any, by which a specified floating market rate exceeds the cap strike interest rate. The floating interest rate is reset at the end of each three-month period. The contract expires on January 19, 2025.
We had $533.1 million of outstanding borrowings under our Term Loan and no outstanding borrowings under our Revolving Credit Facility as of December 31, 2023. For the year ended December 31, 2023, the effect of a hypothetical 100 basis point increase or decrease in overall interest rates would have changed our interest expense by approximately $5.4 million.
We had cash and cash equivalents of $575.0 million as of December 31, 2023. Given the short-term nature of our investments, we do not believe there is any material risk to the value of our investments with increases or decreases in interest rates.
Foreign Currency Risk
All of our revenue is denominated in U.S. dollars. Although approximately 51.2% of our revenue for the year ended December 31, 2023 was derived from international sales, primarily in Europe and Asia Pacific, all of these sales are denominated in U.S. dollars. The majority of our expenses are generally denominated in the currencies in which they are incurred, which is primarily in the United States. As we expand our presence in international markets, to the extent we are required to enter into agreements denominated in a currency other than the U.S. dollar, results of operations and cash flows may increasingly be subject to fluctuations due to changes in foreign currency exchange rates and may be adversely affected in the future due to changes in foreign currency exchange rates. To date, we have not entered into any hedging arrangements with respect to foreign currency risk. As our international operations grow, we will continue to reassess our approach to manage our risk relating to fluctuations in currency rates.
Item 8. Financial Statements and Supplementary Data
INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
Report of Independent Registered Public Accounting Firm
To the Stockholders and the Board of Directors of Maravai LifeSciences Holdings, Inc.
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of Maravai LifeSciences Holdings, Inc. (the Company) as of December 31, 2023 and 2022, the related consolidated statements of operations, comprehensive (loss) income, changes in stockholders’ equity and cash flows for each of the three years in the period ended December 31, 2023, and the related notes (collectively referred to as the “consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company at December 31, 2023 and 2022, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2023, in conformity with U.S. generally accepted accounting principles.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (PCAOB), the Company's internal control over financial reporting as of December 31, 2023, based on criteria established in Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework), and our report dated February 29, 2024 expressed an unqualified opinion thereon.
Basis for Opinion
These financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on the Company’s financial statements based on our audits. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
Critical Audit Matters
The critical audit matters communicated below are matters arising from the current period audit of the financial statements that were communicated or required to be communicated to the audit committee and that: (1) relate to accounts or disclosures that are material to the financial statements and (2) involved our especially challenging, subjective or complex judgments. The communication of critical audit matters does not alter in any way our opinion on the consolidated financial statements, taken as a whole, and we are not, by communicating the critical audit matters below, providing separate opinions on the critical audit matters or on the accounts or disclosures to which they relate.
| | | | | | | | |
| | Goodwill Impairment Assessment |
| | |
Description of the Matter | | At December 31, 2023, the Company has recorded goodwill of $326.0 million. As discussed in Note 1 to the consolidated financial statements, goodwill is tested at the reporting unit level for impairment at least annually or more frequently if indicators of potential impairment exist. Under the goodwill impairment assessment, if the carrying amount of a reporting unit exceeds its fair value, an impairment loss is recognized in an amount equal to the amount of the excess carrying amount of the reporting unit over its fair value. During the current year the Company executed a quantitative assessment over the goodwill balance assigned to each reporting unit. |
| | |
| | Auditing the Company’s recoverability test for goodwill impairment assessment was challenging due to subjective estimates and assumptions used by the Company to determine fair value of the reporting units. The estimates were subject to higher uncertainty due to management judgements over significant assumptions, including revenue growth rates and valuation related discount rates. |
| | | | | | | | |
| | |
How We Addressed the Matter in Our Audit | | We obtained an understanding, evaluated the design, and tested the operating effectiveness of controls over the Company’s goodwill impairment process, including controls over management’s review of its projected financial information utilized in the valuation of its reporting units. |
| | |
| | Our audit procedures over the Company’s goodwill impairment assessment included, among others, assessing the reasonableness of significant assumptions, specifically revenue growth rates, discount rates and assessing the completeness and accuracy of the underlying data used by the Company in its analyses. We evaluated whether significant assumptions were reasonable by comparing them to industry data and current market forecasts, and whether such assumptions were consistent with evidence obtained in other areas of the audit. We performed various sensitivity analyses around the assumptions to understand the impact on the fair value calculation. We also involved our valuation specialists to assist us in evaluating the reasonableness of the Company’s valuation methodologies and certain significant assumptions used by the Company. |
| | |
| | Payable to related parties pursuant to a Tax Receivable Agreement |
| | |
Description of the Matter | | As discussed in Notes 1 and 14 of the December 31, 2023 consolidated financial statements, the Company has a Tax Receivable Agreement (TRA) with certain related party investors (TRA investors). The TRA liability represents a contractual commitment to distribute 85% of any tax benefits, realized or deemed to be realized by the Company to the TRA investors. As of December 31, 2023, the Company has recorded a TRA liability of $7.1 million after concluding it was probable that, based on estimates of future taxable income, the Company will owe a payment to the TRA investors. As of December 31, 2023, the Company has not recognized the remaining $665.3 million liability under the TRA after concluding it was not probable that the Company will be able to realize benefits based on estimates of future taxable income. |
| | |
| | Auditing management’s accounting for the TRA liability is complex because of the application of the tax laws used to determine the tax basis upon which to calculate the corresponding TRA liability and assumptions around the timing and amount of taxable income in the future which impacts the recognition of the TRA liability. These factors involved subjective auditor judgment and audit effort in performing procedures and evaluating the appropriateness of the calculation of the tax basis. In addition, the audit effort involved the use of professionals with specialized skill and knowledge to assist in performing these procedures and evaluating the audit evidence obtained from these procedures. |
| | |
How We Addressed the Matter in Our Audit | | We obtained an understanding, evaluated the design, and tested the operating effectiveness of controls over the computation and recognition of the Company’s TRA liability, including testing management's controls over the completeness and accuracy of the underlying data used in the valuation and recognition, and controls over management's review of the assumptions discussed above. |
| | |
| | Our audit procedures included, among others, testing the information used in the calculation and recognition of the TRA liability including projections of future taxable income and the involvement of professionals with specialized skills and knowledge to assist in developing an independent calculation of the tax basis, comparing the independent calculation to management’s calculations to evaluate the reasonableness of the tax basis, assessing management’s application of the tax laws, and verifying the calculation of the TRA liability was in accordance with the terms set out in the TRA. |
| | |
| | Revenue with distributors |
| | |
Description of the Matter | | During the year ended December 31, 2023, the Company’s revenues were $288.9 million, of which a portion relates to products sold to distributors. Its distributor customers resell the products to end users. Revenues from product sales are recognized when control is transferred to the Company’s customer. |
| | |
| | | | | | | | |
| | Auditing the Company’s product sales to distributors was challenging, specifically related to the effort required to audit the respective sales activity to assess whether incentives were provided that were not properly recognized. This involved judgmentally assessing factors including distributor customer ordering patterns, contractual terms, incentives offered and after shipment credits or free goods as described in Note 1 to the consolidated financial statements. |
| | |
How We Addressed the Matter in Our Audit | | We obtained an understanding, evaluated the design and tested the operating effectiveness of internal controls over the Company’s process to monitor appropriate terms and conditions for these transactions. This includes testing relevant controls over the information systems that are important to the initiation, recording and billing of revenue transactions as well as controls over the completeness and accuracy of the data used. |
| | |
| | Our audit procedures over the Company’s product sales to distributor customers included, among others, performing analytical procedures to detect and investigate anomalies within the data. We also examined the terms and conditions of selected new or amended contracts with distributor customers and its impact on the Company’s recognition model. We also confirmed the terms and conditions of contracts directly with a selection of distributor customers, including whether there are side agreements and terms not formally included in the contract that may impact the Company’s revenue recognition. In addition, we obtained written representations from members of the commercial organization regarding the completeness of the terms and conditions reported to the legal and accounting departments. |
/s/ Ernst & Young LLP
We have served as the Company’s auditor since 2016.
San Mateo, California
February 29, 2024
MARAVAI LIFESCIENCES HOLDINGS, INC.
CONSOLIDATED BALANCE SHEETS
(in thousands, except par value)
| | | | | | | | | | | |
| December 31, |
| 2023 | | 2022 |
| Assets | | | |
| Current assets: | | | |
| Cash and cash equivalents | $ | 574,962 | | | $ | 632,138 | |
| Accounts receivable, net | 54,605 | | | 138,624 | |
| Inventory | 51,397 | | | 43,152 | |
| Prepaid expenses and other current assets | 17,830 | | | 25,798 | |
| Government funding receivable | 1,118 | | | 8,190 | |
| Total current assets | 699,912 | | | 847,902 | |
| Property and equipment, net | 162,900 | | | 52,694 | |
| Goodwill | 326,029 | | | 283,668 | |
| Intangible assets, net | 220,987 | | | 216,663 | |
| Deferred tax assets | — | | | 765,799 | |
| Other assets | 77,622 | | | 115,589 | |
| Total assets | $ | 1,487,450 | | | $ | 2,282,315 | |
| Liabilities and stockholders’ equity | | | |
| Current liabilities: | | | |
| Accounts payable | $ | 10,729 | | | $ | 5,991 | |
| Accrued expenses and other current liabilities | 60,237 | | | 53,371 | |
| Deferred revenue | 3,360 | | | 3,088 | |
| Current portion of payable to related parties pursuant to the Tax Receivable Agreement | 7,069 | | | 42,254 | |
| Current portion of long-term debt | 5,440 | | | 5,440 | |
| Current portion of finance lease liabilities | 633 | | | — | |
| Total current liabilities | 87,468 | | | 110,144 | |
| Long-term debt, less current portion | 518,707 | | | 521,997 | |
| Finance lease liabilities, less current portion | 31,897 | | | — | |
| | | |
| Payable to related parties pursuant to the Tax Receivable Agreement, less current portion | — | | | 675,956 | |
| Other long-term liabilities | 59,494 | | | 68,975 | |
| Total liabilities | 697,566 | | | 1,377,072 | |
Commitments and contingencies (Note 9) | | | |
| Stockholders’ equity: | | | |
Class A common stock, $0.01 par value - 500,000 shares authorized; 132,228 and 131,692 shares issued and outstanding as of December 31, 2023 and 2022, respectively | 1,322 | | | 1,317 | |
Class B common stock, $0.01 par value - 300,000 shares authorized; 119,094 and 123,669 shares issued and outstanding as of December 31, 2023 and 2022 | 1,191 | | | 1,237 | |
| Additional paid-in capital | 128,503 | | | 137,898 | |
| Retained earnings | 285,737 | | | 404,766 | |
| | | |
| Total stockholders’ equity attributable to Maravai LifeSciences Holdings, Inc. | 416,753 | | | 545,218 | |
| Non-controlling interest | 373,131 | | | 360,025 | |
| Total stockholders’ equity | 789,884 | | | 905,243 | |
| Total liabilities and stockholders’ equity | $ | 1,487,450 | | | $ | 2,282,315 | |
The accompanying notes are an integral part of these consolidated financial statements.
MARAVAI LIFESCIENCES HOLDINGS, INC.
CONSOLIDATED STATEMENTS OF OPERATIONS
(in thousands, except per share amounts)
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 | | 2021 |
| Revenue | $ | 288,945 | | | $ | 883,001 | | | $ | 799,240 | |
| Operating expenses: | | | | | |
| Cost of revenue | 148,743 | | | 168,957 | | | 140,561 | |
| Selling, general and administrative | 151,390 | | | 129,259 | | | 100,064 | |
| Research and development | 17,280 | | | 18,369 | | | 15,219 | |
| Change in estimated fair value of contingent consideration | (3,286) | | | (7,800) | | | — | |
| | | | | |
Restructuring | 6,466 | | | — | | | — | |
| Gain on sale of business | — | | | — | | | (11,249) | |
| | | | | |
| Total operating expenses | 320,593 | | | 308,785 | | | 244,595 | |
| (Loss) income from operations | (31,648) | | | 574,216 | | | 554,645 | |
| Other income (expense): | | | | | |
| Interest expense | (45,892) | | | (20,414) | | | (30,260) | |
| Interest income | 27,727 | | | 2,338 | | | — | |
| Loss on extinguishment of debt | — | | | (208) | | | — | |
| Change in payable to related parties pursuant to the Tax Receivable Agreement | 668,886 | | | (4,102) | | | 6,101 | |
| Other (expense) income | (1,337) | | | (358) | | | 279 | |
Income before income taxes | 617,736 | | | 551,472 | | | 530,765 | |
| Income tax expense | 756,111 | | | 60,809 | | | 61,515 | |
| Net (loss) income | (138,375) | | | 490,663 | | | 469,250 | |
| Net (loss) income attributable to non-controlling interests | (19,346) | | | 270,458 | | | 287,213 | |
| Net (loss) income attributable to Maravai LifeSciences Holdings, Inc. | $ | (119,029) | | | $ | 220,205 | | | $ | 182,037 | |
| | | | | |
| Net (loss) income per Class A common share attributable to Maravai LifeSciences Holdings, Inc.: | | | | | |
| Basic | $ | (0.90) | | | $ | 1.67 | | | $ | 1.59 | |
| Diluted | $ | (0.90) | | | $ | 1.67 | | | $ | 1.56 | |
| | | | | |
Weighted average number of Class A common shares outstanding: | | | | | |
| Basic | 131,919 | | | 131,545 | | | 114,791 | |
| Diluted | 131,919 | | | 255,323 | | | 257,803 | |
The accompanying notes are an integral part of these consolidated financial statements.
MARAVAI LIFESCIENCES HOLDINGS, INC.
CONSOLIDATED STATEMENTS OF COMPREHENSIVE (LOSS) INCOME
(in thousands)
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 | | 2021 |
Net (loss) income | $ | (138,375) | | | $ | 490,663 | | | $ | 469,250 | |
| Other comprehensive income: | | | | | |
| Foreign currency translation adjustments | — | | | — | | | 55 | |
Total other comprehensive (loss) income | (138,375) | | | 490,663 | | | 469,305 | |
Comprehensive (loss) income attributable to non-controlling interests | (19,346) | | | 270,458 | | | 287,224 | |
Total comprehensive (loss) income attributable to Maravai LifeSciences Holdings, Inc. | $ | (119,029) | | | $ | 220,205 | | | $ | 182,081 | |
The accompanying notes are an integral part of the consolidated financial statements.
MARAVAI LIFESCIENCES HOLDINGS, INC.
CONSOLIDATED STATEMENTS OF CHANGES IN STOCKHOLDERS’ EQUITY
(in thousands)
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Class A Common Stock | | Class B Common Stock | | | | | | | | | | |
| Shares | | Amount | | Shares | | Amount | | Additional Paid-In Capital | | Retained Earnings | | Accumulated Other Comprehensive Income (Loss) | | Non-controlling Interest | | Total Stockholders’ Equity |
| December 31, 2020 | 96,647 | | $ | 966 | | | 160,974 | | $ | 1,610 | | | $ | 85,125 | | | $ | 854 | | | $ | (44) | | | $ | 66,235 | | | $ | 154,746 | |
| Cumulative effect of adoption of ASC 842, net of tax | — | | — | | | — | | | — | | | — | | | 1,670 | | | — | | | 2,784 | | | 4,454 | |
| Effect of exchanges of LLC Units | 34,734 | | 348 | | | (34,734) | | | (348) | | | 31,003 | | | — | | | — | | | (31,003) | | | — | |
| Recognition of impact of Tax Receivable Agreement due to exchanges of LLC Units | — | | — | | | — | | — | | | 53,000 | | | — | | | — | | | — | | | 53,000 | |
| Issuance of Class A common stock under employee equity plans, net of shares withheld for employee taxes | 107 | | 1 | | | — | | — | | | 1,669 | | | — | | | — | | | — | | | 1,670 | |
| Impact of cash contribution to Topco LLC, exchange and forfeiture of LLC Units, and forfeiture of Class B common stock by MLSH 1 | — | | — | | | (2,571) | | | (25) | | | (46,206) | | | — | | | — | | | 51,451 | | | 5,220 | |
| Non-controlling interest adjustment for changes in proportionate ownership in Topco LLC | — | | — | | | — | | — | | | (809) | | | — | | | — | | | 809 | | | — | |
Equity-based compensation | — | | — | | | — | | — | | | 4,645 | | | — | | | — | | | 5,813 | | | 10,458 | |
| Distribution for tax liabilities to non-controlling interest holder | — | | — | | | — | | — | | | (41) | | | — | | | — | | | (153,451) | | | (153,492) | |
| Net income | — | | — | | | — | | — | | | — | | | 182,037 | | | — | | | 287,213 | | | 469,250 | |
| Foreign currency translation adjustment | — | | — | | | — | | — | | | — | | | — | | | 44 | | | 11 | | | 55 | |
| December 31, 2021 | 131,488 | | 1,315 | | | 123,669 | | 1,237 | | | 128,386 | | | 184,561 | | | — | | | 229,862 | | | 545,361 | |
| Issuance of Class A common stock under employee equity plans, net of shares withheld for employee taxes | 204 | | | 2 | | | — | | | — | | | 2,303 | | | — | | | — | | | — | | | 2,305 | |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Class A Common Stock | | Class B Common Stock | | | | | | | | | | |
| Shares | | Amount | | Shares | | Amount | | Additional Paid-In Capital | | Retained Earnings | | Accumulated Other Comprehensive Income (Loss) | | Non-controlling Interest | | Total Stockholders’ Equity |
| Non-controlling interest adjustment for changes in proportionate ownership in Topco LLC | — | | | — | | | — | | | — | | | (864) | | | — | | | — | | | 864 | | | — | |
Equity-based compensation | — | | | — | | | — | | | — | | | 9,623 | | | — | | | — | | | 9,047 | | | 18,670 | |
| Distribution for tax liabilities to non-controlling interest holder | — | | | — | | | — | | | — | | | 141 | | | — | | | — | | | (150,206) | | | (150,065) | |
| Impact of change to deferred tax asset associated with cash contribution to Topco LLC | — | | | — | | | — | | | — | | | (1,691) | | | — | | | — | | | — | | | (1,691) | |
| Net income | — | | | — | | | — | | | — | | | — | | | 220,205 | | | — | | | 270,458 | | | 490,663 | |
| December 31, 2022 | 131,692 | | 1,317 | | | 123,669 | | 1,237 | | | 137,898 | | | 404,766 | | | — | | | 360,025 | | | 905,243 | |
| Effects of Structuring Transactions | — | | | — | | | (4,575) | | | (46) | | | (25,404) | | | — | | | — | | | 26,392 | | | 942 | |
| Issuance of Class A common stock under employee equity plans, net of shares withheld for employee taxes | 536 | | | 5 | | | — | | | — | | | 116 | | | — | | | — | | | — | | | 121 | |
| Non-controlling interest adjustment for changes in proportionate ownership in Topco LLC | — | | | — | | | — | | | — | | | 754 | | | — | | | — | | | (754) | | | — | |
Equity-based compensation | — | | | — | | | — | | | — | | | 18,167 | | | — | | | — | | | 16,421 | | | 34,588 | |
| Distribution for tax liabilities to non-controlling interest holder | — | | | — | | | — | | | — | | | — | | | — | | | — | | | (9,607) | | | (9,607) | |
Impact of change to deferred tax asset associated with equity-based compensation | — | | | — | | | — | | | — | | | (3,028) | | | — | | | — | | | — | | | (3,028) | |
| Net loss | — | | | — | | | — | | | — | | | — | | | (119,029) | | | — | | | (19,346) | | | (138,375) | |
| December 31, 2023 | 132,228 | | $ | 1,322 | | | 119,094 | | $ | 1,191 | | | $ | 128,503 | | | $ | 285,737 | | | $ | — | | | $ | 373,131 | | | $ | 789,884 | |
The accompanying notes are an integral part of the consolidated financial statements.
MARAVAI LIFESCIENCES HOLDINGS, INC.
CONSOLIDATED STATEMENTS OF CASH FLOWS
(in thousands)
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 | | 2021 |
| Operating activities: | | | | | |
| Net (loss) income | $ | (138,375) | | | $ | 490,663 | | | $ | 469,250 | |
Adjustments to reconcile net (loss) income to net cash provided by operating activities: | | | | | |
| Depreciation | 12,898 | | | 7,566 | | | 6,413 | |
| Amortization of intangible assets | 27,356 | | | 24,269 | | | 18,339 | |
Amortization of operating lease right-of-use assets | 8,527 | | | 6,268 | | | 8,792 | |
| Amortization of deferred financing costs | 2,929 | | | 2,788 | | | 2,676 | |
| Equity-based compensation expense | 34,588 | | | 18,670 | | | 10,458 | |
| Loss on extinguishment of debt | — | | | 208 | | | — | |
| Deferred income taxes | 754,942 | | | 42,318 | | | 46,904 | |
| | | | | |
| Change in estimated fair value of contingent consideration | (3,286) | | | (7,800) | | | — | |
| Gain on sale of business | — | | | — | | | (11,249) | |
| Revaluation of liabilities under the Tax Receivable Agreement | (668,886) | | | 4,102 | | | (6,101) | |
| Other | (2,313) | | | (7,993) | | | (281) | |
| Changes in operating assets and liabilities: | | | | | |
| Accounts receivable | 84,395 | | | (22,272) | | | (70,391) | |
| Inventory | 649 | | | 9,459 | | | (21,574) | |
| Prepaid expenses and other assets | 8,136 | | | (52,873) | | | (9,513) | |
| Government funding receivable | — | | | 16,973 | | | — | |
| Accounts payable | 5,284 | | | (1,578) | | | 676 | |
| Accrued expenses and other current liabilities | 15,108 | | | 8,503 | | | (3,457) | |
| Deferred revenue | 250 | | | (7,123) | | | (67,851) | |
| Other long-term liabilities | (15,978) | | | 3,829 | | | (4,521) | |
| Net cash provided by operating activities | 126,224 | | | 535,977 | | | 368,570 | |
| Investing activities: | | | | | |
| Cash paid for acquisition, net of cash acquired | (69,622) | | | (238,969) | | | — | |
| Purchases of property and equipment | (65,553) | | | (17,090) | | | (14,850) | |
Proceeds from government assistance allocated to property and equipment | 12,865 | | | 1,105 | | | — | |
| Prepaid lease payments on finance lease yet to commence | — | | | (13,278) | | | — | |
| Proceeds from sale of building | — | | | — | | | 548 | |
| Proceeds from sale of business, net of cash divested | — | | | 620 | | | 119,957 | |
| Net cash (used in) provided by investing activities | (122,310) | | | (267,612) | | | 105,655 | |
| Financing activities: | | | | | |
| Distributions to non-controlling interests holders | (9,607) | | | (150,206) | | | (153,451) | |
| Proceeds from borrowings of long-term debt, net of discount | — | | | 8,455 | | | — | |
| Principal repayments of long-term debt | (5,440) | | | (13,895) | | | (6,000) | |
| Payments of finance lease liabilities | (332) | | | — | | | — | |
| Proceeds from derivative instruments | 6,168 | | | — | | | — | |
| Payment of acquisition consideration holdback | (9,706) | | | — | | | — | |
| Payments to MLSH 1 pursuant to the Tax Receivable Agreement | (35,661) | | | (29,108) | | | (1,115) | |
| Payments to MLSH 2 pursuant to the Tax Receivable Agreement | (6,492) | | | (5,103) | | | (192) | |
| Shares withheld for employee taxes, net of proceeds from issuance of Class A common stock under employee equity plans | (20) | | | 2,358 | | | 1,709 | |
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 | | 2021 |
Net cash used in financing activities | (61,090) | | | (187,499) | | | (159,049) | |
| Effects of exchange rate changes on cash | — | | | — | | | (88) | |
| Net (decrease) increase in cash and cash equivalents | (57,176) | | | 80,866 | | | 315,088 | |
| Cash and cash equivalents, beginning of period | 632,138 | | | 551,272 | | | 236,184 | |
| Cash and cash equivalents, end of period | $ | 574,962 | | | $ | 632,138 | | | $ | 551,272 | |
| | | | | |
| Supplemental cash flow information: | | | | | |
| Cash paid for interest | $ | 44,256 | | | $ | 20,198 | | | $ | 27,234 | |
| Cash (refunded) paid for income taxes, net | $ | (2,987) | | | $ | 23,032 | | | $ | 22,473 | |
| | | | | |
| Supplemental disclosures of non-cash activities: | | | | | |
| Property and equipment included in accounts payable and accrued expenses | $ | 2,011 | | | $ | 1,701 | | | $ | 2,149 | |
| | | | | |
| Accrued receivable for capital expenditures to be reimbursed under a government contract | $ | 1,118 | | | $ | — | | | $ | — | |
| Right-of-use assets obtained in exchange for new finance lease liabilities | $ | 32,862 | | | $ | — | | | $ | — | |
| Right-of-use assets obtained in exchange for new operating lease liabilities | $ | 3,931 | | | $ | 17,513 | | | $ | — | |
| | | | | |
| Fair value of contingent consideration liability recorded in connection with acquisition of a business | $ | 5,289 | | | $ | 7,800 | | | $ | — | |
| Accrued consideration payable for MyChem acquisition | $ | — | | | $ | 10,000 | | | $ | — | |
Recognition of deferred tax assets as a result of exchanges of LLC Units and cash contribution | $ | — | | | $ | — | | | $ | 423,361 | |
| Recognition of liabilities under the Tax Receivable Agreement | $ | — | | | $ | — | | | $ | 366,179 | |
The accompanying notes are an integral part of the consolidated financial statements.
MARAVAI LIFESCIENCES HOLDINGS, INC.
NOTES TO THE CONSOLIDATED FINANCIAL STATEMENTS
1.Organization and Significant Accounting Policies
Description of Business
Maravai LifeSciences Holdings, Inc. (the “Company”, and together with its consolidated subsidiaries, “Maravai”, “we”, “us”, and “our”) provides critical products to enable the development of drugs, therapeutics, diagnostics, vaccines and support research on human diseases. Our products address the key phases of biopharmaceutical development and include complex nucleic acids for diagnostic and therapeutic applications and antibody-based products to detect impurities during the production of biopharmaceutical products.
The Company is headquartered in San Diego, California and has historically operated in three principal businesses: Nucleic Acid Production, Biologics Safety Testing and Protein Detection. In September 2021, the Company completed the divestiture of its Protein Detection business (see Note 2). Our Nucleic Acid Production business manufactures and sells products used in the fields of gene therapy, vaccines, nucleoside chemistry, oligonucleotide therapy and molecular diagnostics, including reagents used in the chemical synthesis, modification, labelling and purification of deoxyribonucleic acid (“DNA”) and ribonucleic acid (“RNA”). Our core Nucleic Acid Production offerings include messenger ribonucleic acid (“mRNA”), long and short oligonucleotides, our proprietary CleanCap® capping technology and oligonucleotide building blocks, and custom enzyme development and manufacturing. Our Biologics Safety Testing business sells highly specialized analytical products for use in biologic manufacturing process development, including custom product-specific development antibody and assay development services. Our Protein Detection business sold innovative labeling and detection reagents for researchers in immunohistochemistry.
Organization
We were incorporated as a Delaware corporation in August 2020 for the purpose of facilitating an initial public offering (“IPO”). Immediately prior to the IPO, we effected a series of organizational transactions (the “Organizational Transactions”), which, together with the IPO, were completed in November 2020, that resulted in the Company operating, controlling all of the business affairs and becoming the ultimate parent company of Maravai Topco Holdings, LLC (“Topco LLC”) and its consolidated subsidiaries. Maravai Life Sciences Holdings, LLC (“MLSH 1”), which is controlled by investment entities affiliated with GTCR, is the only other member of Topco LLC.
The Company is the sole managing member of Topco LLC, which operates and controls TriLink Biotechnologies, LLC (“TriLink”), Glen Research, LLC, MockV Solutions, LLC, Cygnus Technologies, LLC and Alphazyme, LLC (“Alphazyme”) and their respective subsidiaries. Prior to the Company’s divestiture of its Protein Detection business in September 2021, Topco LLC also operated and controlled Vector Laboratories, Inc. and its subsidiaries (“Vector”).
Basis of Presentation
The Company operates and controls all of the business and affairs of Topco LLC, and, through Topco LLC and its subsidiaries, conducts its business. Because we manage and operate the business and control the strategic decisions and day-to-day operations of Topco LLC and also have a substantial financial interest in Topco LLC, we consolidate the financial results of Topco LLC, and a portion of our net (loss) income is allocated to the non-controlling interests in Topco LLC held by MLSH 1.
The accompanying consolidated financial statements have been prepared in accordance with accounting principles generally accepted in the United States of America ("U.S. GAAP") pursuant to the rules and regulations of the Securities and Exchange Commission (“SEC”) and include our accounts and the accounts of our subsidiaries.
All intercompany transactions and accounts between the businesses comprising the Company have been eliminated in the accompanying consolidated financial statements.
Variable Interest Entities
The Company consolidates all entities that it controls through a majority voting interest or as the primary beneficiary of a variable interest entity (“VIE”). In determining whether the Company is the primary beneficiary of an entity, the Company applies a qualitative approach that determines whether it has both (i) the power to direct the economically significant activities of the entity and (ii) the obligation to absorb losses of, or the right to receive benefits from, the entity that could potentially be significant to that entity. The Company’s determination about whether it should consolidate such VIEs is made continuously as changes to existing relationships or future transactions may result in a consolidation event.
Use of Estimates
The preparation of consolidated financial statements in accordance with GAAP requires the Company to make judgements, estimates and assumptions that affect the reported amounts of assets, liabilities, equity, revenue and expenses, and related disclosures. These estimates form the basis for judgments the Company makes about the carrying values of assets and liabilities that are not readily apparent from other sources. The Company bases its estimates and judgments on historical experience and on various other assumptions that the Company believes are reasonable under the circumstances. These estimates are based on management’s knowledge about current events and expectations about actions the Company may undertake in the future. Significant estimates include, but are not limited to, the measurement of right-of-use assets and lease liabilities and related incremental borrowing rate, the payable to related parties pursuant to the Tax Receivable Agreement (as defined in Note 14), the realizability of our net deferred tax assets, and valuation of goodwill and intangible assets acquired in business combinations. Actual results could differ materially from those estimates.
Revenue Recognition
The Company generates revenue primarily from the sale of products, and to a much lesser extent, services in the fields of nucleic acid production and biologics safety testing. Prior to September 2021, the Company also generated revenue from its Protein Detection business. Products are sold primarily through a direct sales force and through distributors in certain international markets where the Company does not have a direct commercial presence.
Revenue is recognized when control of promised goods or services is transferred to a customer or distributor in an amount that reflects the consideration to which the entity expects to be entitled in exchange for those goods or services. Distributors are the principal in all sales transactions with its customers. To determine revenue recognition for its arrangements with customers, the Company performs the following five steps: (i) identify the contract(s) with a customer; (ii) identify the performance obligations in the contract; (iii) determine the transaction price; (iv) allocate the transaction price to the performance obligations in the contract; and (v) recognize revenue when (or as) the entity satisfies a performance obligation.
The majority of the Company’s contracts include only one performance obligation. A performance obligation is a promise in a contract to transfer a distinct good or service to the customer and is defined as the unit of account for revenue recognition. The Company also recognizes revenue from other contracts that may include a combination of products and services, the provision of solely services, or from license fee arrangements which may be associated with the delivery of product. Where there is a combination of products and services, the Company accounts for the promises as individual performance obligations if they are concluded to be distinct. Performance obligations are considered distinct if they are both capable of being distinct and distinct within the context of the contract. In determining whether performance obligations meet the criteria for being distinct, the Company considers a number of factors, such as the degree of interrelation and interdependence between obligations, and whether or not the good or service significantly modifies or transforms another good or service in the contract. As a practical expedient, we do not adjust the transaction price for the effects of a significant financing component if, at contract inception, the period between customer payment and the transfer of goods or services is expected to be one year or less. Contracts with customers are evaluated on a contract-by-contract basis as contracts may include multiple types of goods and services as described below.
Revenue from sales to customers through distributors are recognized consistent with the policies and practices for direct sales to customers, as described above.
Nucleic Acid Production
Nucleic Acid Production revenue is generated from the manufacture and sale of highly modified, complex nucleic acids products to support the needs of our of customers’ research, therapeutic and vaccine programs. The primary offering of products includes CleanCap, mRNA, specialized oligonucleotides, and enzymes. Contracts typically consist of a single performance obligation. We also sell nucleic acid products for labeling and detecting proteins in cells and tissue samples research. The Company recognizes revenue from these products in the period in which the performance obligation is satisfied by transferring control to the customer or distributor. Revenue for nucleic acid catalog products is recognized at a single point in time, generally upon shipment to the customer or distributor. Revenue for contracts for certain custom nucleic acid products, with an enforceable right to payment and a reasonable margin for work performed to date, is recognized over time, based on a cost-to-cost input method over the manufacturing period. Payments received from customers in advance of manufacturing their products is recorded as deferred revenue until the products were delivered.
Biologics Safety Testing
The Company’s Biologics Safety Testing revenue is associated with the sale of host cell protein, bioprocess impurity detection, viral clearance prediction kits and associated products. We also enter into contracts that include custom
antibody development, assay development, antibody affinity extraction and mass spectrometry services. These products and services enable the detection of impurities that occur in the manufacturing of biologic drugs and other therapeutics including cell and gene therapies. The Company recognizes revenue from the sale of kits and products in the period in which the performance obligation is satisfied by transferring control to the customer. Custom antibody development contracts consist of a single performance obligation, typically with an enforceable right to payment and a reasonable margin for work performed to date. Revenue is recognized over time based on a cost-to-cost input method over the contract term. Where an enforceable right to payment does not exist, revenue is recognized at a point in time when control is transferred to the customer. Assay development service contracts consist of a single performance obligation, revenue is recognized at a point in time when a successful antigen test and report is provided to the customer. Affinity extraction, mass spectrometry and other analytical services, which generally occur over a short period of time, consist of a single performance obligation to perform the service and provide a summary report to the customer. Revenue is recognized upon delivery of the report to the customer.
The Company elected the practical expedient to not disclose the unfulfilled performance obligations for contracts with an original length of one year or less. The Company had no material unfulfilled performance obligations for contracts with an original length greater than one year for any period presented.
The Company accepts returns only if the products do not meet customer specifications and historically, the Company’s volume of product returns has not been significant. Further, no warranties are provided for promised goods and services other than assurance type warranties.
Revenue for an individual contract is recognized at the related transaction price, which is the amount the Company expects to be entitled to in exchange for transferring the products and/or services. The transaction price for product sales is calculated at the contracted product selling price. The transaction price for a contract with multiple performance obligations is allocated to the separate performance obligations on a relative standalone selling price basis. Standalone selling prices for products are determined based on the prices charged to customers, which are directly observable. Standalone selling price of services are mostly based on time and materials. Generally, payments from customers are due when goods and services are transferred. As most contracts contain a single performance obligation, the transaction price is representative of the standalone selling price charged to customers. Revenue is recognized only to the extent that it is probable that a significant reversal of the cumulative amount recognized will not occur in future periods. Variable consideration has not been material to our consolidated financial statements.
Sales taxes
Sales taxes collected by the Company are not included in the transaction price as revenue as they are ultimately remitted to a governmental authority.
Shipping and handling costs
The Company has elected to account for shipping and handling activities related to contracts with customers as costs to fulfill the promise to transfer the associated products. Accordingly, revenue for shipping and handling is recognized at the same time that the related product revenue is recognized.
Contract costs
The Company recognizes the incremental costs of obtaining contracts as an expense when incurred when the amortization period of the assets that otherwise would have been recognized is one year or less. These costs are included in sales and marketing and general and administrative expenses. The costs to fulfill the contracts are determined to be immaterial and are recognized as an expense when incurred.
Contract balances
Contract assets are generated when contractual billing schedules differ from revenue recognition timing and the Company records contract receivable when it has an unconditional right to consideration. There were no contract asset balances as of December 31, 2023 or 2022.
Contract liabilities include billings in excess of revenue recognized, such as customer deposits and deferred revenue. Customer deposits, which are included in accrued expenses, are recorded when cash payments are received or due in advance of performance. Deferred revenue is recorded when the Company has unsatisfied performance obligations. Total contract liabilities were $5.5 million and $4.8 million as of December 31, 2023 and 2022, respectively. Contract liabilities are expected to be recognized into revenue within the next twelve months.
Disaggregation of Revenue
The following tables summarize the revenue by segment and region for the periods presented (in thousands):
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, 2023 |
| Nucleic Acid Production | | Biologics Safety Testing | | Total |
| North America | $ | 114,459 | | $ | 26,596 | | $ | 141,055 |
| Europe, the Middle East and Africa | 34,390 | | 15,532 | | 49,922 |
| Asia Pacific | 75,716 | | 21,725 | | 97,441 |
| Latin and Central America | 204 | | 323 | | 527 |
| Total revenue | $ | 224,769 | | $ | 64,176 | | $ | 288,945 |
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, 2022 |
| Nucleic Acid Production | | Biologics Safety Testing | | Total |
| North America | $ | 312,119 | | $ | 27,354 | | $ | 339,473 |
| Europe, the Middle East and Africa | 415,298 | | 17,628 | | 432,926 |
| Asia Pacific | 85,508 | | 24,286 | | 109,794 |
| Latin and Central America | 144 | | 664 | | 808 |
| Total revenue | $ | 813,069 | | $ | 69,932 | | $ | 883,001 |
| | | | | | | | | | | | | | | | | | | | | | | |
| Year Ended December 31, 2021 |
| Nucleic Acid Production | | Biologics Safety Testing | | Protein Detection | | Total |
| North America | $ | 280,369 | | $ | 25,686 | | $ | 11,016 | | $ | 317,071 |
| Europe, the Middle East and Africa | 377,325 | | 15,597 | | 4,752 | | 397,674 |
| Asia Pacific | 54,114 | | 26,471 | | 3,068 | | 83,653 |
| Latin and Central America | 56 | | 663 | | 123 | | 842 |
| Total revenue | $ | 711,864 | | $ | 68,417 | | $ | 18,959 | | $ | 799,240 |
Total revenue is attributed to geographic regions based on the bill-to location of the transaction. For all periods presented, the majority of our revenue was recognized at a point in time.
Shipping and Handling Costs
Shipping and handling costs, which are charged to customers, are included in revenue. Shipping and handling charges included in revenue were approximately $3.5 million, $3.2 million and $3.6 million for the years ended December 31, 2023, 2022 and 2021, respectively. Freight and supplies costs directly associated with shipping products to customers are included as a component of cost of revenue.
Research and Development
Research and development (“R&D”) expenses include personnel costs, including salaries, benefits and equity-based compensation for laboratory personnel, outside contracted services, and costs of supplies. R&D costs are expensed as incurred. Payments made prior to the receipt of goods or services to be used in R&D are recognized as prepaid assets until the goods are received or services are rendered.
Advertising Costs
The Company expenses advertising costs as incurred. Advertising costs incurred were approximately $2.9 million, $2.5 million and $1.3 million during the years ended December 31, 2023, 2022 and 2021, respectively.
Restructuring Costs
Restructuring costs relate to a cost realignment plan implemented by the Company in November 2023 to optimize business operations and match them to current market conditions. Restructuring costs are comprised of severance and other employee-related costs, facility and other exit costs, professional fees and other restructuring costs.
Employee separation costs principally consist of one-time termination benefits and other post-employment benefits. One-time termination benefits are expensed at the date the entity notifies the employee, unless the employee must provide future service, in which case the benefits are expensed over the future service period. Other post-employment benefits are expensed when the obligation is probable and the benefit amounts are estimable. Other costs associated with restructuring activities, including facility and other exist costs and professional fees, are expensed as they are incurred.
Equity-Based Compensation
Stock-Based Compensation
The Company recognized stock-based compensation for all equity awards made to employees based upon the awards’ estimated grant date fair value. For equity awards that vest subject to the satisfaction of service requirements, compensation expense is measured based on the fair value of the award on the date of grant and expense is recognized on a straight-line basis over the requisite service period, which is typically between two to four years. We account for forfeitures as they occur. Stock-based compensation is classified in the accompanying consolidated statements of operations based on the function to which the related services are provided.
The Company estimates the fair value of stock option grants using the Black-Scholes option pricing model. The assumptions used in estimating the fair value of these awards, such as expected term, expected dividend yield, volatility and risk-free interest rate, represent management’s best estimates and involve inherent uncertainties and the application of management’s judgment. If actual results are not consistent with the Company’s assumptions and judgments used in making these estimates, the Company may be required to increase or decrease compensation expense, which could be material to the Company’s consolidated results of operations.
The fair value of restricted stock units (“RSUs”) is determined based on the number of shares granted and the quoted market price of the Company’s Class A common stock on the date of grant.
For performance stock units (“PSUs”) which are subject to service and market conditions, compensation expense is measured based on the fair value of the award on the date of grant and expense is recognized on a straight-line basis over the requisite service period regardless if the market condition is satisfied. If the grantee is terminated prior to meeting both conditions, any previously recognized expense is reversed. The Company estimates the fair value of PSUs using the Monte Carlo simulation model. The assumptions used in estimating the fair value of these awards, such as expected term, volatility and risk-free interest rate, represent management’s best estimates and involve inherent uncertainties and the application of management’s judgment.
For PSUs which are subject to a performance condition, compensation expense is recognized on a straight-line basis over the requisite service period when the achievement of such performance condition is determined probable, and upon achieving such performance condition that was not previously considered as probable, records a cumulative catch-up adjustment to reflect the portion of the grantee’s requisite service that has been provided to date. If a performance condition is not determined probable or is not met, no compensation expense is recognized, and any previously recognized expense is reversed. The fair value of such PSUs is determined based on the quoted market price of the Company’s Class A common stock on the date of grant.
Unit-Based Compensation
Up until the IPO, MLSH 1 had granted unit-based awards to certain executives of Topco LLC who are also executives of the Company in the form of non-vested units. Topco LLC’s controlled subsidiary, MLSC, also granted unit-based awards only to certain employees of its subsidiaries (collectively, the “Incentive Units”). All awards of Incentive Units were measured based on the fair value of the award on the date of grant. The Company recognizes compensation expense for MLSH 1 awards in its consolidated financial statements as MLSH 1 is considered to be the economic interest holder in Topco LLC. Compensation expense for the Incentive Units is recognized over their requisite service period. Forfeitures are recognized when they occur.
The grant date fair value of Incentive Unit awards was determined by the Company’s Board of Directors with the assistance of management and an independent third-party valuation specialist.
Income Taxes
We are subject to U.S. federal and state income taxes. We are the controlling member of Topco LLC, which has been, and will continue to be, treated as a partnership for U.S. federal and state income tax purposes. Topco LLC’s previously wholly-owned U.S. subsidiary, Maravai Life Sciences, Inc. (“Maravai Inc.”) and its subsidiaries, were taxpaying entities in the U.S., Canada, and the U.K. Maravai Inc.’s subsidiaries were sold and Maravai Inc. ceased to be a regarded entity and was deemed liquidated for U.S. tax purposes during the year ended December 31, 2021. Topco LLC’s wholly-owned subsidiary, Maravai LifeSciences International Holdings, Inc., is a taxpaying entity for U.S. and foreign jurisdictions and had limited activity subject to a transfer pricing arrangement during the year ended December 31, 2023. Topco LLC’s other subsidiaries are treated as pass-through
entities for federal and state income tax purposes. The income or loss generated by these entities is not taxed at the LLC level. As required by U.S. tax law, income or loss generated by these LLCs passes through to their owners. As such, our tax provision consists solely of the activities of Maravai Inc. and its subsidiaries, prior to their disposal, and Maravai LifeSciences International Holdings, Inc., as well as our share of income or loss generated by Topco LLC.
We account for income taxes under the asset and liability method of accounting. Current income tax expense or benefit represents the amount of income taxes expected to be payable or refundable for the current year. We recognize deferred tax assets and liabilities for the future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities and their respective tax bases, as well as for operating loss and tax credit carryforwards. We measure deferred tax assets and liabilities using enacted tax rates expected to apply to taxable income in the years in which we expect to recover or settle those temporary differences. We recognize the effect of a change in tax rates on deferred tax assets and liabilities in the results of operations in the period that includes the enactment date. We reduce the measurement of a deferred tax asset, if necessary, by a valuation allowance if it is more likely than not that we will not realize some or all of the deferred tax asset.
The Company’s tax positions are subject to income tax audits. We account for uncertain tax positions by recognizing the financial statement effects of a tax position only when, based upon technical merits, it is more likely than not that the position will be sustained upon examination. Significant judgment is required in determining the accounting for income taxes. In the ordinary course of business, many transactions and calculations arise where the ultimate tax outcome is uncertain. Our judgments, assumptions and estimates relative to the accounting for income taxes take into account current tax laws, our interpretation of current tax laws, and possible outcomes of future audits conducted by foreign and domestic tax authorities. Although we believe that our estimates are reasonable, the final tax outcome of matters could be different from our assumptions and estimates used when determining the accounting for income taxes. Such differences, if identified in future periods, could have a material effect on the amounts recorded in our consolidated financial statements. Interest and penalties related to unrecognized tax benefits are recognized in income tax expense in the accompanying consolidated statements of operations. The provision for income taxes includes the effects of any accruals that the Company believes are appropriate, as well as any related net interest and penalties.
Payables to Related Parties Pursuant to the Tax Receivable Agreement
In November 2020, we entered into a Tax Receivable Agreement (“TRA”) with MLSH 1 and MLSH 2. The TRA provides for the payment by us to MLSH 1 and MLSH 2, collectively, of 85% of the amount of tax benefits, if any, that we actually realize, or in some circumstances are deemed to realize from exchanges of LLC Units (together with the corresponding shares of Class B common stock) for Class A common stock, as a result of (i) certain increases in the tax basis of assets of Topco LLC and its subsidiaries resulting from purchases or exchanges of LLC Units, (ii) certain tax attributes of the Organization Transactions and (iii) certain other tax benefits related to our entering into the TRA, including tax benefits attributable to payments that we make under the TRA (collectively, the “Tax Attributes”). The payment obligations under the TRA are not conditioned upon any LLC Unitholder maintaining a continued ownership interest in us or Topco LLC and the rights of MLSH 1 and MLSH 2 under the TRA are assignable. We expect to benefit from the remaining 15% of the tax benefits, if any, that we may actually realize.
We accrue a liability for the payable to related parties for the TRA and a reduction to stockholders’ equity, when it is deemed probable that the Tax Attributes will be used to reduce our taxable income, as the contractual percentage of the benefit of Tax Attributes that we expected to receive over a period of time. The current portion, if any, of the liability is the amount estimated to be paid within one year of the consolidated balance sheet date. For purposes of estimating the value of the payable to related parties for the TRA, the tax benefit deemed realized by us and payable to MLSH 1 and MLSH 2 is computed by taking 85% of the difference of between our undiscounted forecasted cash income tax liability over the term of benefit of the Tax Attributes and the forecasted amount of such taxes that we would have been required to pay had there been no Tax Attributes. The TRA applies to each of our taxable years, beginning with the taxable year that the TRA is entered into. There is no maximum term for the TRA and the TRA will continue until all such tax benefits have been utilized or expired unless we exercise our right to terminate the TRA for an agreed-upon amount equal to the estimated present value of the remaining payments to be made under the agreement. We may record additional liabilities under the TRA when LLC Units of Topco LLC are exchanged in the future and as our estimates of the future utilization of the tax benefits change. If, due to a change in facts, these tax attributes are not utilized in future years, it is reasonably possible no amounts would be paid under the TRA. In this scenario, the reduction of the liability under the TRA would result in a benefit to our consolidated statements of operations. Subsequent adjustments to the payable to related parties for the TRA based on changes in anticipated future taxable income are recorded in our consolidated statements of operations.
Non-Controlling Interests
Non-controlling interests represent the portion of profit or loss, net assets and comprehensive (loss) income of our consolidated subsidiaries that is not allocable to the Company based on our percentage of ownership of such entities.
In November 2020, following the completion of the Organizational Transactions, we became the sole managing member of Topco LLC. As of December 31, 2023, we held approximately 52.6% of the outstanding LLC Units of Topco LLC, and MLSH 1 held approximately 47.4% of the outstanding LLC Units of Topco LLC. Therefore, we report non-controlling interests based on the percentage of LLC Units of Topco LLC held by MLSH 1 on our consolidated balance sheet as of December 31, 2023. Income or loss attributed to the non-controlling interest in Topco LLC is based on the LLC Units outstanding during the period for which the income or loss is generated and is presented on the consolidated statements of operations and consolidated statements of comprehensive (loss) income.
MLSH 1 is entitled to exchange LLC Units, together with an equal number of shares of our Class B common stock (together referred to as “Paired Interests”), for shares of Class A common stock on a one-for-one basis or, at our election, for cash, from a substantially concurrent public offering or private sale (based on the price of our Class A common stock in such public offering or private sale). As such, future exchanges of Paired Interests by MLSH 1 will result in a change in ownership and reduce or increase the amount recorded as non-controlling interests and increase or decrease additional paid-in-capital when Topco LLC has positive or negative net assets, respectively.
Distributions of $9.6 million, $150.2 million and $153.5 million for tax liabilities were made to MLSH 1 during the years ended December 31, 2023, 2022 and 2021, respectively.
Segment Information
The Company has historically operated in three reportable segments. Operating segments are defined as components of an enterprise for which separate financial information is evaluated regularly by the Company’s chief operating decision maker (“CODM”) in deciding how to allocate resources and assessing performance. The CODM allocates resources and assesses performance based upon discrete financial information at the segment level. All of our long-lived assets are located in the United States. After the divestiture of Vector in September 2021, the Company no longer has the Protein Detection segment. The Company has reported the historical results of the Protein Detection business as such discrete financial information evaluated by the CODM for the periods presented included the information for this legacy segment. As of December 31, 2023, the Company operated in two reportable segments: Nucleic Acid Production and Biologics Safety Testing.
Cash and Cash Equivalents
The Company considers all highly liquid investments with original maturities of three months or less to be cash equivalents. The carrying value of these cash equivalents approximates fair value. Cash and cash equivalents consist of deposits held at financial institutions and money market funds.
Accounts Receivable and Allowance for Credit Losses
Accounts receivable primarily consist of amounts due from customers for product sales and services. The Company’s expected credit losses are developed using an estimated loss rate method that considers historical collection experience, current conditions, and reasonable and supportable forecasts that affect the collectability of the reported amount. The estimated loss rates are applied to trade receivables with similar risk characteristics such as the length of time the balance has been outstanding, liquidity and financial position of the customer, and the geographic location of the customer. In certain instances, the Company may identify individual accounts receivable assets that do not share risk characteristics with other accounts receivable, in which case the Company records its expected credit losses on an individual asset basis.
The allowance for credit losses was approximately $1.4 million and $2.2 million as of December 31, 2023 and 2022, respectively. Write-offs of accounts receivable were $0.7 million during the year ended December 31, 2023. Write-offs of accounts receivable were not significant during the years ended December 31, 2022 and 2021. There were $0.5 million of recoveries during the year ended December 31, 2023. Recoveries were not significant during the years ended December 31, 2022 and 2021.
Inventory
Inventories consist of raw materials, work-in-process and finished goods. Inventories are stated at the lower of cost (weighted average cost) or net realizable value. Inventory costs, which relate to the purchase or production of inventories, include materials, direct labor and manufacturing overhead. The Company regularly monitors for excess and obsolete inventory based on its estimates of expected sales volumes, production capacity and expiration of raw materials, work-in-process and finished products, and reduces the carrying value of inventory accordingly. The Company writes down inventory that has become
obsolete, inventory that has a cost basis in excess of its expected net realizable value, and inventory in excess of expected manufacturing requirements. Any write-downs of inventories are charged to cost of revenue.
A change in the estimated timing or amount of demand for the Company’s products could result in reduction to the recorded value of inventory quantities on hand. Any significant unanticipated changes in demand or unexpected quality failures could have a significant impact on the value of inventory and reported operating results. During all periods presented in the accompanying consolidated financial statements, there have been no material adjustments related to a revised estimate of our inventory valuations.
Government Assistance
The consideration awarded to the Company by the U.S. Department of Defense is outside the scope of the contracts with customers, income tax, funded research and development, and contribution guidance. This is because the awarding entity is not considered to be a customer, the receipt of the funding is not predicated on the Company’s income tax position, there are no refund provisions, and the entity is not receiving reciprocal value for their support provided to the Company. The Company’s elected policy is to recognize such assistance as a reduction to the carrying amount of the assets associated with the award when it is reasonably assured that the funding will be received as evidenced through the existence of an arrangement, amounts eligible for reimbursement are determinable and have been incurred or paid, the applicable conditions under the arrangement have been met, and collectability of amounts due is reasonably assured.
Property and Equipment
Property and equipment are stated at cost, less accumulated depreciation. Depreciation is computed using the straight-line method over the following estimated useful lives:
| | | | | | | | |
| Assets | | Estimated Useful Life |
| | |
| Leasehold improvements | | 12 years |
| Furniture, fixtures, equipment and software | | 3 - 7 years |
Leasehold improvements are amortized over the shorter of the related lease term or useful life.
Maintenance and repairs are charged to operations when incurred, while betterments or renewals are capitalized. When property and equipment are sold or otherwise disposed of, the asset account and related accumulated depreciation account are relieved, and any gain or loss is included in the results of operations.
The Company records certain government grants earned related to capital projects as a reduction to property and equipment.
Goodwill
Goodwill represents the excess of consideration transferred over the estimated fair value of assets acquired and liabilities assumed in a business combination. Goodwill is not amortized but is reviewed for impairment. Goodwill is allocated to the Company’s reporting units, which are components of our business for which discrete cash flow information is available one level below its operating segment. The Company conducts a goodwill impairment analysis at least annually and more frequently if changes in facts and circumstances indicate that the fair value of the Company’s reporting units may be less than carrying amount. In performing each annual impairment assessment and any interim impairment assessment, the Company determines if it should qualitatively assess whether it is more likely than not that the fair value of goodwill is less than its carrying amount (the qualitative impairment test). If it is more likely than not that the fair value of the reporting unit is less than its carrying amount, or if the Company elects not to perform the qualitative impairment test, the Company then performs a quantitative impairment test.
The quantitative impairment test is performed using a one-step process. The process is to compare the fair value of a reporting unit with its carrying amount. If the fair value of a reporting unit exceeds its carrying amount, goodwill of the reporting unit is not impaired. If the carrying amount of a reporting unit exceeds its fair value, goodwill of the reporting unit is impaired and an impairment loss is recognized in an amount equal to that excess up to the total amount of goodwill included in the reporting unit.
Intangible Assets
The Company’s finite-lived intangible assets represent purchased intangible assets and primarily consist of trade names, customer relationships, patents, and developed technology. Certain criteria are used in determining whether intangible assets acquired in a business combination must be recognized and reported separately. Finite-lived intangible assets are initially
recognized at fair value, are subject to amortization and are subsequently stated at amortized cost. The Company’s finite-lived intangible assets are amortized using a method that reflects the pattern in which the economic benefits of the intangible assets are consumed or otherwise used. If that pattern cannot be reliably determined, the intangible assets are amortized using the straight-line method over their estimated useful lives and are tested for impairment along with other long-lived assets. Amortization related to patents and developed technology is allocated to cost of revenue whereas amortization associated with trade names and customer relationships is allocated to selling, general and administrative expenses.
Impairment of Long-Lived and Intangible Assets
The Company periodically reviews long-lived assets, including property and equipment, right-of-use lease assets and finite-lived intangible assets, to determine whether current events or circumstances indicate that such carrying amounts may not be recoverable. If such facts or circumstances are determined to exist, an estimate of the undiscounted future cash flows of these assets is compared to the carrying value of the assets to determine whether impairment exists. If the assets are determined to be impaired, the loss is measured based on the difference between the fair value and carrying value of the assets. If we determine that events and circumstances warrant a revision to the remaining period of amortization or depreciation for a specific long-lived asset, its remaining estimated useful life will be revised, and the remaining carrying amount of the long-lived asset will be depreciated or amortized prospectively over the revised remaining estimated useful life. No impairment loss was recognized for long-lived or intangible assets for any period presented.
Debt Issuance Costs
Costs incurred in connection with obtaining new debt financing are deferred and amortized over the life of the related financing. If such financing is settled or replaced prior to maturity with debt instruments that have substantially different terms, the settlement is treated as an extinguishment and the unamortized costs are charged to gain or loss on extinguishment of debt. If such financing is settled or replaced with debt instruments from the same lender that do not have substantially different terms, the new debt agreement is accounted for as a modification for the prior debt agreement and the unamortized costs remain capitalized, the new original issuance discount costs are capitalized, and any new third-party costs are charged to expense. Deferred costs are recognized as a direct reduction in the carrying amount of the debt instrument on the consolidated balance sheets and are amortized to interest expense over the term of the related debt using the effective interest method.
Accumulated Other Comprehensive Income (Loss)
Comprehensive income (loss) and its components encompass all changes in equity other than those with stockholders or member. Comprehensive income (loss) for the Company consists of foreign currency translation adjustments. There were no reclassifications out of accumulated other comprehensive loss during the periods presented.
Fair Value of Financial Instruments
The Company defines fair value as the amount that would be received to sell an asset, or paid to transfer a liability, in an orderly transaction between market participants at the measurement date. The Company follows accounting guidance that has a three-level hierarchy for fair value measurements based upon the transparency of inputs to the valuation of the asset or liability as of the measurement date. Instruments with readily available actively quoted prices, or for which fair value can be measured from actively quoted prices in an orderly market, will generally have a higher degree of market price transparency and a lesser degree of judgment used in measuring fair value. The three levels of the hierarchy are defined as follows:
Level 1—Observable inputs that reflect quoted prices (unadjusted) for identical assets or liabilities in active markets;
Level 2—Include other inputs that are directly or indirectly observable in the marketplace; and
Level 3—Unobservable inputs which are supported by little or no market activity.
As of December 31, 2023 and 2022, the carrying value of current assets and liabilities approximates fair value due to the short maturities of these instruments. The fair values of the Company’s long-term debt approximate carrying value, excluding the effect of unamortized debt discount, as it is based on borrowing rates currently available to the Company for debt with similar terms and maturities (Level 2 inputs).
Acquisitions
The Company evaluates mergers, acquisitions and other similar transactions to assess whether or not the transaction should be accounted for as a business combination or an acquisition of assets. The Company first identifies the acquiring entity by determining if the target is a legal entity or a group of assets or liabilities. If control over a legal entity is being evaluated, the Company also evaluates if the target is a variable interest or voting interest entity. For acquisitions of voting interest entities, the
Company applies a screen test to determine if substantially all of the fair value of the gross assets acquired is concentrated in a single identifiable asset or group of similar identifiable assets. If the screen test is met, the transaction is accounted for as an acquisition of assets. If the screen is not met, further determination is required as to whether or not the Company has acquired inputs and processes that have the ability to create outputs which would meet the definition of a business.
The Company accounts for its business combinations using the acquisition method of accounting which requires that the assets acquired and liabilities assumed of acquired businesses be recorded at their respective fair values at the date of acquisition. The purchase price, which includes the fair value of consideration transferred, is attributed to the fair value of the assets acquired and liabilities assumed. The purchase price may also include contingent consideration. The Company assesses whether such contingent consideration is subject to liability classification and fair value measurement or meets the definition of a derivative. Contingent consideration liabilities are recognized at their estimated fair value on the acquisition date. Contingent consideration arrangements that are determined to be compensatory in nature are recognized as post combination expense in our consolidated statements of operations ratably over the implied service period beginning in the period it becomes probable such amounts will become payable. The excess of the purchase price of the acquisition over the fair value of the identifiable net assets of the acquiree is recorded as goodwill. The fair value of assets acquired and liabilities assumed in certain cases may be subject to revision based on the final determination of fair value during a period of time not to exceed twelve months from the acquisition date. The results of acquired businesses are included in the Company’s consolidated financial statements from the date of acquisition. Transaction costs directly attributable to acquired businesses are expensed as incurred.
Determining the fair value of assets acquired and liabilities assumed requires management to use significant judgment and estimates, including the selection of valuation methodologies and assumptions about future net cash flows, discount rates and market participants. Each of these factors can significantly affect the value attributed to the identifiable intangible asset acquired in a business combination.
Contingent Consideration
Contingent consideration represents additional consideration that may be transferred to former owners of an acquired entity in the future if certain future events occur or conditions are met. Contingent consideration resulting from the acquisition of a business is recorded at fair value on the acquisition date. Such contingent consideration is re-measured to its estimated fair value at each reporting date with the change in fair value recognized within operating expenses in the Company’s consolidated statements of operations. Subsequent changes in the fair value of the contingent consideration are classified as an adjustment to cash flows from operating activities in the consolidated statements of cash flows because the change in fair value is an input in determining net (loss) income. Cash paid in settlement of contingent consideration liabilities are classified as cash flows from financing activities up to the acquisition date fair value with any excess classified as cash flows from operating activities.
Changes in the fair value of contingent consideration liabilities associated with the acquisition of a business can result from updates to assumptions such as the expected timing or probability of achieving customer-related performance targets, specified sales milestones, changes in projected revenue or changes in discount rates. Judgment is used in determining those assumptions as of the acquisition date and for each subsequent reporting period. Therefore, any changes in the fair value will impact the Company’s results of operations in such reporting period, thereby resulting in potential variability in the Company’s operating results until such contingencies are resolved.
Leases
The Company determines whether the arrangement is or contains a lease based on the unique facts and circumstances present at the inception of the arrangement and if such a lease is classified as a finance lease or operating lease. Finance leases with a term greater than one year are included in property and equipment, current portion of finance lease liabilities, and finance lease liabilities, less current portion on our consolidated balance sheets. Operating leases with a term greater than one year are included in other assets, accrued expenses and other current liabilities, and other long-term liabilities on our consolidated balance sheets. The Company has elected not to recognize on the consolidated balance sheet leases with terms of one year or less.
Right-of-use (“ROU”) assets represents the Company’s right to use an underlying asset for the lease term and lease liabilities represent the Company’s obligation to make lease payments arising from the lease contract. Lease liabilities and their corresponding ROU assets are recorded based on the present value of lease payments over the expected lease term. In determining the net present value of lease payments, the interest rate implicit in lease contracts is typically not readily determinable. As such, the Company utilizes the appropriate incremental borrowing rate, which is the rate incurred to borrow on a collateralized basis over a similar term an amount equal to the lease payments in a similar economic environment. Certain adjustments to the ROU asset may be required for items such as initial direct costs paid or incentives received and impairment charges if we determine the ROU asset is impaired.
The Company considers a lease term to be the noncancelable period that it has the right to use the underlying asset, including any periods where it is reasonably assured the Company will exercise the option to extend the contract. Periods covered by an option to extend are included in the lease term if the lessor controls the exercise of that option.
The Company recognizes lease expense on a straight-line basis over the expected lease term. Variable lease payments, for items such as maintenance and utilities, are not included in the calculation of the ROU asset and the related lease liability and are recognized as this lease expense is incurred.
The Company has elected to not separate lease and non-lease components for its leased assets and accounts for all lease and non-lease components of its agreements as a single lease component. The lease components resulting in a ROU asset have been recorded on the balance sheet and amortized as lease expense on a straight-line basis over the lease term.
Concentration of Credit Risk
Financial instruments that potentially subject the Company to significant concentrations of credit risk consist principally of cash and accounts receivable. The Company maintains the majority of its cash balances at multiple financial institutions that management believes are of high-credit quality and financially stable. Cash is deposited with major financial institutions in excess of Federal Deposit Insurance Corporation (“FDIC”) insurance limits. The Company believes it is not exposed to significant credit risk due to the financial strength of the depository institutions in which the cash is held. The Company provides credit, in the normal course of business, to international and domestic distributors as well as certain customers, which are geographically dispersed. The Company attempts to limit its credit risk by performing ongoing credit evaluations of its customers and maintaining adequate allowances for potential credit losses.
The following table summarizes revenue from each of our customers who individually accounted for 10% or more of our total revenue or accounts receivable for the periods presented:
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Revenue | | Accounts Receivable, net |
| Years Ended December 31, | | As of December 31, |
| 2023 | | 2022 | | 2021 | | 2023 | | 2022 |
| Nacalai USA, Inc. | 19.3 | % | | * | | * | | 27.3 | % | | 20.3 | % |
| CureVac N.V. | * | | * | | 15.3 | % | | 13.0 | % | | 15.7 | % |
| BioNTech SE | * | | 34.8 | % | | 29.5 | % | | * | | 12.0 | % |
| Pfizer Inc. | * | | 26.4 | % | | 23.3 | % | | * | | 19.2 | % |
____________________
*Less than 10%
For the year ended December 31, 2023, all of the revenue recorded for Nacalai USA, Inc. was generated by the Nucleic Acid Production Segment. For the year ended December 31, 2022, substantially all of the revenue recorded for BioNTech SE and Pfizer Inc. was generated by our Nucleic Acid Production segment. For the year ended December 31, 2021, substantially all of the revenue recorded for BioNTech SE, Pfizer Inc and CureVac N.V. was generated by our Nucleic Acid Production segment.
Net (Loss) Income per Class A Common Share Attributable to Maravai LifeSciences Holdings, Inc.
Basic net (loss) income per Class A common share attributable to Maravai LifeSciences Holdings, Inc. is computed by dividing net (loss) income attributable to us by the weighted average number of Class A common shares outstanding during the period. Diluted net income per Class A common share is calculated by giving effect to all potential weighted average dilutive stock options, restricted stock units, performance stock units and Topco LLC Units, that together with an equal number of shares of our Class B common stock are convertible into shares of our Class A common stock. The dilutive effect of outstanding awards, if any, is reflected in diluted earnings per share by application of the treasury stock method or if-converted method, as applicable. In periods in which the Company reports a net loss attributable to Maravai LifeSciences Holdings, Inc., diluted net loss per Class A common share attributable to the Company is the same as basic net loss per Class A common share attributable to the Company, since dilutive equity instruments are not assumed to have been issued if their effect is anti-dilutive. The Company reported a net loss attributable to Maravai LifeSciences Holdings, Inc. for the year ended December 31, 2023.
Recently Adopted Accounting Pronouncements Not Yet Adopted
In November 2023, the Financial Accounting Standards Board (“FASB”) issued Accounting Standards Update (“ASU”) 2023-07, Segment Reporting (Topic 280) - Improvements to Reportable Segment Disclosures (“ASU 2023-07”), which improves segment disclosure requirements, primarily through enhanced disclosures about significant expenses. ASU 2023-07 requires disclosures to include significant segment expenses that are regularly provided to the CODM and included within each
reported measure of segment profit or loss, an amount for other segment items by reportable segment and a description of its composition, any additional measures of a segment’s profit or loss used by the CODM when deciding how to allocate resources, and the title and position of the CODM and an explanation of how the CODM uses the reported measures of segment profit or loss in assessing segment performance and deciding how to allocate resources. The ASU also requires all annual disclosures currently required by Topic 280 to be included in interim periods. ASU 2023-07 is effective for the Company for fiscal years beginning after December 15, 2023, and interim periods within fiscal years beginning after December 15, 2024, with early adoption permitted. The amendments in this ASU should be applied retrospectively to all prior periods presented in the consolidated financial statements. The Company is currently evaluating the impact of adopting this standard on its consolidated financial statements and disclosures.
In December 2023, the FASB issued ASU 2023-09, Income Taxes (Topic 740) - Improvements to Income Tax Disclosures (“ASU 2023-09”). The amendments in this ASU address investor requests for more transparency about income tax information through improvements to tax disclosures primarily related to the rate reconciliation and income taxes paid information. The ASU also includes certain other amendments to improve the effectiveness of income tax disclosures. ASU 2023-09 is effective for the Company for annual periods beginning after December 15, 2024, with early adoption permitted. The amendments in this ASU should be applied on a prospective basis, with retrospective application permitted. The Company is currently evaluating the impact of adopting this standard on its consolidated financial statements and disclosures.
2.Acquisitions and Divestiture
Acquisitions
Alphazyme, LLC
On January 18, 2023, the Company completed the acquisition of Alphazyme, LLC (“Alphazyme”), a privately-held original equipment manufacturer (“OEM”) and provider of custom, scalable, molecular biology enzymes to customers in the genetic analysis and nucleic acid synthesis markets. The acquisition will expand the Company’s internal enzyme product portfolio and increase the Company’s differentiated mRNA manufacturing services and product offerings. Alphazyme’s ability to manufacture custom enzymes allows the Company to expand into near adjacent markets and raise our enzyme vertical.
The Company acquired Alphazyme for a total purchase consideration of $75.3 million, which is inclusive of net working capital adjustments. As a result of the acquisition, the Company owns all the outstanding equity interest in Alphazyme. The total cash consideration was paid using existing cash on hand. The transaction was accounted for as an acquisition of a business as Alphazyme consisted of inputs and processes applied to those inputs that had the ability to contribute to the creation of outputs.
For the year ended December 31, 2023, the Company incurred $4.1 million in transaction costs associated with the acquisition of Alphazyme, which were recorded within selling, general and administrative expenses in the consolidated statements of operations.
The acquisition date fair value of consideration transferred to acquire Alphazyme consisted of the following (in thousands):
| | | | | |
Cash paid (1) | $ | 70,037 | |
| Fair value of contingent consideration | 5,289 | |
| Total consideration transferred | $ | 75,326 | |
____________________
(1)Represents cash consideration paid at closing of $70.1 million, net of a purchase price adjustment received in June 2023 of $0.1 million.
Pursuant to the Securities Purchase Agreement (the “Alphazyme SPA”) between the Company and sellers of Alphazyme, additional payments to the sellers of Alphazyme are dependent upon meeting or exceeding defined revenue targets during fiscal years 2023 through 2025 (the “Alphazyme Performance Payments”). The Alphazyme SPA provides for a total maximum Alphazyme Performance Payments of $75.0 million. The Alphazyme Performance Payments were recorded as contingent consideration and was included as part of the purchase consideration. The Company estimated the fair value of the Alphazyme Performance Payments contingent consideration based on a Monte-Carlo simulation model which utilized an income approach. The estimated fair value was based on Alphazyme revenue projections, expected payout term, volatility and risk adjusted discount rates which are Level 3 inputs (see Note 5). The first performance period applicable to the Alphazyme Performance Payments ended on December 31, 2023, and it was determined that the defined revenue target was not achieved. Consequently, no payment was made to the sellers of Alphazyme.
The Alphazyme SPA also provides that the Company will pay certain employees of Alphazyme an additional amount totaling $9.3 million (the “Alphazyme Retention Payments”) as of various dates but primarily through December 31, 2025 as long as these individuals continue to be employed by the Company. The Company considers the payment of the Alphazyme Retention Payments as probable and is recognizing compensation expense related to these payments in the post-acquisition period ratably over the service period of approximately three years. For the year ended December 31, 2023, the Company recorded $2.2 million of compensation expense related to the Alphazyme Retention Payments within selling, general and administrative expenses in the consolidated statements of operations. Compensation expense related to the Alphazyme Retention Payments recorded within cost of revenue and research and development expenses were not material.
The following table summarizes the estimated fair values of the assets acquired and liabilities assumed at the acquisition date (in thousands):
| | | | | |
| Cash | $ | 288 | |
| Inventory | 7,246 | |
| Other current assets | 660 | |
| Intangible assets, net | 31,680 | |
| Other assets | 5,043 | |
| Total identifiable assets acquired | 44,917 | |
| Current liabilities | (482) | |
| Other long-term liabilities | (11,470) | |
| Total liabilities assumed | (11,952) | |
| Net identifiable assets acquired | 32,965 | |
| Goodwill | 42,361 | |
| Net assets acquired | $ | 75,326 | |
We recorded the preliminary purchase price allocation in the first quarter of 2023. During the third quarter of 2023, we recorded a measurement period adjustment resulting in a decrease to goodwill of $0.4 million, with an equal offset to other long-term liabilities.
The acquisition was accounted for under the acquisition method of accounting, and therefore, the total purchase price was allocated to the identifiable tangible and intangible assets acquired and the liabilities assumed based on their respective fair values as of the acquisition date. Purchase consideration in excess of the amounts recognized for the net assets acquired was recognized as goodwill. Goodwill is primarily attributable to expanded synergies expected from the acquisition associated with a vertical supply integration. All of the goodwill acquired in connection with the acquisition of Alphazyme was allocated to the Company’s Nucleic Acid Production segment. None of the goodwill recognized is expected to be deductible for income tax purposes.
Upon closing of the acquisition, approximately $1.5 million was placed into escrow to cover potential working capital adjustments and approximately $3.0 million was placed into escrow to secure certain representations and warranties pursuant to the terms of the Alphazyme SPA. These amounts are included in the total purchase consideration of $75.3 million. The $1.5 million was released from escrow during the second quarter of 2023, of which the Company received $0.1 million related to net working capital adjustments. Because the remaining $3.0 million held in escrow is not controlled by the Company, this amount is not included in the accompanying consolidated balance sheet as of December 31, 2023.
The following table summarizes the estimated fair values of Alphazyme’s identifiable intangible assets as of the date of acquisition and their estimated useful lives:
| | | | | | | | | | | |
| Estimated Fair Value
(in thousands) | | Estimated Useful Life
(in years) |
| Trade names | $ | 220 | | | 5 |
| Developed technology | 31,000 | | | 12 |
| Customer relationships | 460 | | | 12 |
| Total | $ | 31,680 | | | |
The trade name and customer relationship intangible assets are related to Alphazyme’s name, customer loyalty and customer relationships. The developed technology intangible asset is related to its unique manufacturing process optimization capability to both scale production and achieve quality standards. The fair value of these intangible assets was based on Alphazyme’s projected revenues and was estimated using an income approach, specifically the relief from royalty method for trade names, the multi-period excess earnings method for developed technology, and the distributor method for customer relationships. Under the income approach, an intangible asset’s fair value is equal to the present value of future economic benefits to be derived from ownership of the asset. The estimated fair value was developed by discounting future net cash flows to their present value at market-based rates of return utilizing Level 3 inputs. The useful lives for these intangible assets were determined based upon the remaining period for which the assets were expected to contribute directly or indirectly to future cash flows. Key quantitative assumptions used in the determination of fair value of the developed technology intangible included revenue growth rates ranging from 3.0% to 55.0%, a discount rate of 17.8% and an assumed technical obsolescent curve of 5.0%.
The carrying value of the remaining assets acquired or liabilities assumed was estimated to equal their fair values based on their short-term nature. These estimates were based on the assumption that the Company believes to be reasonable; however, actual results may differ from these estimates.
MyChem, LLC
On January 27, 2022, the Company completed the acquisition of MyChem, LLC (“MyChem”), a privately-held San Diego, California-based provider of ultra-pure nucleotides to customers in the diagnostics, pharma, genomics and research markets. The acquisition will vertically integrate the Company’s supply chain and expand its product offerings for inputs used in the development of therapeutics and vaccines.
The Company acquired MyChem for a total purchase consideration of $257.9 million, which is inclusive of net working capital adjustments. As a result of the acquisition, the Company owns all the outstanding equity interest in MyChem. The total cash consideration was paid using existing cash on hand. The transaction was accounted for as an acquisition of a business as MyChem consisted of inputs and processes applied to those inputs that had the ability to contribute to the creation of outputs.
For the year ended December 31, 2022, the Company incurred $3.5 million in transaction costs associated with the acquisition of MyChem, which were recorded within selling, general and administrative expenses in the consolidated statements of operations.
The acquisition date fair value of consideration transferred to acquire MyChem consisted of the following (in thousands):
| | | | | |
Cash paid (1) | $ | 240,145 | |
| Consideration payable | 10,000 | |
| Fair value of contingent consideration | 7,800 | |
| Total consideration transferred | $ | 257,945 | |
____________________
(1)Represents cash consideration paid at closing of $240.0 million and a purchase price adjustment paid in November 2022 of $0.1 million.
Pursuant to the Securities Purchase Agreement (the “MyChem SPA”) between the Company and sellers of MyChem, additional payments to the sellers of MyChem are dependent upon meeting or exceeding defined revenue targets during fiscal 2022 (the “MyChem Performance Payment”). The MyChem SPA provides for a total maximum Performance Payment of $40.0 million. The MyChem Performance Payment was recorded as contingent consideration and was included as part of the purchase consideration. The Company estimated the fair value of the MyChem Performance Payment contingent consideration based on a Monte-Carlo simulation model which utilized an income approach. The estimated fair value was based on MyChem revenue projections, expected payout term, volatility and risk adjusted discount rates which are Level 3 inputs (see Note 5). The performance period applicable to the MyChem Performance Payment ended as of December 31, 2022 and it was determined that none of the defined revenue thresholds were achieved. Consequently, no payment was made to the sellers of MyChem.
The MyChem SPA also provides that the Company will pay to the sellers of MyChem an additional $20.0 million (the “MyChem Retention Payment”) as of the second anniversary of the closing of the acquisition date as long as two senior employees who are also the sellers of MyChem continue to be employed by TriLink. The Company considers the payment of the Retention Payment as probable and is recognizing compensation expense related to this payment in the post-acquisition period ratably over the expected service period of two years. For the year ended December 31, 2023, the Company recorded $4.3 million of compensation expense related to the MyChem Retention Payment within cost of revenue in the consolidated statements of operations. For the years ended December 31, 2023 and 2022, the Company recorded $5.1 million and $9.3
million, respectively, of compensation expense related to the MyChem Retention Payment within research and development expenses in the consolidated statements of operations.
The MyChem SPA further provides that the Company will pay to the sellers of MyChem an additional amount of up to $10.0 million subject to the completion of certain calculations associated with acquired inventory, which has been recorded within accrued expenses and other current liabilities on the consolidated balance sheet as of December 31, 2022. During the first quarter of 2023, but subsequent to the end of the measurement period, these calculations were completed and a payment of $9.7 million was made by the Company to the sellers. The remaining $0.3 million was recorded as non-cash gain within current year operations.
The following table summarizes the estimated fair values of the assets acquired and liabilities assumed at the acquisition date (in thousands):
| | | | | |
| Cash | $ | 1,176 | |
| Current assets | 2,741 | |
| Intangible assets, net | 123,360 | |
| Other assets | 8,585 | |
| Total identifiable assets acquired | 135,862 | |
| Current liabilities | (420) | |
| Other long-term liabilities | (8,399) | |
| Total liabilities assumed | (8,819) | |
| Net identifiable assets acquired | 127,043 | |
| Goodwill | 130,902 | |
| Net assets acquired | $ | 257,945 | |
We recorded the preliminary purchase price allocation in the first quarter of 2022. During the fourth quarter of 2022, we recorded measurement period adjustments resulting in an increase to goodwill of $0.1 million and a decrease to other assets and current liabilities of $0.7 million.
The acquisition was accounted for under the acquisition method of accounting, and therefore, the total purchase price was allocated to the identifiable tangible and intangible assets acquired and the liabilities assumed based on their respective fair values as of the acquisition date. Purchase consideration in excess of the amounts recognized for the net assets acquired was recognized as goodwill. Goodwill is primarily attributable to expanded synergies expected from the acquisition associated with a vertical supply integration. There were no tax impacts associated with the acquisition due to the pass-through income tax treatment of MyChem. All of the goodwill acquired in connection with the acquisition of MyChem was allocated to the Company’s Nucleic Acid Production segment and is deductible to Topco LLC for income tax purposes.
Upon closing of the acquisition, approximately $1.0 million was placed into escrow to cover potential working capital adjustments and approximately $12.5 million was placed into escrow to secure certain representations and warranties pursuant to the terms of the MyChem SPA. These amounts are included in the total purchase consideration of $257.9 million. The Company released the $1.0 million in escrow and paid out an additional $0.1 million related to net working capital adjustments during the fourth quarter of 2022. During the first quarter of 2023, but subsequent to the end of the measurement period, $12.4 million of the amounts in escrow to secure certain representations and warranties was released to the sellers and the remaining $0.1 million was released to the Company for indemnification of pre-closing liabilities, which was recorded within current year operations.
The following table summarizes the estimated fair values of MyChem’s identifiable intangible assets as of the date of acquisition and their estimated useful lives:
| | | | | | | | | | | |
| Estimated Fair Value
(in thousands) | | Estimated Useful Life
(in years) |
| Trade names | $ | 460 | | | 3 |
| Developed technology | 121,000 | | | 12 |
| Customer relationships | 1,900 | | | 12 |
| Total | $ | 123,360 | | | |
The trade name and customer relationship intangible assets are related to MyChem’s name, customer loyalty and customer relationships. The developed technology intangible asset is related to processes and techniques for synthesizing and developing ultra-pure nucleotides. The fair value of these intangible assets was based on MyChem’s projected revenues and was estimated using an income approach, specifically the multi-period excess earnings method. Under the income approach, an intangible asset’s fair value is equal to the present value of future economic benefits to be derived from ownership of the asset. The estimated fair value was developed by discounting future net cash flows to their present value at market-based rates of return utilizing Level 3 inputs. The useful lives for these intangible assets were determined based upon the remaining period for which the assets were expected to contribute directly or indirectly to future cash flows. Key quantitative assumptions used in the determination of fair value of the developed technology intangible included revenue growth rates ranging from 3.0% to 30.6%, a discount rate of 16.5% and an assumed technical obsolescent curve range of 5.0% to 7.5%.
Pursuant to the terms of the MyChem SPA, the Company recognized an indemnification asset of $8.0 million within other assets, which represented the seller’s obligation to reimburse pre-acquisition income tax liabilities assumed in the acquisition and was recorded within other long-term liabilities.
The carrying value of the remaining assets acquired or liabilities assumed was estimated to equal their fair values based on their short-term nature.
Divestiture
Vector Laboratories, Inc.
In August 2021, the Company entered into a definitive agreement to sell Vector to Voyager Group Holdings, Inc. (“Voyager”), a third-party unrelated to the Company, for an all cash sale price of $124.0 million, subject to purchase price adjustments. The Company determined that the fair value of Vector, less estimated costs to sell, exceeded the book value of the Vector Disposal Group and there were no other indicators of asset impairment prior to the sale. The divestiture was completed in September 2021, and final net proceeds were $120.7 million, which were inclusive of working capital adjustments.
As a result of the divestiture, during the year ended December 31, 2021, the Company recognized a pre-tax gain on sale of $11.2 million, net of transactions costs of $0.9 million, in the consolidated statements of operations.
The Company’s Protein Detection segment was comprised of Vector. The sale of Vector represents a strategic shift as the Company will no longer be in the protein detection business after the sale. However, the sale did not qualify for presentation as discontinued operations since the sale of the Protein Detection segment did not have a major effect on the Company’s operations or financial results.
In connection with the divestiture, the Company entered into a Transition Services Agreement (“TSA”) with Voyager to help support its ongoing operations. Under the TSA, the Company will provide certain transition services to Voyager, including information technology, finance and ERP, marketing and commercial, human resources, employee benefits, and other limited services. Depending on the service, the initial period ranges from one month to five months and the extension period ranges from one month to eight months. Income from performing services under the TSA was recorded within other income in the consolidated statements of operations and was not significant for the year ended December 31, 2021.
In August 2020, the Company entered into an agreement with an executive of Vector whereby the executive received incentive units of MLSH 1. In connection with the divestiture, MLSH 1 amended this executive’s incentive units resulting in the recognition of incremental unit-based compensation expense in the Company’s consolidated financial statements of $2.4 million. This unit-based compensation expense was recorded within selling, general and administrative expenses in the consolidated statements of operations for the year ended December 31, 2021.
3.Restructuring
In November 2023, the Company implemented a cost realignment plan (the “Cost Realignment Plan”) that included the termination of approximately 15% of the Company’s workforce, the termination of certain leases, and other actions to reduce expenses, all as part of a plan to optimize business operations and match them to current market conditions. The reduction in force was completed on January 5, 2024, following the end of the sixty-day notification period required by the Worker Adjustment and Retraining Notification Act. The Company expects the remaining actions under the Cost Realignment Plan to be substantially complete during the first quarter of 2024.
The Company’s restructuring charges by segment and unallocated corporate costs, which are recorded as restructuring expenses on the consolidated statements of operations, were as follows for the year ended December 31, 2023 (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Severance and Other Employee Costs | | Stock-Based Compensation Expense (Benefit) | | Facility and Other Exit Costs | | Professional Fees and Other | | Total |
Nucleic Acid Production | $ | 2,470 | | | $ | 168 | | | $ | 638 | | | $ | 190 | | | $ | 3,466 | |
| | | | | | | | | |
Corporate | 1,833 | | | (269) | | | 1,351 | | | 85 | | | 3,000 | |
Total | $ | 4,303 | | | $ | (101) | | | $ | 1,989 | | | $ | 275 | | | $ | 6,466 | |
The following table summarizes the activity for accrued restructuring costs, which is recorded within accrued expenses and other current liabilities on the consolidated balance sheets, for the period presented (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| Severance and Other Employee Costs | | Stock-Based Compensation Expense (Benefit) | | Facility and Other Exit Costs | | Professional Fees and Other | | Total |
| Balance as of December 31, 2022 | $ | — | | | $ | — | | | $ | — | | | $ | — | | | $ | — | |
Charges | 4,303 | | | (101) | | | 1,989 | | | 275 | | | 6,466 | |
Non-cash charges | — | | | 101 | | | — | | | — | | | 101 | |
Cash payments | (1,760) | | | — | | | (1,989) | | | (4) | | | (3,753) | |
| Balance as of December 31, 2023 | $ | 2,543 | | | $ | — | | | $ | — | | | $ | 271 | | | $ | 2,814 | |
The Company does not expect to incur additional restructuring costs relating to the Cost Realignment Plan, however the Company expects an additional benefit totaling $1.2 million for the forfeiture of equity awards upon the termination of certain impacted employees in January 2024, of which $0.8 million relates to the Nucleic Acid Production segment and $0.4 million relates to unallocated corporate costs.
4.Goodwill and Intangible Assets
The Company’s goodwill of $326.0 million and $283.7 million as of December 31, 2023 and 2022, respectively, represents the excess of purchase consideration over the fair value of assets acquired and liabilities assumed. As of December 31, 2023, the Company had four reporting units, three of which are contained in the Nucleic Acid Production segment. During the year ended December 31, 2023, the Company recorded goodwill of $42.4 million in connection with the acquisition of Alphazyme that was completed in January 2023 (see Note 2). As of December 31, 2022, the Company had three reporting units, two of which were contained in the Nucleic Acid Production segment.
Due to the sustained decline in its stock price and the announcement of the Cost Realignment Plan in November 2023, the Company performed a quantitative goodwill impairment analysis on each of its four reporting units during the fourth quarter of 2023 and concluded that the fair value of goodwill exceeded its carrying value. The Company has not recognized any goodwill impairment charges in any of the periods presented.
The following table summarizes the activity in the Company’s goodwill by segment for the period presented (in thousands):
| | | | | | | | | | | | | | | | | |
| Nucleic Acid Production | | Biologics Safety Testing | | Total |
| Balance as of December 31, 2022 | $ | 163,740 | | | $ | 119,928 | | | $ | 283,668 | |
| Acquisition | 42,361 | | | — | | | 42,361 | |
| | | | | |
| Balance as of December 31, 2023 | $ | 206,101 | | | $ | 119,928 | | | $ | 326,029 | |
Intangible assets are being amortized on a straight-line basis, which reflects the expected pattern in which the economic benefits of the intangible assets are being obtained, over an estimated useful life ranging from 3 to 14 years.
The following are components of finite-lived intangible assets and accumulated amortization as of the periods presented (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| December 31, 2023 |
| Gross
Carrying
Amount | | Accumulated
Amortization | | Net
Carrying
Amount | | Estimated
Useful
Life | | Weighted
Average
Remaining
Amortization
Period |
| | | (in thousands) | | | | (in years) | | (in years) |
| Trade Names | $ | 7,800 | | | $ | 6,369 | | | $ | 1,431 | | | 3 - 10 | | 2.8 |
| Patents and Developed Technology | 319,649 | | | 109,800 | | | 209,849 | | | 10 - 14 | | 8.9 |
| Customer Relationships | 22,313 | | | 12,606 | | | 9,707 | | | 10 - 12 | | 5.9 |
| Total | $ | 349,762 | | | $ | 128,775 | | | $ | 220,987 | | | | | 8.7 |
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| December 31, 2022 |
| Gross Carrying Amount | | Accumulated Amortization | | Net Carrying Amount | | Estimated Useful Life | | Weighted Average Remaining Amortization Period |
| | | (in thousands) | | | | (in years) | | (in years) |
| Trade Names | $ | 7,580 | | | $ | 5,746 | | | $ | 1,834 | | | 3 - 10 | | 3.5 |
| Patents and Developed Technology | 288,649 | | | 85,058 | | | 203,591 | | | 10 - 14 | | 9.5 |
| Customer Relationships | 21,853 | | | 10,615 | | | 11,238 | | | 10 - 12 | | 6.5 |
| Total | $ | 318,082 | | | $ | 101,419 | | | $ | 216,663 | | | | | 9.3 |
During the first quarter of 2023, the Company recorded intangible assets of $31.7 million in connection with the acquisition of Alphazyme that was completed in January 2023 (see Note 2).
The Company recognized $24.8 million, $21.5 million and $12.4 million of amortization expense from intangible assets directly linked with revenue generating activities within cost of revenue in the consolidated statements of operations for the years ended December 31, 2023, 2022 and 2021, respectively. Amortization expense for intangible assets that are not directly related to sales generating activities of $2.6 million, $2.8 million and $5.9 million was recorded as selling, general and administrative expenses for the years ended December 31, 2023, 2022 and 2021, respectively.
As of December 31, 2023, the estimated future amortization expense for finite-lived intangible assets were as follows (in thousands):
| | | | | |
| 2024 | $ | 27,478 | |
| 2025 | 27,335 | |
| 2026 | 27,098 | |
| 2027 | 26,082 | |
| 2028 | 25,862 | |
| Thereafter | 87,132 | |
| Total estimated amortization expense | $ | 220,987 | |
5.Fair Value Measurements
The following table summarizes the Company’s financial assets and liabilities that are measured at fair value on a recurring basis by level within the fair value hierarchy as of the periods presented (in thousands):
| | | | | | | | | | | | | | | | | | | | | | | |
| Fair Value Measurements as of December 31, 2023 |
| Level 1 | | Level 2 | | Level 3 | | Total |
| Assets | | | | | | | |
Money market funds | $ | 418,685 | | | $ | — | | | $ | — | | | $ | 418,685 | |
| Interest rate cap | — | | | 8,559 | | | — | | | 8,559 | |
| Total assets | $ | 418,685 | | | $ | 8,559 | | | $ | — | | | $ | 427,244 | |
| | | | | | | |
| Liabilities | | | | | | | |
| Current portion of contingent consideration | $ | — | | | $ | — | | | $ | 131 | | | $ | 131 | |
| Contingent consideration, non-current | — | | | — | | | 1,872 | | | 1,872 | |
| Total liabilities | $ | — | | | $ | — | | | $ | 2,003 | | | $ | 2,003 | |
| | | | | | | | | | | | | | | | | | | | | | | |
| Fair Value Measurements as of December 31, 2022 |
| Level 1 | | Level 2 | | Level 3 | | Total |
| Assets | | | | | | | |
| Interest rate cap | $ | — | | | $ | 11,362 | | | $ | — | | | $ | 11,362 | |
| | | | | | | |
| | | | | | | |
Contingent Consideration
In connection with the acquisition of Alphazyme (see Note 2), the Company is required to make contingent payments to the sellers of up to $75.0 million, subject to achieving certain revenue thresholds. The preliminary fair value of the liability for the contingent payments recognized upon the acquisition as part of the purchase accounting opening balance sheet totaled $5.3 million. The preliminary fair value of the contingent consideration was determined using a Monte-Carlo simulation-based model discounted to present value. Assumptions used in this calculation are expected revenue, a discount rate of 17.8% and various probability factors. The ultimate settlement of the contingent consideration could deviate from current estimates based on the actual results of these financial measures. The contingent consideration has three performance payments spanning over three years beginning 2024. This liability is considered to be a Level 3 financial liability that is remeasured each reporting period. Changes in fair value of contingent consideration are recognized as a gain or loss and recorded within change in estimated fair value of contingent consideration in the consolidated statements of operations. During the year ended December 31, 2023, the Company recorded a decrease of $3.3 million in the estimated fair value of contingent consideration. This was due to a change in estimates associated with Alphazyme revenue projections reaching thresholds that would trigger a contingent payment per the Alphazyme SPA.
In connection with the acquisition of MyChem (see Note 2), the Company is required to make contingent payments to the sellers of up to $40.0 million, subject to achieving certain revenue thresholds. The preliminary fair value of the liability for the contingent payments recognized upon the acquisition as part of the purchase accounting opening balance sheet totaled $7.8 million. The preliminary fair value of the contingent consideration was determined using a Monte-Carlo simulation-based model discounted to present value. Assumptions used in this calculation are expected revenue, a discount rate of 16.9% and various probability factors. The ultimate settlement of the contingent consideration could deviate from current estimates based on the actual results of these financial measures. The contingent consideration projected year of payment was 2023. This liability is considered to be a Level 3 financial liability that is remeasured each reporting period. Changes in fair value of contingent consideration are recognized as a gain or loss and recorded within change in estimated fair value of contingent consideration in the consolidated statements of operations. During the second quarter of 2022, the Company recorded a $7.8 million decrease in the estimated fair value of contingent consideration. This was due to a change in the estimate associated with MyChem revenue projections reaching thresholds that would trigger a contingent payment per the MyChem SPA. The contingent consideration expired as of December 31, 2022 and the revenue thresholds were not achieved.
The following table provides a reconciliation of liabilities measured at fair value on a recurring basis using significant unobservable inputs (Level 3) for the periods presented (in thousands):
| | | | | |
| Contingent Consideration |
| Balance as of December 31, 2021 | $ | — | |
| Contingent consideration related to the acquisition of MyChem | 7,800 | |
| Change in estimated fair value of contingent consideration | (7,800) | |
| Balance as of December 31, 2022 | — | |
| Contingent consideration related to the acquisition of Alphazyme | 5,289 | |
| Change in estimated fair value of contingent consideration | (3,286) | |
| Balance as of December 31, 2023 | $ | 2,003 | |
6.Balance Sheet Components
Inventory
Inventory consisted of the following as of the periods presented (in thousands):
| | | | | | | | | | | |
| December 31, 2023 | | December 31, 2022 |
| Raw materials | $ | 19,338 | | | $ | 13,486 | |
| Work-in-process | 12,680 | | | 21,950 | |
| Finished goods | 19,379 | | | 7,716 | |
| Total inventory | $ | 51,397 | | | $ | 43,152 | |
Property and equipment
Property and equipment consisted of the following as of the periods presented (in thousands):
| | | | | | | | | | | |
| December 31, 2023 | | December 31, 2022 |
Finance lease right-of-use assets | $ | 78,599 | | | $ | — | |
| Leasehold improvements | 24,874 | | | 20,095 | |
| Furniture, fixtures, and equipment | 48,793 | | | 35,907 | |
| Software | 3,211 | | | 3,004 | |
| Total | 155,477 | | | 59,006 | |
| Less accumulated depreciation | (32,214) | | | (19,502) | |
| Total | 123,263 | | | 39,504 | |
| Construction in-progress | 39,637 | | | 13,190 | |
| Total property and equipment, net | $ | 162,900 | | | $ | 52,694 | |
Depreciation expense totaled approximately $12.9 million, $7.6 million and $6.4 million for the years ended December 31, 2023, 2022 and 2021, respectively.
Other assets
Other assets consisted of the following as of the periods presented (in thousands):
| | | | | | | | | | | |
| December 31, 2023 | | December 31, 2022 |
Operating lease right-of-use assets | $ | 59,746 | | | $ | 63,896 | |
| Interest rate cap | 8,559 | | | 11,362 | |
Indemnification asset (see Note 2) | 6,388 | | | 7,682 | |
| Prepaid lease payments | — | | | 27,253 | |
| Other | 2,929 | | | 5,396 | |
| Total other assets | $ | 77,622 | | | $ | 115,589 | |
Accrued expenses and other current liabilities
Accrued expenses consisted of the following as of the periods presented (in thousands):
| | | | | | | | | | | |
| December 31, 2023 | | December 31, 2022 |
Accrued MyChem Retention Payments, current portion (see Note 2) | $ | 19,446 | | | $ | — | |
| Employee related | 12,905 | | | 19,873 | |
| Accrued interest payable | 9,202 | | | 7,700 | |
Operating lease liabilities, current portion | 6,780 | | | 6,269 | |
Accrued restructuring costs (see Note 3) | 2,814 | | | — | |
| Professional services | 2,277 | | | 4,093 | |
| Customer deposits | 2,156 | | | 1,665 | |
| Sales and use tax liability | 1,001 | | | 1,029 | |
| | | |
| | | |
| Inventory holdback liability | — | | | 10,000 | |
| Other | 3,656 | | | 2,742 | |
| Total accrued expenses and other current liabilities | $ | 60,237 | | | $ | 53,371 | |
Other long-term liabilities
Other long-term liabilities consisted of the following as of the periods presented (in thousands):
| | | | | | | | | | | |
| December 31, 2023 | | December 31, 2022 |
Operating lease liabilities, non-current | $ | 47,510 | | | $ | 51,556 | |
Acquisition related tax liability (see Note 2) | 6,388 | | | 7,682 | |
Accrued Alphayzme Retention Payments, non-current (see Note 2) | 3,202 | | | — | |
Contingent consideration, non-current | 1,872 | | | — | |
Accrued MyChem Retention Payments, non-current (see Note 2) | — | | | 9,324 | |
| Other | 522 | | | 413 | |
| Total other long-term liabilities | $ | 59,494 | | | $ | 68,975 | |
7.Government Assistance
Cooperative Agreement
In May 2022, TriLink entered into a cooperative agreement (the “Cooperative Agreement”) with the U.S. Department of Defense, as represented by the Joint Program Executive Office for Chemical, Biological, Radiological and Nuclear Defense on behalf of the Biomedical Advanced Research and Development Authority (“BARDA”), within the U.S. Department of Health and Human Services (“HHS”), to advance the development of domestic manufacturing capabilities and to expand TriLink’s domestic production capacity in its San Diego manufacturing campus (the “Flanders San Diego Facility”) for products critical to the development and manufacture of mRNA vaccines and therapeutics. The Cooperative Agreement has since transitioned from the U.S. Department of Defense to the HHS as of January 2023. The Flanders San Diego Facility consists of two buildings (“Flanders I” and “Flanders II”), however, the Cooperative Agreement is exclusively involved in Flanders I.
The Cooperative Agreement requires the Company to provide the U.S. Government with conditional priority access and certain preferred pricing obligations for a 10-year period from the completion of the construction project for the production of a medical countermeasure (or a component thereof) that the Company manufactures in the Flanders San Diego Facility during a declared public health emergency.
Pursuant to certain requirements, BARDA awarded TriLink an amount equal to $38.8 million or 50% of the construction and validation costs currently budgeted for the Flanders San Diego Facility. The contract period of performance is May 2022 through January 2034, which is the effective date of the Cooperative Agreement through the anticipated expiration of the 10-year conditional priority access period. Amounts reimbursed are subject to audit and may be recaptured by the HHS in certain circumstances.
During the year ended December 31, 2023, the Company has received $12.9 million of reimbursements under the Cooperative Agreement with an equal offset recorded to property and equipment on the consolidated balance sheet. As of December 31, 2023, the Company has recorded a receivable of $1.1 million, with an equal offset to property and equipment on the consolidated balance sheet.
During the year ended December 31, 2022, the Company has received $18.1 million of reimbursements under the Cooperative Agreement, with offsets recorded to: (i) prepaid lease payments associated with Flanders I within other assets of $17.0 million; and (ii) property and equipment of $1.1 million. As of December 31, 2022, the Company has recorded a receivable of $8.2 million, with an equal offset recorded to prepaid lease payments associated with Flanders I within other assets on the consolidated balance sheet.
8.Leases
All of the Company's facilities, including office, laboratory and manufacturing space, are occupied under long-term non-cancelable lease arrangements with various expiration dates through 2038, some of which include options to extend up to 20 years. The Company does not have any leases that include residual value guarantees.
In January 2023, the Company assumed Alphazyme’s existing facility lease in Jupiter, Florida, in connection with the acquisition of Alphazyme (see Note 2). The lease term began in January 2023 and will end in January 2032. The lease is for 10 years with the option to extend for one additional 5-year period.
In February 2023, the Company entered into an agreement to expand the existing Alphazyme facility lease for additional space. The lease term will run concurrently with and as part of the initial lease term.
In March 2023 and June 2023, the Company’s leases for Flanders I and Flanders II, respectively, commenced. The Company entered into the lease agreement in August 2021. The leases are for eleven years with the option to extend for one additional 5-year period. The Company is reasonably certain to execute the renewal option and has, therefore, recognized this as part of its ROU assets and lease liabilities. The lease includes tenant improvement provisions, rent abatement clauses, and escalating rent payments over the life of the lease.
In December 2023, as part of the Cost Realignment Plan, the Company terminated a facility lease in San Diego, California and recorded a non-cash loss for early lease termination in the consolidated statements of operations (see Note 3).
The Company has a $0.5 million outstanding letter of credit as security for a lease agreement for a facility in San Diego, California, which reduced the availability of credit under the Revolving Credit Facility (see Note 10).
The following table presents supplemental balance sheet information related to the Company's leases as of the periods presented below (in thousands):
| | | | | | | | | | | | | | | | | |
| Line Item in the Consolidated Balance Sheets | | December 31, 2023 | | December 31, 2022 |
| Right-of-use assets | | | | | |
| Finance leases | Property and equipment, net | | $ | 75,382 | | | $ | — | |
| Operating leases | Other assets | | 59,746 | | | 63,896 | |
| Total right-of-use assets | | $ | 135,128 | | | $ | 63,896 | |
| | | | | |
| Current lease liabilities | | | | | |
| Finance leases | Current portion of finance lease liabilities | | $ | 633 | | | $ | — | |
| Operating leases | Accrued expenses and other current liabilities | | 6,780 | | | 6,269 | |
| Total current lease liabilities | | $ | 7,413 | | | $ | 6,269 | |
| | | | | |
| Non-current lease liabilities | | | | | |
| Finance leases | Finance lease liabilities, less current portion | | $ | 31,897 | | | $ | — | |
| Operating leases | Other long-term liabilities | | 47,510 | | | 51,556 | |
| Total non-current lease liabilities | | $ | 79,407 | | | $ | 51,556 | |
The components of the net lease costs reflected in the Company's consolidated statements of operations were as follows for the periods presented (in thousands):
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 | | 2021 |
| Finance lease costs: | | | | | |
| Depreciation of leased assets | $ | 3,217 | | | $ | — | | | $ | — | |
| Interest on lease liabilities | 1,696 | | | — | | | — | |
| Total finance lease costs | 4,913 | | | — | | | — | |
| | | | | |
| Operating lease costs | 12,417 | | | 8,800 | | | 8,792 | |
| Variable lease costs | 3,940 | | | 2,742 | | | 1,759 | |
| Total lease costs | $ | 21,270 | | | $ | 11,542 | | | $ | 10,551 | |
The weighted average remaining lease term and weighted average discount rate related to the Company's ROU assets and lease liabilities for its leases were as follows as of the periods presented below:
| | | | | | | | | | | |
| December 31, 2023 | | December 31, 2022 |
| Weighted average remaining lease term (in years): | | | |
| Finance leases | 14.2 | | * |
| Operating leases | 7.3 | | 7.9 |
| | | |
| Weighted average discount rate: | | | |
| Finance leases | 8.4 | % | | * |
| Operating leases | 6.7 | % | | 6.5 | % |
____________________
*The Company did not have any finance leases as of December 31, 2022.
Supplemental information concerning the cash flow impact arising from the Company's leases recorded in the Company's consolidated statements of cash flows is detailed in the following table for the periods presented (in thousands):
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 | | 2021 |
| Cash paid for amounts included in lease liabilities: | | | | | |
| Financing cash flows used for finance leases | $ | 332 | | | $ | — | | | $ | — | |
| Operating cash flows used for finance leases | 1,696 | | | — | | | — | |
| Operating cash flows used for operating leases | 10,306 | | | 7,049 | | | 6,335 | |
| | | | | |
| Non-cash transactions: | | | | | |
| Right-of-use assets obtained in exchange for new finance lease liabilities | $ | 32,862 | | | $ | — | | | $ | — | |
| Right-of-use assets obtained in exchange for new operating lease liabilities | 3,931 | | | 17,513 | | | — | |
As of December 31, 2023, the Company expects that its future minimum lease payments will become due and payable as follows (in thousands):
| | | | | | | | | | | | | | | | | |
| Finance Leases | | Operating Leases | | Total |
| 2024 | $ | 3,327 | | | $ | 10,224 | | | $ | 13,551 | |
| 2025 | 3,427 | | | 10,392 | | | 13,819 | |
| 2026 | 3,530 | | | 10,039 | | | 13,569 | |
| 2027 | 3,636 | | | 8,561 | | | 12,197 | |
| 2028 | 3,745 | | | 8,666 | | | 12,411 | |
| Thereafter | 40,357 | | | 25,432 | | | 65,789 | |
| Total minimum lease payments | 58,022 | | | 73,314 | | | 131,336 | |
| Less: interest | (25,492) | | | (19,024) | | | (44,516) | |
| Total lease liabilities | $ | 32,530 | | | $ | 54,290 | | | $ | 86,820 | |
9.Commitments and Contingencies
Unconditional Purchase Obligations
In the ordinary course of business, we enter into certain unconditional purchase obligations with our suppliers. These are agreements to purchase products and services that are enforceable, legally binding, and specify terms that include provisions with respect to quantities, pricing and timing of purchases.
Amounts purchased under these obligations totaled $3.0 million for the year ended December 31, 2023. Such amounts were not material for the years ended December 31, 2022 and 2021.
As of December 31, 2023, future minimum commitments under these obligations totaled $3.3 million which relates to the year ending December 31, 2024.
Legal Proceedings
The Company is involved in various legal proceedings arising in the normal course of business. The Company accrues for a loss contingency when it determines that it is probable, after consultation with counsel, that a liability has been incurred and the amount of such loss can be reasonably estimated. The Company believes that the results of any such contingencies, either individually or in the aggregate, will not have a material adverse effect on the Company’s consolidated financial position, results of operations or cash flows.
Indemnification Agreements
In the ordinary course of business, we may provide indemnification of varying scope and terms to vendors, lessors, customers and other parties with respect to certain matters including, but not limited to, losses arising out of breach of such agreements or
from intellectual property infringement claims made by third parties, and losses arising from breach of representations, warranties and covenants to counterparties set forth in agreements with such parties. We have also agreed to our directors and officers to the maximum extent permitted under applicable state laws pursuant to standard director and officer indemnification agreements and our corporate charter and bylaws. The maximum potential amount of future payments that we could be required to make under these indemnification agreements is, in many cases, unlimited. We have not incurred any material costs as a result of such indemnifications and are not currently aware of any indemnification claims.
10.Long-Term Debt
Credit Agreement
In October 2020, Maravai Intermediate Holdings, LLC (“Intermediate”), a wholly-owned subsidiary of Topco LLC, along with certain of its subsidiaries (together with Intermediate, the “Borrowers”), entered into a credit agreement (as amended, the “Credit Agreement”), which provides for a term loan facility and a revolving credit facility. In January 2022, the Company entered into an amendment (the “Amendment”) to refinance the term loan and to replace London Interbank Offered Rate (“LIBOR”) with a Term Secured Overnight Financing Rate (“SOFR”) based rate.
As amended, the Credit Agreement provides for a $600.0 million term loan facility, maturing October 2027 (the “Tranche B Term Loan”), and a $180.0 million revolving credit facility (the “Revolving Credit Facility”). The interest rate margins applicable to the Tranche B Term Loan and Revolving Credit Facility is 3.00%, with respect to each Term SOFR-based loan, and 2.00%, with respect to each Base Rate-based loan. Further, the interest rate floor for Base Rate term loans, Term SOFR-based term loans, and Term SOFR-based revolving loans are 1.50%, 0.50% and 0.00%, respectively.
As of December 31, 2023, the interest rate on the Tranche B Term Loan was 8.40% per annum.
The Credit Agreement also provides for a $20.0 million limit for letters of credit. As of December 31, 2023, the Company had a $0.5 million outstanding letter of credit as security for a lease agreement, which reduced the availability of credit under the Revolving Credit Facility by $0.5 million.
Borrowings under the Credit Agreement are unconditionally guaranteed by Topco LLC, together with the existing and future material domestic subsidiaries of Topco LLC (subject to certain exceptions), as specified in the respective guaranty agreements. Borrowings under the Credit Agreement are also secured by a first-priority lien and security interest in substantially all of the assets (subject to certain exceptions) of existing and future material domestic subsidiaries of Topco LLC that are loan parties.
The accounting related to entering into the Amendment was evaluated on a creditor-by-creditor basis to determine whether each transaction should be accounted for as a modification or extinguishment. Certain creditors under the Tranche B Term Loan did not participate in this refinancing transaction, were repaid their principal and interest of $8.5 million and ceased being creditors of the Company and the repayment of their related outstanding debt balances has been accounted for as an extinguishment of debt. Proceeds of borrowings from new lenders of $8.5 million were accounted for as a new debt financing. The Company recorded a loss on extinguishment of debt of $0.2 million in the accompanying consolidated statements of operations during the year ended December 31, 2022. For the remainder of the creditors, this transaction was accounted for as a modification because the change in present value of cash flows between the two term loans before and after the transaction was less than 10% on a creditor-by-creditor basis. As part of the refinancing, the Company incurred $0.9 million of various costs, of which an insignificant amount was related to an original issuance discount, and were all capitalized in the accompanying balance sheet within long-term debt and are subject to amortization over the term of the refinanced debt as an adjustment to interest expense using the effective interest method.
We also incurred $0.3 million of financing-related fees related to the Revolving Credit Facility in connection with the debt refinancing activities in January 2022. As of December 31, 2023, unamortized debt issuance costs totaled $1.4 million and are recorded as assets within other assets on the accompanying consolidated balance sheet as there is no balance outstanding related to the Revolving Credit Facility.
Commencing with the fiscal year ended December 31, 2021, and each fiscal year thereafter, the Credit Agreement requires that we make mandatory prepayments on the Tranche B Term Loan principal upon certain excess cash flow, subject to certain step-downs based on the Company’s first lien net leverage ratio. The excess cash flow shall be reduced to 25% or 0% of the calculated excess cash flow if the Company’s first lien net leverage ratio was equal to or less than 4.75:1.00 or 4.25:1.00, respectively, however, no prepayment shall be required to the extent excess cash flow calculated for the respective period is equal to or less than $10.0 million. As of December 31, 2023, the Company’s first lien net leverage ratio was less than 4.25:1.00. Thus, a mandatory prepayment on the Tranche B Term Loan out of our excess cash flow was not required.
The Tranche B Term Loan is repayable in quarterly payments of $1.4 million which began in March 2022, with all remaining outstanding principal due in October 2027. The Tranche B Term Loan includes prepayment provisions that allow the Company,
at our option, to repay all or a portion of the principal amount at any time. The Revolving Credit Facility allows the Company to repay and borrow from time to time until October 2025, at which time all amounts borrowed must be repaid. Subject to certain exceptions and limitations, we are required to repay borrowings under the Tranche B Term Loan and Revolving Credit Facility with the proceeds of certain occurrences, such as the incurrence of debt, certain equity contributions and certain asset sales or dispositions.
Accrued interest under the Credit Agreement is payable by us (a) quarterly in arrears with respect to Base Rate loans, (b) at the end of each interest rate period (or at each three-month interval in the case of loans with interest periods greater than three months) with respect to Term SOFR Rate loans, (c) on the date of any repayment or prepayment and (d) at maturity (whether by acceleration or otherwise). An annual commitment fee is applied to the daily unutilized amount under the Revolving Credit Facility at 0.375% per annum, with one stepdown to 0.25% per annum based on Intermediate’s first lien net leverage ratio calculation.
The Credit Agreement contains certain covenants, including, among other things, covenants limiting our ability to incur or prepay certain indebtedness, pay dividends or distributions, dispose of assets, engage in mergers and consolidations, make acquisitions or other investments and make changes to the nature of the business. Additionally, the Credit Agreement also requires us to maintain a certain net leverage ratio if the outstanding debt balance on the Revolving Credit Facility exceeds 35.0% of the aggregate amount of available credit of $180.0 million. The Company was in compliance with these covenants as of December 31, 2023.
Interest Rate Cap
In the first quarter of 2021, the Company entered into an interest rate cap agreement to manage a portion of its variable interest rate risk on its outstanding long-term debt. The contract, which was effective March 31, 2021, entitles the Company to receive from the counterparty at each calendar quarter end the amount, if any, by which a specified defined floating market rate exceeds the cap strike interest rate, applied to the contract’s notional amount of $415.0 million The floating rate of interest is reset at the end of each three month period. The contract was set to expire on March 31, 2023.
In May 2022, the Company amended the interest rate cap agreement, effective June 30, 2022, to increase the contract’s notional amount to $500.0 million and to extend the maturity date to January 19, 2025. Additionally, the floating rate option changed from a LIBOR-based rate to a SOFR-based rate. Other provisions remained unchanged as a result of the amendment. Premiums paid to amend the interest rate cap agreement were immaterial.
The interest rate cap agreement has not been designated as a hedging relationship and has been recognized on the consolidated balance sheet at fair value of $8.6 million within other assets with changes in fair value recognized within interest expense in the consolidated statements of operations. Proceeds from the interest rate cap agreement are reflected in cash flows used in financing activities in the consolidated statements of cash flows.
The Company’s long-term debt consisted of the following as of the periods presented (in thousands):
| | | | | | | | | | | |
| December 31, 2023 | | December 31, 2022 |
| Tranche B Term Loan | $ | 533,120 | | | $ | 538,560 | |
| Unamortized debt issuance costs | (8,973) | | | (11,123) | |
| Total long-term debt | 524,147 | | | 527,437 | |
| Less: current portion | (5,440) | | | (5,440) | |
| Total long-term debt, less current portion | $ | 518,707 | | | $ | 521,997 | |
There were no balances outstanding on the Company’s Revolving Credit Facility as of December 31, 2023 and 2022.
As of December 31, 2023, the aggregate future principal maturities of the Company’s debt obligations based on contractual due dates were as follows (in thousands):
| | | | | |
| 2024 | $ | 5,440 | |
| 2025 | 5,440 | |
| 2026 | 5,440 | |
| 2027 | 516,800 | |
| |
| |
| Total long-term debt | $ | 533,120 | |
11.Stockholders’ Equity
Amendment and Restatement of Certificate of Incorporation
In November 2020, in connection with the Organizational Transactions, the Company’s certificate of incorporation was amended and restated to, among other things, provide for the (i) authorization of 500,000,000 shares of Class A common stock with a par value of $0.01 per share; (ii) authorization of 300,000,000 shares of Class B common stock with a par value of $0.01 per share; (iii) authorization of 50,000,000 shares of preferred stock with a par value of $0.01 per share.
Holders of Class A and Class B common stock are entitled to one vote per share. Except as otherwise required in the Certificate of Incorporation or by applicable law, the holders of Class A common stock and Class B common stock shall vote together as a single class on all matters on which stockholders are generally entitled to vote. Holders of the Class A common stock are entitled to receive dividends, and upon the Company’s dissolution or liquidation, after payment in full of all amounts required to be paid to creditors and to the holders of preferred stock having liquidation preferences, if any, the holders of shares of Class A common stock will be entitled to receive the Company’s pro rata remaining assets available for distribution. Holders of Maravai’s Class B common stock are not entitled to receive dividends and will not be entitled to receive any distributions upon dissolution or liquidation of Maravai. Holders of Class A and Class B common stock do not have preemptive or subscription rights. As of December 31, 2023, no preferred stock was outstanding.
We are required to, at all times, maintain (i) a one-to-one ratio between the number of shares of Class A common stock outstanding and the number of LLC Units owned by us and (ii) a one-to-one ratio between the number of shares of Class B common stock owned by the MLSH 1 and the number of LLC Units owned by the MLSH 1. We may issue shares of Class B common stock only to the extent necessary to maintain these ratios. Shares of Class B common stock are transferable only together with an equal number of LLC Units if we, at the election of MLSH 1, exchange LLC Units for shares of Class A common stock. All Class B common stock that is transferred shall be automatically retired and cancelled and shall no longer be outstanding.
Exchanges and Secondary Offerings
April 2021 Exchange and Secondary Offering
In April 2021, MLSH 1 executed an exchange of 17,665,959 Topco LLC units (the “LLC Units”) (paired with the corresponding shares of Class B common stock) in return for 17,665,959 shares of the Company’s Class A common stock. The corresponding shares of Class B common stock were subsequently cancelled and retired. The Company immediately completed a secondary offering (“April 2021 Secondary Offering”) of 20,700,000 shares of its Class A common stock by MLSH 1 and Maravai Life Sciences Holdings 2, LLC (“MLSH 2”), which included 3,034,041 shares of Class A common stock previously held by MLSH 2, which included the full exercise of the underwriters’ option to purchase up to 2,700,000 additional shares of Class A common stock, at a price of $31.25 per share.
The selling stockholders were responsible for the underwriting discounts and commissions of the April 2021 Secondary Offering and received all of the net proceeds of $624.2 million from the sale of shares of Class A common stock. The Company was responsible for the offering costs associated with the April 2021 Secondary Offering of $1.0 million which were recorded within selling, general and administrative expenses in the consolidated statements of operations.
September 2021 Exchange and Secondary Offering
In September 2021, MLSH 1 executed an exchange of 17,068,559 LLC Units (paired with the corresponding shares of Class B common stock) in return for 17,068,559 shares of the Company’s Class A common stock. The corresponding shares of Class B common stock were subsequently cancelled and retired. Shortly after the exchange, the Company completed a secondary offering (“September 2021 Secondary Offering”) of 20,000,000 shares of its Class A common stock by MLSH 1 and MLSH 2, which included 2,931,441 shares of Class A common stock previously held by MLSH 2 at a price of $50.00 per share.
The selling stockholders were responsible for the underwriting discounts and commissions of the September 2021 Secondary Offering and received all of the net proceeds of $977.5 million from the sale of shares of Class A common stock. The Company was responsible for the offering costs associated with the September 2021 Secondary Offering of $0.9 million which were recorded within selling, general and administrative expenses in the consolidated statements of operations.
Cash Contribution, Exchange, and Forfeiture Agreement
In December 2021, the Company entered into a Cash Contribution, Exchange and Forfeiture Agreement (the “Contribution Agreement”) with Topco LLC and MLSH 1, a related party. Pursuant to the Contribution Agreement, the Company contributed $110.0 million of cash to Topco LLC in exchange for 2,732,919 newly-issued units LLC Units of Topco LLC at a price per unit of $40.25, which was equal to the 50-day volume-weighted average price of the Company’s Class A common stock as
calculated on December 31, 2021. Immediately following the contribution, the Company and MLSH 1 agreed to forfeit 2.036% of their respective LLC Units of Topco LLC and an equal number of shares of the Company’s Class B common stock, par value $0.01 per share, for no consideration. The purpose of the Contribution Agreement was to reduce the excess cash that had accumulated at the Company as a result of quarterly tax distributions it has received from Topco LLC since its IPO.
Structuring Transactions
In connection with the Company’s acquisition of Alphazyme (see Note 2), the Company undertook a series of structuring transactions (the “Structuring Transactions”), including:
•On January 18, 2023, the Company acquired all of the outstanding membership interests in Alphazyme (see Note 2).
•On January 19, 2023, the Company entered into a contribution agreement (the “Contribution Agreement”) with Alphazyme Holdings, Inc. (“Alphazyme Holdings”), a wholly owned subsidiary of the Company, pursuant to which the Company contributed all such membership interests in Alphazyme (the “Alphazyme Membership Interest”) to Alphazyme Holdings.
•On January 22, 2023, Alphazyme Holdings entered into a contribution and exchange agreement (the “Contribution and Exchange Agreement”) with Topco LLC, pursuant to which it contributed all of the Alphazyme Membership Interests to TopCo LLC in exchange for 5,059,134 newly-issued LLC Units of Topco LLC at a price per unit of $13.87, which was equal to the 50-day volume-weighted average price of the Company’s Class A common stock as calculated on January 18, 2023 (the “Contribution and Exchange”).
•Immediately following the Contribution and Exchange, the Company entered into a forfeiture agreement (the “Forfeiture Agreement”) with Alphazyme Holdings, TopCo LLC and MLSH 1, a related party, pursuant to which each of the Company (together with Alphazyme Holdings) and MLSH 1 agreed to forfeit 5,059,134 and 4,871,970 LLC Units, respectively, representing 3.7% of the Company’s (together with Alphazyme Holdings) and MLSH 1’s respective LLC Units of Topco LLC, and an equal number of shares of the Company’s Class B common stock, par value $0.01 per share, were forfeited by MLSH 1, in each case for no consideration.
These were considered transactions between entities under common control. As a result, the consolidated financial statements for periods prior to the these transactions have been adjusted to combine the previously separate entities for presentation purposes.
12.Net (Loss) Income Per Class A Common Share Attributable to Maravai LifeSciences Holdings, Inc.
Basic net (loss) income per Class A common share has been calculated by dividing net (loss) income for the period, adjusted for net (loss) income attributable to non-controlling interests, by the weighted average number of Class A common shares outstanding during the period. Diluted net (loss) income per Class A common share gives effect to potentially dilutive securities by application of the treasury stock method or if-converted method, as applicable. Diluted net (loss) income per Class A common share attributable to the Company is computed by adjusting the net (loss) income and the weighted average number of Class A common shares outstanding to give effect to potentially diluted securities. In periods in which the Company reports a net loss attributable to Maravai LifeSciences Holdings, Inc., diluted net loss per Class A common share attributable to the Company is the same as basic net loss per Class A common share attributable to the Company, since dilutive equity instruments are not assumed to have been issued if their effect is anti-dilutive. The Company reported a net loss attributable to Maravai LifeSciences Holdings, Inc. for the year ended December 31, 2023.
The following table presents the computation of basic and diluted net (loss) income per common share attributable to the Company for the periods presented (in thousands, except per share amounts):
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 | | 2021 |
| Numerator: | | | | | |
Net (loss) income | $ | (138,375) | | | $ | 490,663 | | | $ | 469,250 | |
Less: loss (income) attributable to common non-controlling interests | 19,346 | | | (270,458) | | | (287,213) | |
Net (loss) income attributable to Maravai LifeSciences Holdings, Inc.—basic | (119,029) | | | 220,205 | | | 182,037 | |
Net (loss) income effect of dilutive securities: | | | | | |
| Effect of dilutive employee stock purchase plan, RSUs and options | $ | — | | | 87 | | | 132 | |
| Effect of the assumed conversion of Class B common stock | — | | | 205,984 | | | 220,187 | |
Net (loss) income attributable to Maravai LifeSciences Holdings, Inc.—diluted | $ | (119,029) | | | $ | 426,276 | | | $ | 402,356 | |
| | | | | |
| Denominator: | | | | | |
Weighted average Class A common shares outstanding—basic | 131,919 | | | 131,545 | | | 114,791 | |
| Weighted average effect of dilutive securities: | | | | | |
| Effect of dilutive employee stock purchase plan, RSUs and options | — | | | 109 | | | 153 | |
| Effect of the assumed conversion of Class B common stock | — | | | 123,669 | | | 142,859 | |
Weighted average Class A common shares outstanding—diluted | 131,919 | | | 255,323 | | | 257,803 | |
| | | | | |
Net (loss) income per Class A common share attributable to Maravai LifeSciences Holdings, Inc.: | | | | | |
| Basic | $ | (0.90) | | | $ | 1.67 | | | $ | 1.59 | |
| Diluted | $ | (0.90) | | | $ | 1.67 | | | $ | 1.56 | |
Shares of Class B common stock do not share in the earnings or losses of the Company, and are therefore not participating securities. As such, a separate presentation of basic and diluted net (loss) income per share for Class B common stock under the two-class method has not been presented.
The following table presents potentially dilutive securities excluded from the computation of diluted net (loss) income per share for the periods presented because their effect would have been anti-dilutive for the periods presented (in thousands):
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 | | 2021 |
| | | | | |
| Restricted stock units | 3,181 | | | 74 | | | — | |
| Stock options | 4,246 | | | 2,769 | | | 355 | |
| Shares estimated to be purchased under employee stock purchase plan | — | | | 13 | | | 12 | |
| Shares of Class B common stock | 119,094 | | | — | | | — | |
| Total | 126,521 | | | 2,856 | | | 367 | |
Shares underlying contingently issuable awards that have not met the necessary conditions as of the end of a reporting period are not included in the calculation of diluted net (loss) income per Class A common share attributable to the Company for that period. The Company had contingently issuable PSUs outstanding that did not meet the market and performance conditions as of December 31, 2023 and 2022 and, therefore, were excluded from the calculation of diluted net (loss) income per Class A common share attributable to the Company. The maximum number of potentially dilutive shares that could be issued upon vesting for such awards was insignificant as of December 31, 2023 and 2022. These amounts were also excluded from the potentially dilutive securities in the table above. The Company had no contingently issuable PSUs outstanding as of December 31, 2021.
13.Equity Incentive Plans
Stock-Based Compensation
In November 2020, the Company’s board of directors adopted the 2020 Omnibus Incentive Plan (the “2020 Plan”). The 2020 Plan provides for an automatic increase in the number of shares reserved for issuance thereunder on January 1 of each of the first 10 calendar years during the term of the 2020 Plan, by the lesser of (i) 4% of the total number of shares of Class A common stock outstanding on each December 31 immediately prior to the date of increase or (ii) such number of shares of Class A common stock determined by our board of directors or compensation committee. Shares of Class A common stock subject to an award that expires or is cancelled, forfeited, exchanged, settled in cash or otherwise terminated without delivery of shares and shares withheld to pay the exercise price of, or to satisfy the withholding obligations with respect to, an award will again be available for delivery pursuant to other awards under the 2020 Plan.
All awards granted under the 2020 Plan are intended to be treated as (i) stock options, including incentive stock options (“ISOs”), (ii) stock appreciation rights (“SARs”), (iii) restricted share awards (“RSAs”), (iv) restricted stock units (“RSUs”), (v) performance awards, (vi) dividend equivalents, or (vii) other stock or cash awards as may be determined by the plan’s administrator from time to time. The term of each option award shall be no more than 10 years from the date of grant. The exercise price of a stock option shall not be less than 100% (or, in the case of an ISO granted to a ten percent stockholder, 110%) of the fair market value of the shares on the date of grant. As of December 31, 2023, only stock options, RSUs and PSUs have been issued.
In November 2020, the Company adopted the 2020 Employee Stock Purchase Plan (the “ESPP”) to assist employees in acquiring a stock ownership interest in the Company and to encourage them to remain in the employment of the Company. The ESPP permits eligible employees to purchase shares of Class A common stock at a discount through payroll deductions during specified six-month purchase periods. The price of shares purchased under the ESPP is equal to the lower of the grant date price less a 15% discount or a 15% discount to the market closing price on the date of purchase.
Compensation expense recognized for the ESPP was insignificant for all periods presented.
The Company began issuing PSUs during 2022 to certain executive employees under the 2020 Plan. Certain PSUs vest only if the executive employee satisfies a service-based vesting condition and market condition. The executive employee must remain employed through the third anniversary of the grant date. The award is eligible to vest based on the achievement of certain price targets of the Company’s stock price over a defined performance period. Certain other PSUs are subject to a performance condition being satisfied. The award is eligible to vest upon achievement of certain revenue-based performance goals and are subject to continued service over a defined performance period.
Compensation expense recognized for these PSUs were insignificant for the years ended December 31, 2023 and 2022. There was no compensation expense related to PSUs during the year ended December 31, 2021.
Stock Options
The following table summarizes information related to stock options:
| | | | | | | | | | | | | | | | | | | | | | | |
| Number of Stock Options
(in thousands) | | Weighted Average Exercise Price per Stock Option | | Weighted Average Remaining Contractual Life
(in years) | | Aggregate Intrinsic Value
(in thousands) |
| Outstanding as of December 31, 2022 | 2,893 | | | $ | 26.45 | | | 8.9 | | $ | — | |
| Granted | 2,270 | | | 14.76 | | | | | |
| | | | | | | |
| Cancelled | (858) | | | 25.11 | | | | | |
| Outstanding as of December 31, 2023 | 4,305 | | | $ | 20.55 | | | 8.5 | | $ | 19 | |
| Exercisable as of December 31, 2023 | 1,456 | | | $ | 24.76 | | | 7.7 | | $ | — | |
The Company uses the Black-Scholes option pricing model to estimate the fair value of each option grant on the date of grant or any other measurement date. The assumptions and estimates are as follows:
•Expected term - The expected term represents the period that stock-based awards are expected to be outstanding and is determined using the simplified method. Our historical share option exercise information is limited due to a lack of sufficient data points and does not provide a reasonable basis upon which to estimate an expected term.
•Expected volatility - The expected volatility was derived from the historical stock volatilities of peer public companies within our industry that are considered to be comparable to our business over a period equivalent to the expected term of the stock-based awards, since our stock trading history is limited.
•Risk-free interest rate - The risk-free interest rate is based on the U.S. Treasury yield curve in effect at the date of grant for zero-coupon U.S. Treasury notes with maturities approximately equal to the stock-based awards’ expected term.
•Expected dividend yield - The expected dividend yield is zero as we have no plans to make dividend payments.
A summary of the assumptions used to estimate the fair value of stock option grants for the years presented is as follows:
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 | | 2021 |
| Expected volatility | 48.0 | % | | 51.3 | % | | 57.2 | % |
| Risk-free interest rate | 3.6 | % | | 2.8 | % | | 1.0 | % |
| Expected term (in years) | 6.5 | | 6.1 | | 6.1 |
| Expected dividend yield | — | % | | — | % | | — | % |
Stock-based compensation expense related to stock options was $11.5 million, $8.1 million and $4.6 million for the years ended December 31, 2023, 2022 and 2021, respectively. The total fair value of stock options vested was $11.9 million, $7.7 million and $4.3 million for the years ended December 31, 2023, 2022 and 2021, respectively.
As of December 31, 2023, the total unrecognized stock-based compensation related to stock options was $26.3 million, which is expected be recognized over a weighted-average period of approximately 2.7 years.
Restricted Stock Units
The Company has granted restricted stock unit awards to employees and non-employee directors and contractors. The following table summarizes information related to RSUs:
| | | | | | | | | | | |
| Restricted Stock Units
(in thousands) | | Weighted Average Fair Value per RSU at Grant Date |
| Balance as of December 31, 2022 | 1,331 | | | $ | 21.04 | |
| Granted | 3,507 | | | 13.66 | |
| Vested | (278) | | | 23.66 | |
| Forfeited | (616) | | | 20.30 | |
| Balance as of December 31, 2023 | 3,944 | | | $ | 15.35 | |
Stock-based compensation expense related to RSUs was $20.2 million, $8.2 million and $0.8 million for the years ended December 31, 2023, 2022 and 2021, respectively. The total fair value of RSUs vested was $5.0 million, $1.0 million and $0.9 million for the years ended December 31, 2023, 2022 and 2021, respectively.
As of December 31, 2023, the total unrecognized equity-based compensation related to RSUs was $43.7 million, which is expected be recognized over a weighted-average period of approximately 2.0 years.
Unit-Based Compensation
Prior to the IPO, the Company’s parent, MLSH 1, granted unit-based awards (“MLSH 1 Incentive Units”) to certain executives of the Company in the form of non-vested units.
MLSH 1 Incentive Units
Prior to the Organizational Transactions, Topco LLC entered into agreements with certain executives and board members whereby those employees and board members were granted incentive units in MLSH 1, a related party. All MLSH 1 Incentive Unit awards were subject to a market condition which is subject to the achievement of a certain investment return threshold that increased on a compounding basis annually and a service condition subject to their continued employment. Certain MLSH 1 Incentive Unit awards contained a performance condition tied to the achievement of certain cash distribution multiples. All vested MLSH 1 Incentive Unit awards are subject to repurchase for fair value at MLSH 1’s option upon a voluntary or involuntary separation event that is not deemed to be for cause.
The MLSH 1 Incentive Unit awards that include market and service conditions provide for cliff-vesting generally over four or five years. The MLSH 1 Incentive Unit awards that include market and performance conditions provide for full vesting upon meeting the performance condition. The fair value of MLSH 1 Incentive Unit awards was measured at the grant date and recognized as expense over the requisite service period for the awards.
In connection with the divestiture of its Protein Detection business, the Company recognized incremental unit-based compensation expense of $2.4 million related to an amended agreement with an executive of Vector (see Note 2). This unit-based compensation expense was recorded within selling, general and administrative expenses in the consolidated statements of operations for the year ended December 31, 2021.
Unit-based compensation expense related to MLSH 1 Incentive Unit awards was approximately $0.2 million, $0.7 million and $3.9 million for the years ended December 31, 2023, 2022 and 2021, respectively.
MLSH 1 Incentive Unit award activity during year ended December 31, 2023 is as follows:
| | | | | | | | | | | |
| Number of Unvested MLSH 1 Incentive Units
(in thousands) | | Weighted Average Grant Date Fair Value Per Unit |
| Balance as of December 31, 2022 | 77 | | | $ | 24.34 | |
| Forfeited | (12) | | | 20.08 | |
| Vested | (33) | | | 22.24 | |
| Balance as of December 31, 2023 | 32 | | | $ | 28.15 | |
As of December 31, 2023, total unrecognized compensation cost related to unvested MLSH 1 Incentive Units subject to service condition is $0.1 million which is expected to be recognized over a weighted average period of 1.0 year.
Equity-Based Compensation
The following table summarizes the total equity-based compensation expense included in the Company’s consolidated statements of operations for the periods presented (in thousands):
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 | | 2021 |
| Cost of sales | $ | 7,324 | | | $ | 4,192 | | | $ | 1,915 | |
| Selling, general and administrative | 24,650 | | | 13,349 | | | 8,263 | |
| Research and development | 2,715 | | | 1,129 | | | 280 | |
Restructuring | (101) | | | — | | | — | |
| Total equity-based compensation | $ | 34,588 | | | $ | 18,670 | | | $ | 10,458 | |
14.Income Taxes
As of December 31, 2023 and 2022, we are subject to U.S. federal and state income taxes with respect to our allocable share of any taxable income or loss of Topco LLC, as well as any stand-alone income or loss we generate. Topco LLC is organized as a limited liability company and treated as a partnership for federal tax purposes and generally does not pay income taxes on its taxable income in most jurisdictions. Instead, Topco LLC’s taxable income or loss is passed through to its members, including us.
Components of income from continuing operations before income taxes for the periods presented were as follows (in thousands):
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 | | 2021 |
| U.S. | $ | 617,681 | | | $ | 551,472 | | | $ | 530,853 | |
| International | 55 | | | — | | | (88) | |
| Total income from continuing operations | $ | 617,736 | | | $ | 551,472 | | | $ | 530,765 | |
Income tax expense consisted of the following for the periods presented (in thousands):
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 | | 2021 |
Current tax expense | | | | | |
| Federal | $ | 405 | | | $ | 16,312 | | | $ | 9,291 | |
| State and local | 756 | | | 2,173 | | | 1,623 | |
| International | 8 | | | 6 | | | 3,697 | |
Total current tax expense | 1,169 | | | 18,491 | | | 14,611 | |
| | | | | |
Deferred tax expense | | | | | |
| Federal | $ | 663,968 | | | $ | 39,924 | | | $ | 36,564 | |
| State and local | 90,974 | | | 2,394 | | | 10,340 | |
| | | | | |
Total deferred tax expense | 754,942 | | | 42,318 | | | 46,904 | |
| | | | | |
| Total provision for income taxes | $ | 756,111 | | | $ | 60,809 | | | $ | 61,515 | |
A reconciliation between the Company’s effective tax rate and the applicable U.S. federal statutory income tax rate as of the periods presented is summarized as follows:
| | | | | | | | | | | | | | | | | |
| December 31, 2023 | | December 31, 2022 | | December 31, 2021 |
| Federal statutory rate | 21.0 | % | | 21.0 | % | | 21.0 | % |
| State and local taxes, net of federal benefits | 14.9 | | | 0.6 | | | 2.2 | |
| Deferred tax revaluation | 1.2 | | | 0.3 | | | — | |
| Income of non-controlling interest | 0.8 | | | (10.3) | | | (11.4) | |
| | | | | |
| Taxable (loss) gain on subsidiary liquidation | — | | | — | | | (0.7) | |
| Equity-based compensation | — | | | — | | | 0.1 | |
| Research and development credits | — | | | (0.1) | | | (0.4) | |
| | | | | |
| Valuation allowance | 87.6 | | | 0.1 | | | 0.1 | |
Nondeductible TRA movement | (3.0) | | | — | | | — | |
| Other | — | | | (0.6) | | | 0.7 | |
| Effective tax rate | 122.5 | % | | 11.0 | % | | 11.6 | % |
Deferred income taxes reflect the net tax effects of temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used for income tax purposes and operating loss and tax credit carryforwards. Significant items comprising the net deferred tax assets were as follows as of the periods presented below (in thousands):
| | | | | | | | | | | |
| December 31, 2023 | | December 31, 2022 |
| Deferred tax assets | | | |
| Investment in Topco LLC | $ | 595,796 | | $ | 636,498 |
Net operating loss | 40,980 | | — |
| Deductions to be received for the Tax Receivable Agreement payments | 1,408 | | 148,681 |
| Capital loss carryforward | 3,256 | | 3,265 |
| Other | 712 | | 1,131 |
| Total deferred tax assets | 642,152 | | 789,575 |
| Valuation allowance | (642,152) | | (23,776) |
| Total deferred tax assets, net of valuation allowance | $ | — | | $ | 765,799 |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
| | | |
As a result of the Organizational Transactions, IPO, and subsequent exchanges and financing, we acquired LLC Units and recognized a deferred tax asset for the difference between the financial reporting and tax basis of our investment in Topco LLC
which included net deferred tax assets of $0.0 million primarily associated with: (i) $595.8 million related to temporary differences in the book basis as compared to the tax basis of our Company’s investment in Topco LLC, (ii) $1.4 million related to tax benefits from future deductions attributable to payments under the TRA, (iii) $3.3 million related to the capital loss carryforwards generated during the sale of Vector, (iv) $41.0 million related to net operating loss carryforwards, and (v) $642.2 million valuation allowance on these and other items.
The valuation allowance increased by $618.4 million and $0.7 million during the years ended December 31, 2023 and 2022, respectively.
The realizability of the Company’s deferred tax asset related to its investment in Topco LLC depends on the Company receiving allocations of tax deductions for its tax basis in the investment and on the Company generating sufficient taxable income to fully offset such deductions. Management assesses the available positive and negative evidence to estimate whether sufficient future taxable income will be generated to permit use of existing deferred tax assets. A significant piece of objective evidence evaluated during the year ended December 31, 2023 was our current year and projected future pre-tax losses. Due to our recent history of current year and projected near-term pre-tax losses, we determined that the negative evidence outweighs the positive evidence and so it is more likely than not that our deferred tax assets will not be utilized, and therefore the Company recorded a full valuation allowance on its U.S. federal and state deferred tax assets. The objective negative evidence is difficult to overcome and limits the ability to consider other subjective evidence, such as projections of future growth. It is possible in the foreseeable future that there may be sufficient positive evidence, and that the objective negative evidence related to pre-tax losses will no longer be present, in which event the Company could release a portion or all of the valuation allowance. Release of any amount of valuation allowance would result in a benefit to income tax expense for the period the release is recorded, which could have a material impact on net earnings.
Alphazyme was treated as a regarded corporation for U.S. federal and state income tax purposes at the time of acquisition. The Company recorded the initial contribution of Alphazyme to Topco LLC through its deferred tax asset related to the investment in Topco LLC, which was offset by a valuation allowance against the deferred tax asset. The Company also recorded a deferred tax liability for the difference between book basis and tax basis in the net assets of Alphazyme through purchase accounting. However, for the year ended December 31, 2023, Alphazyme became a disregarded entity for U.S. federal and state income tax purposes, which became effective immediately following the acquisition and prior to the contribution to Topco LLC. The change in Alphazyme’s tax status resulted in an income tax benefit of $8.8 million from the reversal of the Company’s deferred tax liability related to its ownership of Alphazyme, as well as an income tax expense of $17.1 million from the impact of the reversal to the Company’s deferred tax asset for its investment in Topco LLC.
Net operating loss (“NOL”) and tax credit carryforwards as of December 31, 2023 were as follows (in millions):
| | | | | | | | | | | |
| Amount | | Expiration Years |
Net operating losses, federal | $ | 36.3 | | | Does not expire |
Net operating losses, state | 4.7 | | | Varies by state |
| Capital loss carryforward | 3.3 | | | 2026 |
| Tax credits, federal | 0.3 | | | 2043 |
| Tax credits, state | 0.3 | | | CA - Do not expire |
As of December 31, 2023 and 2022, the Company had $5.2 million and $6.3 million of unrecognized tax benefits, all of which would affect the effective tax rate if recognized. The Company expects our unrecognized tax benefits may decrease by $2.6 million in the next twelve months due to statute expiration. The Company recognizes interest related to uncertain tax benefits as a component of income tax expense, including $0.3 million recognized during the year ended December 31, 2023.
The aggregate changes in the balance of the Company’s unrecognized tax benefits were as follows for the periods presented (in thousands):
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 | | 2021 |
| Balance, beginning of year | $ | 6,257 | | $ | 241 | | $ | 220 |
| Gross increases based on tax positions related to current year | 99 | | 130 | | 232 |
| Gross increases based on tax positions related to prior years | — | | 6,775 | | — |
| Gross decreases based on tax positions related to prior years | (1,158) | | (889) | | (211) |
| Balance, end of year | $ | 5,198 | | $ | 6,257 | | $ | 241 |
The Company files income tax returns in the U.S. federal jurisdiction and various states and is not under audit by taxing authorities in any of these jurisdictions. With exceptions for certain states, the Company is no longer subject to U.S. federal, state, and local, or non-U.S. income tax examinations for years before 2020.
Payable to Related Parties Pursuant to the Tax Receivable Agreement
We are a party to a TRA with MLSH 1 and MLSH 2. The TRA provides for the payment by us to MLSH 1 and MLSH 2, collectively, of 85% of the amount of certain tax benefits, if any, that we actually realize, or in some circumstances are deemed to realize, as a result of the Organizational Transactions, IPO and any subsequent purchases or exchanges of LLC Units of Topco LLC. The Company expects to benefit from the remaining 15% of any cash tax savings that it realizes.
We recognize the amount of TRA payments expected to be paid within the next 12 months and classify this amount as current. This determination is based on our estimate of taxable income for the year ended December 31, 2023. As of December 31, 2023, the current liability under the TRA was $7.1 million.
As of December 31, 2023, the Company has derecognized the remaining $665.3 million non-current liability under the TRA after concluding it was not probable that the Company will be able to realize the remaining tax benefits based on estimates of future taxable income. The estimation of liability under the TRA is by its nature imprecise and subject to significant assumptions regarding the amount, character, and timing of the taxable income in the future. If the Company concludes in a future period that the tax benefits are more likely than not to be realized and releases its valuation allowance, the corresponding TRA liability amounts may be considered probable at that time and recorded on the consolidated balance sheet and within earnings.
We made payments of $42.6 million to MLSH 1 and MLSH 2 pursuant to the TRA during the year ended December 31, 2023, of which $0.4 million is related to interest. We made payments of $35.3 million to MLSH 1 and MLSH 2 pursuant to the TRA during the year ended December 31, 2022, of which $1.1 million was related to interest. As of December 31, 2023 and 2022, our liabilities under the TRA were $7.1 million and $718.2 million, respectively.
Tax Distributions to Topco LLC’s Owners
Topco LLC is subject to an operating agreement put in place at the date of the Organizational Transactions (“LLC Operating Agreement”). The LLC Operating Agreement has numerous provisions related to allocations of income and loss, as well as timing and amounts of distributions to its owners. This agreement also includes a provision requiring cash distributions enabling its owners to pay their taxes on income passing through from Topco LLC. These tax distributions are computed based on an assumed income tax rate equal to the sum of (i) the maximum combined marginal federal and state income tax rate applicable to an individual and (ii) the net investment income tax. The assumed income tax rate ranges from 46.7% to 54.1% in certain cases where the qualified business income deduction is unavailable.
In addition, under the tax rules, Topco LLC is required to allocate taxable income disproportionately to its unit holders. Because tax distributions are determined based on the holder of LLC Units who is allocated the largest amount of taxable income on a per unit basis, but are made pro rata based on ownership, Topco LLC is required to make tax distributions that, in the aggregate, will likely exceed the amount of taxes Topco LLC would have otherwise paid if it were taxed on its taxable income at the assumed income tax rate. Topco LLC is subject to entity level taxation in certain states and certain of its subsidiaries are subject to entity level U.S. and foreign income taxes. As a result, the accompanying consolidated statements of operations include income tax expense related to those states and to U.S. and foreign jurisdictions where Topco LLC or any of our subsidiaries are subject to income tax.
During the year ended December 31, 2023, Topco LLC paid tax distributions of $20.3 million to its owners, including $10.7 million to us. During the year ended December 31, 2022, Topco LLC paid tax distributions of $310.0 million to its owners, including $159.8 million to us. During the year ended December 31, 2021, Topco LLC paid tax distributions of $283.2 million to its owners, including $129.7 million to us.
As of December 31, 2023, no amounts for tax distributions have been accrued as such payments were made during the period.
15.Employee Benefit Plans
The Company sponsors a 401(k) plan (the “Maravai LifeSciences 401(k) Plan”) pursuant to which eligible employees can elect to contribute to the 401(k) Plan, subject to certain limitations, on a pretax basis. The Company provides for a cash match of up to 50% of employee contributions up to the first 6% of salary.
Total contributions by the Company to the Maravai LifeSciences 401(k) Plan was approximately $2.1 million, $1.6 million and $1.3 million for the years ended December 31, 2023, 2022 and 2021, respectively.
16.Related Party Transactions
MLSH 1’s majority owner is GTCR, LLC (“GTCR”). The Company’s Executive Chairman of the Board, Chief Financial Officer (“CFO”) and General Counsel are executives of MLSH 1 and MLSH 2.
Registration Rights Agreement with MLSH 1 and MLSH 2
In connection with the IPO, Company entered into a registration rights agreement with MLSH 1 and MLSH 2. MLSH 1 and MLSH 2 are entitled to request that the Company register their shares of capital stock on a long-form or short-form registration statement on one or more occasions in the future, which registrations may be “shelf registrations.” MLSH 1 and MLSH 2 are also entitled to participate in certain of our registered offerings, subject to the restrictions in the registration rights agreement. During 2021, the Company registered shares of Class A shares held by MLSH 1 which were subsequently sold in an offering as selling shareholders as well as facilitated secondary offering transactions related to the exchanges (see Note 11).
Exchange Agreement with MLSH 1
In connection with the IPO, the Company entered into an exchange agreement with MLSH 1, whereby MLSH 1 may surrender their LLC Units to Topco LLC or, at our election, exchange its LLC Units for shares of our Class A common stock on a one-for-one basis, or, at our election, for cash from a substantially concurrent public offering or private sale. MLSH 1 is also required to deliver to us an equivalent number of shares of Class B common stock to effectuate an exchange. MLSH 1 executed two exchanges under this agreement during 2021 (see Note 11).
Payable to Related Parties Pursuant to the Tax Receivable Agreement
Concurrent with the completion of the IPO, the Company entered into a TRA with MLSH 1 and MLSH 2. During the years ended December 31, 2023, 2022 and 2021, the Company made TRA payments to both MLSH 1 and MLSH 2 (see Note 14).
Cash Contribution, Exchange and Forfeiture Agreement with MLSH 1
In December 2021, the Company entered into a Cash Contribution, Exchange and Forfeiture Agreement with MLSH 1 (see Note 11).
Topco LLC Operating Agreement
MLSH 1 is party to the Topco LLC operating agreement put in place at the date of the Organizational Transactions. This agreement includes a provision requiring cash distributions enabling its owners to pay their taxes on income passing through from Topco LLC. During the years ended December 31, 2023, 2022 and 2021, the Company made distributions of $9.6 million, $150.2 million and $153.5 million for tax liabilities to MLSH 1 under this agreement, respectively.
Contract Development and Manufacturing Agreement with Curia Global
GTCR has significant influence over Curia Global (“Curia”). During the years ended December 31, 2023 and 2022, the Company paid insignificant amounts to Curia for contract manufacturing and development services. During the year ended December 31, 2021, the Company paid $7.4 million to Curia. Such amounts were included in research and development expenses on the consolidated statements of operations.
Maravai LifeSciences Foundation
In December 2021, the Company established a new charitable foundation to promote causes tied to Maravai’s mission. During the year ended December 31, 2021, the Company contributed $2.0 million to the Foundation. The Company does not control the Foundation’s activities, and accordingly, does not consolidate the Foundation.
17.Segments
Operating segments are defined as components of an enterprise about which separate financial information is available that is evaluated regularly by the chief operating decision maker, or decision-making group, in deciding how to allocate resources and in assessing performance. When determining the reportable segments, the Company aggregated operating segments based on their similar economic and operating characteristics. Segment results are presented in the same manner as we present our operations internally to make operating decisions and assess performance. The accounting policies for the segments are the
same as those described in Significant Accounting Policies (see Note 1). The Company’s financial performance is reported in three segments. A description of each segment follows:
•Nucleic Acid Production: focuses on the manufacturing and sale of highly modified nucleic acids products to support the needs of customers’ research, therapeutic and vaccine programs. This segment also provides research products for labeling and detecting proteins in cells and tissue samples.
•Biologics Safety Testing: focuses on the manufacturing and sale of host cell protein, bioprocess impurity detection, viral clearance prediction kits and associated products. This segment also provides services for custom antibody development, assay development, antibody affinity extraction and mass spectrometry that are utilized by our customers in their biologic drug manufacturing spectrum.
•Protein Detection: focused on manufacturing and selling labeling and visual detection reagents to scientific research customers for their tissue-based protein detection and characterization needs. The Company completed the divestiture of its Protein Detection business in September 2021 (see Note 2).
The Company has determined that adjusted earnings before interest, tax, depreciation and amortization (“Adjusted EBITDA”) is the profit or loss measure that the CODM uses to make resource allocation decisions and evaluate segment performance. Adjusted EBITDA assists management in comparing the segment performance on a consistent basis for purposes of business decision-making by removing the impact of certain items that management believes do not directly reflect the core operations and, therefore, are not included in measuring segment performance. The Company defines Adjusted EBITDA as net (loss) income before interest, taxes, depreciation and amortization, certain non-cash items and other adjustments that we do not consider in our evaluation of ongoing operating performance from period to period. Corporate costs, net of eliminations, are managed on a standalone basis and not allocated to segments.
The following schedule includes revenue and adjusted EBITDA for each of the Company’s reportable operating segments (in thousands):
| | | | | | | | | | | | | | | | | |
| Year Ended December 31, |
| 2023 | | 2022 | | 2021 |
| | | | | |
| Revenue: | | | | | |
| Nucleic Acid Production | $ | 224,769 | | | $ | 813,076 | | | $ | 712,520 | |
| Biologics Safety Testing | 64,179 | | | 69,932 | | | 68,417 | |
| Protein Detection | — | | | — | | | 18,959 | |
| Total reportable segments’ revenue | 288,948 | | | 883,008 | | | 799,896 | |
| Intersegment eliminations | (3) | | | (7) | | | (656) | |
| Total | $ | 288,945 | | | $ | 883,001 | | | $ | 799,240 | |
| | | | | |
| Segment adjusted EBITDA: | | | | | |
| Nucleic Acid Production | $ | 82,658 | | | $ | 638,337 | | | $ | 565,254 | |
| Biologics Safety Testing | 46,908 | | | 54,841 | | | 54,440 | |
| Protein Detection | — | | | — | | | 6,391 | |
| Total reportable segments’ adjusted EBITDA | 129,566 | | | 693,178 | | | 626,085 | |
| Reconciliation of total reportable segments’ adjusted EBITDA to income before income taxes | | | | | |
| Amortization | (27,356) | | | (24,269) | | | (18,339) | |
| Depreciation | (12,898) | | | (7,566) | | | (6,413) | |
| Interest expense | (45,892) | | | (20,414) | | | (30,260) | |
| Interest income | 27,727 | | | 2,338 | | | — | |
| Corporate costs, net of eliminations | (64,257) | | | (55,378) | | | (43,265) | |
| Other adjustments: | | | | | |
| Acquisition contingent consideration | 3,286 | | | 7,800 | | | — | |
| Acquisition integration costs | (12,695) | | | (13,362) | | | (44) | |
| | | | | |
| Equity-based compensation | (34,588) | | | (18,670) | | | (10,458) | |
| Gain on sale of business | — | | | — | | | 11,249 | |
| | | | | |
| Merger and acquisition related expenses | (4,392) | | | (2,416) | | | (1,508) | |
| Financing costs | — | | | (1,078) | | | (2,383) | |
| Acquisition related tax adjustment | (1,293) | | | (349) | | | — | |
| Tax Receivable Agreement liability adjustment | 668,886 | | | (4,102) | | | 6,101 | |
| Chief Executive Officer transition costs | (28) | | | (2,426) | | | — | |
Restructuring costs (1) | (6,567) | | | — | | | — | |
| Other | (1,763) | | | (1,814) | | | — | |
| Income before income taxes | 617,736 | | | 551,472 | | | 530,765 | |
| Income tax expense | (756,111) | | | (60,809) | | | (61,515) | |
Net (loss) income | $ | (138,375) | | | $ | 490,663 | | | $ | 469,250 | |
___________________
(1)Equity-based compensation benefit of $0.1 million related to forfeited equity awards in connection with the restructuring is included on the equity-based compensation line item.
During the years ended December 31, 2023 and 2022, intersegment revenue was immaterial between the Nucleic Acid Production and Biologics Safety Testing segments. During the year ended December 31, 2021, intersegment revenue was $0.7 million between the Nucleic Acid Production and Protein Detection segments. The intersegment sales and the related gross margin on inventory recorded at the end of the period are eliminated for consolidation purposes. Internal selling prices for intersegment sales are consistent with the segment’s normal retail price offered to external parties. There was no commission expense recognized for intersegment sales for the years ended December 31, 2023, 2022 and 2021.
The Company does not allocate assets to its reportable segments as they are not included in the review performed by the CODM for purposes of assessing segment performance and allocating resources.
Item 9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure
None.
Item 9A. Controls and Procedures
Evaluation of Disclosure Controls and Procedures
Under the supervision and with the participation of our management, including our Chief Executive Officer and Chief Financial Officer, we evaluated the effectiveness of the design and operation of our disclosure controls and procedures pursuant to Rule 13a-15(e) and 15(d)-15(e) under the Securities Exchange Act of 1934, as amended (the “Exchange Act”) as of the end of the period covered by this report. Our disclosure controls and procedures are designed to ensure that information required to be disclosed in the reports that are filed or submitted under the Exchange Act is accumulated and communicated to management, including the Chief Executive Officer and Chief Financial Officer, to allow for timely decisions regarding required disclosures. In designing and evaluating the disclosure controls and procedures, management recognized that any controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving the desired control objectives, as ours are designed to do, and management necessarily applies its judgment in evaluating the cost-benefit relationship of possible controls and procedures. Based on such evaluation, our Chief Executive Officer and Chief Financial Officer have concluded that, as of the end of such period, the Company’s disclosure controls and procedures were effective at a reasonable assurance level.
Management’s Report on Internal Control over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting. Our internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with accounting principles generally accepted in the United States. Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Therefore, even those systems determined to be effective can provide only reasonable assurance of achieving their control objectives.
Our management assessed the effectiveness of our internal control over financial reporting as of December 31, 2023. In making this assessment, we used the criteria set forth by the Committee of Sponsoring Organizations of the Treadway Commission (COSO) in Internal Control – Integrated Framework (2013 Framework). Based on its assessment, management concluded that, as of December 31, 2023, the Company’s internal control over financial reporting was effective. Ernst & Young LLP, an independent registered public accounting firm, has issued an auditors’ report on our internal control over financial reporting as of December 31, 2023, which is included elsewhere in this Annual Report on Form 10-K.
Changes in Internal Control over Financial Reporting
There have been no changes in our internal control over financial reporting, as such term is defined in Rules 13a-15(f) and 15d-15(f) under the Exchange Act, during the three months ended December 31, 2023 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
Report of Independent Registered Public Accounting Firm
To the Stockholders and the Board of Directors of Maravai LifeSciences Holdings, Inc.
Opinion on Internal Control Over Financial Reporting
We have audited Maravai LifeSciences Holdings, Inc.’s internal control over financial reporting as of December 31, 2023, based on criteria established in Internal Control—Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework) (the COSO criteria). In our opinion, Maravai LifeSciences Holdings, Inc. (the Company) maintained, in all material respects, effective internal control over financial reporting as of December 31, 2023, based on the COSO criteria.
We also have audited, in accordance with the standards of the Public Company Accounting Oversight Board (United States) (PCAOB), the consolidated balance sheets of the Company as of December 31, 2023 and 2022, the related consolidated statements of operations, comprehensive (loss) income, changes in stockholders’ equity and cash flows for each of the three years in the period ended December 31, 2023, and the related notes and our report dated February 29, 2024 expressed an unqualified opinion thereon.
Basis for Opinion
The Company’s management is responsible for maintaining effective internal control over financial reporting and for its assessment of the effectiveness of internal control over financial reporting included in the accompanying Management's Report on Internal Control over Financial Reporting. Our responsibility is to express an opinion on the Company’s internal control over financial reporting based on our audit. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audit in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether effective internal control over financial reporting was maintained in all material respects.
Our audit included obtaining an understanding of internal control over financial reporting, assessing the risk that a material weakness exists, testing and evaluating the design and operating effectiveness of internal control based on the assessed risk, and performing such other procedures as we considered necessary in the circumstances. We believe that our audit provides a reasonable basis for our opinion.
Definition and Limitations of Internal Control Over Financial Reporting
A company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles. A company’s internal control over financial reporting includes those policies and procedures that (1) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the company; (2) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with generally accepted accounting principles, and that receipts and expenditures of the company are being made only in accordance with authorizations of management and directors of the company; and (3) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the company’s assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
/s/ Ernst & Young LLP
San Mateo, California
February 29, 2024
Item 9B. Other Information
Insider Trading Arrangements
None of the Company’s directors or officers (as defined in Section 16 of the Exchange Act) adopted or terminated a “Rule 10b5-1 trading arrangement” or a “non-Rule 10b5-1 trading arrangement” (each as defined in Item 408(a) and (c) of Regulation S-K) during the Company’s fiscal quarter ended December 31, 2023.
Item 9C. Disclosure Regarding Foreign Jurisdictions that Prevent Inspections
Not applicable.
Part III.
Item 10. Directors, Executive Officers and Corporate Governance
The information required by this Item is incorporated by reference to the Company’s 2024 Proxy Statement (the “2024 Proxy Statement”) to be filed with the SEC within 120 days after December 31, 2023 in connection with the solicitation of proxies for the Company’s 2024 annual meeting of stockholders.
Item 11. Executive Compensation
The information required by this Item is incorporated by reference to the 2024 Proxy Statement, which is expected to be filed no later than 120 days after the end of our fiscal year ended December 31, 2023.
Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters
The information required by this Item is incorporated by reference to the 2024 Proxy Statement, which is expected to be filed no later than 120 days after the end of our fiscal year ended December 31, 2023.
Item 13. Certain Relationships and Related Transactions and Director Independence
The information required by this Item is incorporated by reference to the 2024 Proxy Statement, which is expected to be filed no later than 120 days after the end of our fiscal year ended December 31, 2023.
Item 14. Principal Accounting Fees and Services
The information required by this Item is incorporated by reference to the 2024 Proxy Statement, which is expected to be filed no later than 120 days after the end of our fiscal year ended December 31, 2023.
Part IV.
Item 15. Exhibits and Financial Statement Schedules
(a) The following documents are filed as a part of this report:
(1) Consolidated Financial Statements (included in Item 8):
(2) Financial Statement Schedules
All schedules have been omitted because they are not applicable or not required, or because the required information is included either in the consolidated financial statements or in the notes thereto.
(3) Exhibits
| | | | | | | | |
| Exhibit Number | | Description |
| | |
| 2.1§ | | Agreement and Plan of Merger, dated as of August 5, 2021, among Maravai Life Sciences, Inc., Voyager Group Holdings, Inc., Maravai LifeSciences Holdings, Inc., and Maravai Intermediate Holdings, LLC (incorporated by reference to Exhibit 2.1 to Maravai LifeSciences Holdings, Inc.’s Form 8-K filed on August 10, 2021). |
| | |
| 2.2§ | | Amendment No. 1, dated as of September 2, 2021, to the Agreement and Plan of Merger, dated as of August 5, 2021, among Maravai Life Sciences, Inc., Voyager Group Holdings, Inc., Maravai LifeSciences Holdings, Inc., and Maravai Intermediate Holdings, LLC (incorporated by reference to Exhibit 2.1 to Maravai LifeSciences Holdings, Inc.’s Form 8-K filed on September 3, 2021). |
| | |
| 2.3 | | Letter Agreement, dated November 24, 2021, amending the Agreement and Plan of Merger, dated as of August 5, 2021, among Maravai Life Sciences, Inc., Voyager Group Holdings, Inc., Maravai LifeSciences Holdings, Inc., and Maravai Intermediate Holdings, LLC (incorporated by reference to Exhibit 2.3 to Maravai LifeSciences Holdings, Inc.'s Form 10-K filed on March 1, 2022). |
| | |
2.4 | | Amendment No. 2, dated as of August 30, 2022, to the Agreement and Plan of Merger, dated as of August 5, 2021, among Maravai Life Sciences, LLC (f/k/a Maravai Life Sciences, Inc.), Voyager Group Holdings, Inc., Vector Laboratories, Inc., Maravai LifeSciences Holdings, Inc., and Maravai Intermediate Holdings, LLC (incorporated by reference to Exhibit 2.1 to Maravai LifeSciences Holdings, Inc.’s Form 10-Q filed on November 4, 2022). |
| | |
| 3.1 | | |
| | |
| 3.2 | | |
| | |
| 4.1 | | |
| | |
| 4.2 | | |
| | |
| 10.1+ | | |
| | |
10.2+§ | | |
| | |
| | | | | | | | |
| Exhibit Number | | Description |
10.3+§ | | |
| | |
10.4+§ | | |
| | |
10.6 | | |
| | |
10.7 | | |
| | |
10.8 | | |
| | |
10.9+ | | |
| | |
10.10 | | |
| | |
10.11§ | | |
| | |
10.12§ | | |
| | |
10.13§ | | |
| | |
10.14§ | | |
| | |
10.15§ | | |
| | |
10.16 | | |
| | |
10.17 | | |
| | |
10.18§ | | |
| | |
10.19 | | |
| | |
10.20§ | | Credit Agreement, dated as of October 19, 2020, among Maravai Intermediate Holdings, LLC, Cygnus Technologies, LLC, Trilink Biotechnologies, LLC, Vector Laboratories, Inc., Maravai Topco Holdings, LLC and Morgan Stanley Senior Funding, Inc. (incorporated by reference to Exhibit 10.24 to Maravai LifeSciences Holdings, Inc.’s Form S-1 filed on October 29, 2020). |
| | |
10.21 | | |
| | |
10.22 | | Second Amendment to Credit Agreement, dated as of January 19, 2022, among Maravai Intermediate Holdings, LLC, Cygnus Technologies, LLC, Trilink Biotechnologies, LLC, Maravai Topco Holdings, LLC and Morgan Stanley Senior Funding, Inc. (incorporated by reference to Exhibit 10.1 to Maravai LifeSciences Holdings Inc.’s Form 8-K filed on January 20, 2022). |
| | |
10.23+§ | | |
| | |
| | | | | | | | |
| Exhibit Number | | Description |
10.24+ | | |
| | |
10.25+§ | | |
| | |
10.26+§ | | |
| | |
10.27+§ | | |
| | |
10.28+§ | | Amended and Restated Employment Agreement of William “Trey” Martin, III, effective as of May 8, 2023, among Maravai LifeSciences Holdings, Inc., Maravai Intermediate Holdings, LLC and William “Trey” Martin, III (incorporated by reference to Exhibit 10.3 to Maravai LifeSciences Holdings, Inc.’s Form 10-Q filed on May 9, 2023). |
| | |
10.29+§ | | Amended and Restated Employment Agreement of Peter Leddy, Ph.D., effective as of May 8, 2023, among Maravai LifeSciences Holdings, Inc., Maravai Intermediate Holdings, LLC and Peter Leddy, Ph.D. (incorporated by reference to Exhibit 10.5 to Maravai LifeSciences Holdings, inc.’s Form 10-Q filed on May 9, 2023). |
| | |
10.30+ | | |
| | |
10.31+ | | |
| | |
10.32+ | | |
| | |
10.33+ | | |
| | |
10.34+ | | |
| | |
10.35+ | | |
| | |
10.36+ | | |
| | |
10.37+ | | |
| | |
10.38+ | | |
| | |
| 21.1 | | |
| | |
| 23.1 | | |
| | |
| 31.1 | | |
| | |
| 31.2 | | |
| | |
| 32.1* | | |
| | |
| 32.2* | | |
| | |
97.1 | | |
| | |
| 101.INS | | XBRL Instance Document - the instance document does not appear in the Interactive Data File because its XBRL tags are embedded within the Inline XBRL document. |
| | |
| 101.SCH | | XBRL Taxonomy Extension Schema Document. |
| | |
| 101.CAL | | XBRL Taxonomy Extension Calculation Linkbase Document. |
| | |
| | | | | | | | |
| Exhibit Number | | Description |
| 101.DEF | | XBRL Extension Definition Linkbase Document. |
| | |
| 101.LAB | | XBRL Taxonomy Label Linkbase Document. |
| | |
| 101.PRE | | XBRL Taxonomy Extension Presentation Linkbase Document. |
| | |
| 104 | | Cover Page Interactive Data File (formatted as Inline XBRL and contained in Exhibit 101) |
_______________ | | | | | |
| * | The certifications furnished as Exhibit 32.1 and 32.2 hereto are deemed to accompany this Annual Report on Form 10-K and will not be deemed “filed” for purposes of Section 18 of the Exchange Act, or otherwise subject to the liabilities of that section, nor shall it be deemed incorporated by reference in any filing under the Securities Act or the Exchange Act, except as expressly set forth by specific reference in such filing. |
| + | Indicates a management contract or compensatory plan or agreement. |
| § | Exhibits and schedules have been omitted pursuant to Item 601(a)(5) of Regulation S-K and will be provided on a supplemental basis to the SEC upon request. |
(ii)Financial statement schedules
No financial statement schedules are provided because the information called for is not applicable or is shown in the financial statements or notes.
Item 16. Form 10-K Summary
None.
SIGNATURES
Pursuant to the requirements of the Securities Act of 1934, the registrant has duly caused this Annual Report on Form 10-K to be signed on its behalf by the undersigned, thereunto duly authorized.
| | | | | | | | | | | |
| Maravai LifeSciences Holdings, Inc. |
| | |
| By: | | /s/ William E. Martin, III |
| Name: | | William E. Martin, III |
| Title: | | Chief Executive Officer |
Date: February 29, 2024
***
Pursuant to the requirements of the Securities and Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicted.
| | | | | | | | | | | | | | |
| Signature | | Title | | Date |
| | | | |
| | Chief Executive Officer (Principal Executive Officer) | | February 29, 2024 |
/s/ William E. Martin, III | | |
William E. Martin, III | | |
| | | | |
/s/ Kevin Herde | | Chief Financial Officer (Principal Financial and Accounting Officer) | | February 29, 2024 |
Kevin Herde | | |
| | | | |
/s/ Carl Hull | | Executive Chairman of the Board | | February 29, 2024 |
Carl Hull | | |
| | | | |
/s/ Anat Ashkenazi | | Director | | February 29, 2024 |
Anat Ashkenazi | | |
| | | | |
/s/ Sean Cunningham | | Director | | February 29, 2024 |
Sean Cunningham | | |
| | | | |
/s/ Benjamin Daverman | | Director | | February 29, 2024 |
Benjamin Daverman | | |
| | | | |
/s/ John DeFord | | Director | | February 29, 2024 |
John DeFord, Ph.D. | | |
| | | | |
/s/ Susannah Gray | | Director | | February 29, 2024 |
Susannah Gray | | |
| | | | |
/s/ Jessica Hopfield | | Director | | February 29, 2024 |
Jessica Hopfield, Ph.D. | | |
| | | | |
/s/ Gregory T. Lucier | | Director | | February 29, 2024 |
Gregory T. Lucier | | |
| | | | |
/s/ Luke Marker | | Director | | February 29, 2024 |
Luke Marker | | |
| | | | |
/s/ Constantine Mihas | | Director | | February 29, 2024 |
Constantine Mihas | | |
| | | | |
/s/ Murali K. Prahalad | | Director | | February 29, 2024 |
Murali K. Prahalad | | |
Description of Registrant’s Securities Registered Pursuant to Section 12 of the Securities and Exchange Act of 1934.
General
As of December 31, 2023, Maravai LifeSciences Holdings, Inc. (the “Company,” “we,” “our,” and “us”) had one class of securities, our Class A common stock, par value $0.01 per share, registered under Section 12 of the Securities Exchange Act of 1934, as amended.
The following summary of terms of our Class A common stock is based upon our amended and restated certificate of incorporation (our “certificate” or “certificate of incorporation”) and our amended and restated by-laws (our “bylaws”) currently in effect under Delaware law. This summary is not complete and is subject to, and qualified in its entirety by reference to, the certificate and the bylaws, which are filed as Exhibits 3.1 and 3.2 to our Annual Report on Form 10-K of which this Exhibit 4.2 is a part. We encourage you to read these documents and the applicable portion of the Delaware General Corporation Law, as amended (the “DGCL”), carefully.
Under our certificate, we have the authority to issue 500,000,000 shares of Class A common stock, par value $0.01 per share (“Class A common stock”), 300,000,000 shares of Class B common stock, par value $0.01 per share (“Class B common stock”), and 50,000,000 shares of preferred stock, par value $0.01 per share (“preferred stock”).
Class A Common Stock
As of February 21, 2024, there were 132,305,845 shares of Class A common stock outstanding.
Voting Rights
Holders of shares of our Class A common stock are entitled to one vote for each share held of record on all matters submitted to a vote of shareholders. The holders of our Class A common stock do not have cumulative voting rights in the election of directors.
Holders of shares of our Class A common stock vote together with holders of our Class B common stock as a single class on all matters presented to our shareholders for their vote or approval, except for certain amendments to our certificate of incorporation described below or as otherwise required by applicable law or the certificate.
Dividend Rights
Holders of shares of our Class A common stock are entitled to receive dividends when and if declared by our Board out of funds legally available therefor, subject to any statutory or contractual restrictions on the payment of dividends and to any restrictions on the payment of dividends imposed by the terms of any outstanding preferred stock.
Liquidation Rights
Upon our dissolution or liquidation or the sale of all or substantially all of our assets, after payment in full of all amounts required to be paid to creditors and to the holders of preferred stock having liquidation preferences, if any, the holders of shares of our Class A common stock will be entitled to receive pro rata our remaining assets available for distribution.
Preemptive, Conversion or Redemption Rights
Holders of shares of our Class A common stock do not have preemptive, subscription, redemption or conversion rights. There are no redemption or sinking fund provisions applicable to the Class A common stock.
Class B Common Stock
As of February 21, 2024, there were 119,094,026 shares of Class B common stock outstanding. Maravai Life Sciences Holdings, LLC owns 100% of our outstanding Class B common stock.
Voting Rights
Holders of shares of our Class B common stock are entitled to one vote for each share held of record on all matters submitted to a vote of shareholders. The holders of our Class B common stock do not have cumulative voting rights in the election of directors.
Holders of shares of our Class B common stock vote together with holders of our Class A common stock as a single class on all matters presented to our shareholders for their vote or approval, except for certain amendments to our certificate of incorporation described below or as otherwise required by applicable law or the certificate.
Dividend or Liquidation Rights
Holders of our Class B common stock do not have any right to receive dividends or to receive a distribution upon dissolution or liquidation or the sale of all or substantially all of our assets.
Preemptive, Conversion or Redemption Rights
Holders of shares of our Class B common stock do not have preemptive, subscription, redemption or conversion rights. There are no redemption or sinking fund provisions applicable to the Class B common stock.
Amendments
Any amendment of our certificate of incorporation that gives holders of our Class B common stock (1) any rights to receive dividends or any other kind of distribution, (2) any right to convert into or be exchanged for Class A common stock or (3) any other economic rights will
require, in addition to shareholder approval, the affirmative vote of holders of our Class A common stock voting separately as a class.
Preferred Stock
As of February 21, 2024, there were no shares of preferred stock outstanding.
Our Board is authorized to direct us to issue shares of preferred stock in one or more series without shareholder approval. Our Board has the discretion to determine the rights, preferences, privileges and restrictions, including voting rights, dividend rights, conversion rights, redemption privileges and liquidation preferences, of each series of preferred stock.
The purpose of authorizing our Board to issue preferred stock and determine its rights and preferences is to eliminate delays associated with a shareholder vote on specific issuances. The issuance of preferred stock, while providing flexibility in connection with possible acquisitions, future financings and other corporate purposes, could have the effect of making it more difficult for a third party to acquire, or could discourage a third party from seeking to acquire, a majority of our outstanding voting stock. Additionally, the issuance of preferred stock may adversely affect the holders of our Class A common stock by restricting dividends on the Class A common stock, diluting the voting power of the Class A common stock or subordinating the liquidation rights of the Class A common stock. As a result of these or other factors, the issuance of preferred stock could have an adverse impact on the market price of our Class A common stock.
Forum Selection
Our certificate of incorporation provides that, unless we consent in writing to the selection of an alternative forum, the Court of Chancery of the State of Delaware is the sole and exclusive forum for any state court action for (1) any derivative action or proceeding brought on our behalf, (2) any action asserting a claim of breach of a fiduciary duty owed by any of our directors, officers or other employees to us or our shareholders, (3) any action asserting a claim against Maravai LifeSciences Holdings, Inc. or any director or officer thereof arising pursuant to any provision of the DGCL or as to which the DGCL confers jurisdiction on the Court of Chancery of the State of Delaware, our certificate of incorporation or our bylaws or (4) any other action asserting a claim against Maravai LifeSciences Holdings, Inc. or any director or officer thereof that is governed by the internal affairs doctrine; provided that, for the avoidance of doubt, the forum selection provision that identifies the Court of Chancery of the State of Delaware as the exclusive forum for certain litigation, including any “derivative action,” does not apply to suits to enforce a duty or liability created by the Securities Act, the Exchange Act or any other claim for which the federal courts have exclusive jurisdiction. Our certificate of incorporation also provides that, unless we consent in writing to the selection of an alternative forum, the federal district courts of the United States shall be the exclusive forum for the resolution of any complaint asserting a cause of action arising under the Securities Act. Any person or entity purchasing or otherwise acquiring any interest in shares of our capital stock is deemed to have notice of and to have consented to the provisions of our certificate of incorporation described
above. Although we believe these provisions benefit us by providing increased consistency in the application of Delaware law or the Securities Act, as applicable, for the specified types of actions and proceedings, the provisions may have the effect of discouraging lawsuits against us or our directors and officers. Alternatively, if a court were to find any of the forum selection provisions contained in our amended and restated certificate of incorporation to be inapplicable or unenforceable, we may incur additional costs associated with having to litigate such action in other jurisdictions, which could have an adverse effect on our business, financial condition, results of operations, cash flows and prospects and result in a diversion of the time and resources of our employees, management and board of directors.
Anti-Takeover Provisions
Our certificate, bylaws and the DGCL contain provisions, which are summarized in the following paragraphs, that are intended to enhance the likelihood of continuity and stability in the composition of our Board. These provisions are intended to avoid costly takeover battles, reduce our vulnerability to a hostile change of control and enhance the ability of our Board to maximize shareholder value in connection with any unsolicited offer to acquire us. However, these provisions may have an anti-takeover effect and may delay, deter or prevent a merger or acquisition of us by means of a tender offer, a proxy contest or other takeover attempt that a shareholder might consider in its best interest, including those attempts that might result in a premium over the prevailing market price for the shares of Class A common stock held by shareholders.
These provisions include:
Classified Board. Our certificate provides that our Board is divided into three classes of directors, with the classes as nearly equal in number as possible, and with the directors serving three-year terms. As a result, approximately one-third of our Board is elected each year. The classification of the directors has the effect of making it more difficult for shareholders to change the composition of our Board. Our certificate also provides that, subject to any rights of holders of preferred stock to elect additional directors under specified circumstances, the number of directors is fixed exclusively pursuant to resolutions adopted by our Board. Our Board currently has 11 members.
Shareholder Action by Written Consent. Our certificate precludes shareholder action by written consent at any time when GTCR, LLC (“GTCR”) controls, in the aggregate, less than 35% in voting power of our outstanding common stock.
Special Meetings of Shareholders. Our certificate and bylaws provide that, except as required by law, special meetings of our shareholders may be called at any time only by or at the direction of our Board or the chairman of our Board; provided, however, at any time when GTCR controls, in the aggregate, at least 35% in voting power of our outstanding common stock, special meetings of our shareholders shall also be called by our Board or the chairman of our Board at the request of GTCR. Our bylaws prohibit the conduct of any business at a special meeting other than as specified in the notice for such meeting. These provisions may have the
effect of deferring, delaying or discouraging hostile takeovers, or changes in control or management of us.
Advance Notice Procedures. Our bylaws establish advance notice procedures for shareholder proposals and nomination of candidates for election as directors, other than nominations made by or at the direction of our Board or a committee of our Board, and provided, however, that at any time when GTCR controls, in the aggregate, at least 10% of the voting power of our outstanding common stock, such advance notice procedure does not apply to GTCR. Shareholders at an annual meeting are only be able to consider proposals or nominations specified in the notice of meeting or brought before the meeting by or at the direction of our Board or by a shareholder who was a shareholder of record on the record date for the meeting, who is entitled to vote at the meeting and who has given our Secretary timely written notice, in proper form, of the shareholder’s intention to bring that business before the meeting. Although the bylaws do not give our Board the power to approve or disapprove shareholder nominations of candidates or proposals regarding other business to be conducted at a special or annual meeting, the bylaws may have the effect of precluding the conduct of certain business at a meeting if the proper procedures are not followed or may discourage or deter a potential acquirer from conducting a solicitation of proxies to elect its own slate of directors or otherwise attempting to obtain control of us. These provisions do not apply to nominations by GTCR pursuant to the Director Nomination Agreement.
Removal of Directors; Vacancies. Our certificate provides that a director nominated by GTCR may be removed with or without cause by GTCR; provided, however, that at any time when GTCR controls less than 40% in voting power of our outstanding common stock, all directors, including those nominated by GTCR, may only be removed for cause, and only by the affirmative vote of holders of at least 66 2/3% in voting power of all the then-outstanding shares of capital stock of the company entitled to vote thereon, voting together as a single class. In addition, our certificate also provides that, subject to the rights granted to one or more series of preferred stock then outstanding, any newly created directorship on our Board that results from an increase in the number of directors and any vacancies on our Board will be filled only by the affirmative vote of a majority of the remaining directors, even if less than a quorum, or by a sole remaining director (and not by the shareholders).
Supermajority Approval Requirements. Our certificate and bylaws provide that our Board is expressly authorized to make, alter, amend, change, add to, rescind or repeal, in whole or in part, our bylaws without a shareholder vote in any matter not inconsistent with the laws of the State of Delaware and our certificate. For as long as GTCR controls, in the aggregate, at least 50% in voting power of our outstanding common stock, any amendment, alteration, rescission or repeal of our bylaws by our shareholders requires the affirmative vote of a majority in voting power of the outstanding shares of our stock entitled to vote on such amendment, alteration, change, addition, rescission or repeal. At any time when GTCR controls, in the aggregate, less than 50% in voting power of our outstanding common stock, any amendment, alteration, rescission or repeal of our bylaws by our shareholders requires the affirmative vote of the holders of at least 66 2/3% in voting power of all the then-outstanding shares of stock of the company entitled to vote thereon, voting together as a single class.
The DGCL provides generally that the affirmative vote of a majority of the outstanding shares entitled to vote thereon, voting together as a single class, is required to amend a corporation’s certificate of incorporation, unless the certificate requires a greater percentage.
Our certificate provides that the following provisions in our certificate may be amended, altered, repealed or rescinded only by the affirmative vote of the holders of at least 66 2/3% (as opposed to a majority threshold) in voting power of all the then-outstanding shares of stock entitled to vote thereon, voting together as a single class at any time when GTCR controls, in the aggregate, less than 50% in voting power of our outstanding common stock (except as otherwise noted below):
•the provision requiring a 66 2/3% supermajority vote for shareholders to amend our bylaws;
•the provisions providing for a classified board of directors (the election and term of our directors);
•the provisions regarding resignation and removal of directors;
•the provisions regarding entering into business combinations with interested shareholders;
•the provisions regarding shareholder action by written consent;
•the provisions regarding calling special meetings of shareholders;
•the provisions regarding filling vacancies on our Board and newly created directorships;
•the provision establishing the Court of Chancery of the State of Delaware as the exclusive forum for certain litigation;
•the provision establishing the federal district courts of the United States as the exclusive forum for litigation arising under the Securities Act;
•the provisions eliminating monetary damages for breaches of fiduciary duty by a director (which requires a super majority vote, regardless of GTCR's then-current ownership); and
•the amendment provision requiring that the above provisions be amended only with a 66 2/3% supermajority vote.
The combination of the classification of our Board, the lack of cumulative voting and the supermajority voting requirements will make it more difficult for our existing shareholders to replace our Board as well as for another party to obtain control of us by replacing our Board. Because our Board has the power to retain and discharge our officers, these provisions could also
make it more difficult for existing shareholders or another party to effect a change in management.
Authorized but Unissued Shares. Our authorized but unissued shares of common stock and preferred stock will be available for future issuance without shareholder approval, subject to stock exchange rules. These additional shares of capital stock may be utilized for a variety of corporate purposes, including future public offerings to raise additional capital, corporate acquisitions and employee benefit plans. One of the effects of the existence of authorized but unissued common stock or preferred stock may be to enable our Board to issue shares of capital stock to persons friendly to current management, which issuance could render more difficult or discourage an attempt to obtain control of the company by means of a merger, tender offer, proxy contest or otherwise, and thereby protect the continuity of our management and possibly deprive our shareholders of opportunities to sell their shares of common stock at prices higher than prevailing market prices.
Business Combinations. We are not subject to the provisions of Section 203 of the DGCL. In general, Section 203 prohibits a publicly held Delaware corporation from engaging in a “business combination” with an “interested shareholder” for a three-year period following the time that the person becomes an interested shareholder, unless the business combination is approved in a prescribed manner. A “business combination” includes, among other things, a merger, asset or stock sale or other transaction resulting in a financial benefit to the interested shareholder. An “interested shareholder” is a person who, together with affiliates and associates, owns, or did own within three years prior to the determination of interested shareholder status, 15% or more of the corporation’s voting stock.
Under Section 203, a business combination between a corporation and an interested shareholder is prohibited unless it satisfies one of the following conditions: (1) before the shareholder became an interested shareholder, the Board approved either the business combination or the transaction which resulted in the shareholder becoming an interested shareholder; (2) upon consummation of the transaction which resulted in the shareholder becoming an interested shareholder, the interested shareholder owned at least 85% of the voting stock of the corporation outstanding at the time the transaction commenced, excluding for purposes of determining the voting stock outstanding, shares owned by persons who are directors and also officers, and employee stock plans, in some instances; or (3) at or after the time the shareholder became an interested shareholder, the business combination was approved by the Board and authorized at an annual or special meeting of the shareholders by the affirmative vote of at least two-thirds of the outstanding voting stock which is not owned by the interested shareholder.
A Delaware corporation may “opt out” of these provisions with an express provision in its original certificate of incorporation or an express provision in its certificate of incorporation or bylaws resulting from a shareholders’ amendment approved by at least a majority of the outstanding voting shares.
We opted out of Section 203; however, our certificate contains similar provisions providing that we may not engage in certain “business combinations” with any “interested
shareholder” for a three-year period following the time that the shareholder became an interested shareholder, unless:
•prior to such time, our Board approved either the business combination or the transaction which resulted in the shareholder becoming an interested shareholder;
•upon consummation of the transaction that resulted in the shareholder becoming an interested shareholder, the interested shareholder owned at least 85% of our voting stock outstanding at the time the transaction commenced, excluding certain shares; or
•at or subsequent to that time, the business combination is approved by our Board and by the affirmative vote of holders of at least 66 2/3% of our outstanding voting stock that is not owned by the interested shareholder.
Under certain circumstances, this provision will make it more difficult for a person who would be an “interested shareholder” to effect various business combinations with us for a three-year period. This provision may encourage companies interested in acquiring us to negotiate in advance with our Board because the shareholder approval requirement would be avoided if our Board approves either the business combination or the transaction which results in the shareholder becoming an interested shareholder. These provisions also may have the effect of preventing changes in our Board and may make it more difficult to accomplish transactions which shareholders may otherwise deem to be in their best interests.
Our certificate of incorporation provides that GTCR, and any of its direct or indirect transferees and any group as to which such persons are a party, do not constitute “interested shareholders” for purposes of this provision.
Limitations on Liability and Indemnification of Officers and Directors
The DGCL authorizes corporations to limit or eliminate the personal liability of directors to corporations and their shareholders for monetary damages for breaches of directors’ fiduciary duties, subject to certain exceptions. Our certificate includes a provision that eliminates the personal liability of directors for monetary damages for any breach of fiduciary duty as a director, except to the extent such exemption from liability or limitation thereof is not permitted under the DGCL. The effect of these provisions will be to eliminate the rights of us and our shareholders, through shareholders’ derivative suits on our behalf, to recover monetary damages from a director for breach of fiduciary duty as a director, including breaches resulting from grossly negligent behavior. However, exculpation does not apply to any director if the director has acted in bad faith, knowingly or intentionally violated the law, authorized illegal dividends or redemptions or derived an improper benefit from his or her actions as a director.
Our bylaws provide that we must indemnify and advance expenses to our directors and officers to the fullest extent authorized by the DGCL. We also are expressly authorized to carry directors’ and officers’ liability insurance providing indemnification for our directors, officers and certain employees for some liabilities. We believe that these indemnification and
advancement provisions and insurance are useful to attract and retain qualified directors and officers.
The limitation of liability, indemnification and advancement provisions included in our certificate of incorporation and bylaws may discourage shareholders from bringing a lawsuit against directors for breaches of their fiduciary duty. These provisions also may have the effect of reducing the likelihood of derivative litigation against directors and officers, even though such an action, if successful, might otherwise benefit us and our shareholders. In addition, your investment may be adversely affected to the extent we pay the costs of settlement and damage awards against directors and officers pursuant to these indemnification provisions.
There is currently no pending material litigation or proceeding involving any of our directors, officers or employees for which indemnification is sought.
Corporate Opportunity Doctrine
Delaware law permits corporations to adopt provisions renouncing any interest or expectancy in certain opportunities that are presented to the corporation or its officers, directors or shareholders. Our certificate, to the maximum extent permitted from time to time by Delaware law, renounces any interest or expectancy that we have in, or right to be offered an opportunity to participate in, specified business opportunities that are from time to time presented to certain of our officers, directors or shareholders or their respective affiliates, other than those officers, directors, shareholders or affiliates who are our or our subsidiaries’ employees. Our certificate provides that, to the fullest extent permitted by law, none of GTCR or any director who is not employed by us (including any non-employee director who serves as one of our officers in both his or her director and officer capacities) or its, his or her affiliates have any duty to refrain from (1) engaging in a corporate opportunity in the same or similar lines of business in which we or our affiliates now engage or propose to engage or (2) otherwise competing with us or our affiliates. In addition, to the fullest extent permitted by law, in the event that GTCR or any non-employee director acquires knowledge of a potential transaction or other business opportunity which may be a corporate opportunity for itself, himself or herself or its, his or her affiliates or for us or our affiliates, such person has no duty to communicate or offer such transaction or business opportunity to us or any of our affiliates and they may take any such opportunity for themselves or offer it to another person or entity. Our certificate does not renounce our interest in any business opportunity that is expressly offered to a non-employee director solely in his or her capacity as a director or officer of Maravai LifeSciences Holdings, Inc. To the fullest extent permitted by law, no business opportunity will be deemed to be a potential corporate opportunity for us unless we would be permitted to undertake the opportunity under our certificate, we have sufficient financial resources to undertake the opportunity and the opportunity would be in line with our business.
Dissenters’ Rights of Appraisal and Payment
Under the DGCL, with certain exceptions, our shareholders have appraisal rights in connection with a merger or consolidation of Maravai LifeSciences Holdings, Inc. Pursuant to the DGCL, shareholders who properly request and perfect appraisal rights in connection with
such merger or consolidation have the right to receive payment of the fair value of their shares of capital stock as determined by the Delaware Court of Chancery.
Shareholders’ Derivative Actions
Under the DGCL, any of our shareholders may bring an action in our name to procure a judgment in our favor, also known as a derivative action, provided that the shareholder bringing the action is a holder of our shares of capital stock at the time of the transaction to which the action relates or such shareholder’s stock thereafter devolved by operation of law.
Transfer Agent and Registrar
The transfer agent and registrar for our Class A common stock is Equiniti Trust Company, LLC. Its address is 48 Wall Street, Floor 23, New York, 10005.
Listing
Our Class A common stock is listed on The Nasdaq Global Select Market under the trading symbol “MRVI.”
DocumentLEASE AGREEMENT
BETWEEN
10240 FLANDERS INVESTORS LLC,
a Delaware limited liability company
(LANDLORD)
AND
TRILINK BIOTECHNOLOGIES, LLC,
a Delaware limited liability company
(TENANT)
August 6, 2021
10240-10247 FLANDERS COURT
SAN DIEGO, CALIFORNIA
| | |
The table of contents is empty. Heading styles must be applied in the document and be selected in the table of contents properties panel. |
LIST OF EXHIBITS
| | | | | | | | | | | |
| EXHIBIT SECTION REFERENCE |
| A | - | Project Site Plan | 2.1 |
| B | - | Work Letter Agreement | 2.1 |
| Sch. B-1 | - | Landlord Work | Exhibit B |
| C-1 | - | Form of Memorandum of Lease Terms for Premises A | 3 |
| C-2 | - | Form of Memorandum of Lease Terms for Premises B | 3 |
| D | - | Lease Guaranty | 7.2 |
| E | - | Form of Tenant Estoppel Certificate | 29 |
| F | - | Rules and Regulations | 31 |
| G | - | Parking Rules and Regulations | 40 |
| H | - | Phase I Environmental Site Assessment – Executive Summary | 41.2 |
LEASE AGREEMENT
THIS LEASE AGREEMENT (this “Lease”) is made as of August 6, 2021 (“Effective Date”), by and between 10240 FLANDERS INVESTORS LLC, a Delaware limited liability company (“Landlord”), and TRILINK BIOTECHNOLOGIES, LLC, a Delaware limited liability company (“Tenant”).
Article 1
TERMS AND DEFINITIONS
For the purposes of this Lease, the following terms shall have the following definitions and meanings:
1.1Landlord: 10240 Flanders Investors LLC, a Delaware limited liability company
1.2Landlord’s Address:
10240 Flanders Investors LLC
c/o Bioscience Properties, Inc.
514 Via De La Valle, Suite 300A
Solana Beach, CA 92075
Attention: _____________
1.3Tenant: Trilink Biotechnologies, LLC, a Delaware limited liability company
1.4Tenant’s Address:
Prior to the Commencement Date:
Trilink Biotechnologies, LLC
10770 Wateridge Circle, Suite 110
San Diego, CA 92121
Attn: _________________
As of the Commencement Date:
Trilink Biotechnologies, LLC
10770 Wateridge Circle, Suite 110
San Diego, CA 92121
Attn: _________________
1.5Buildings: The two (2) single-story buildings located at 10240 and 10247 Flanders Court, San Diego, California 92121, which have a total of approximately 65,006 rentable square feet of area (“Rentable Square Feet”).
1.6Premises: (i) Approximately 32,236 Rentable Square Feet within the Building known as 10240 Flanders Court, San Diego, California (“Premises A”), and (ii) approximately 32,770 Rentable Square Feet within the Building known as 10247 Flanders Court, San Diego, California (“Premises B”). Premises A and Premises B comprise of all of the rentable square footage within the Buildings.
1.7Initial Term: Commencing on the Commencement Date for the applicable Premises and expiring on the date which is one hundred twenty-six (126) months after the Premises A Commencement Date.
1.8Tenant’s Vehicle Parking Spaces: (i) For the period beginning on the Premises A Commencement Date and ending on the day prior to the Premises B Commencement Date, 3.7 unreserved parking spaces per 1,000 Rentable Square Feet of Premises A (i.e., 119 parking spaces), and (ii) for the period beginning on the Premises B Commencement Date and continuing through the end of the Term, all spaces within the Parking Area (defined in Article 33 below) (i.e., 220 parking spaces), in each case at no additional charge during the Term, subject to the terms and conditions of Article 33 below.
1.9Tenant Improvement Allowance: Up to One Hundred Seventy-Six and 75/100 Dollars ($176.75) per Rentable Square Foot of the applicable Premises (i.e., up to $5,697,713.00 for Premises A and up to $5,792,097.50 for Premises B) to be contributed by Landlord toward the cost of constructing the Tenant Improvements within the applicable Premises pursuant to the Work Letter Agreement described in Section 2.1 below.
1.10Premises A Commencement Date: The earlier to occur of (i) the date upon which Tenant first commences to conduct business in Premises A, and (ii) the date on which the Landlord Work and Tenant Improvements within Premises A are Substantially Complete pursuant to the terms and conditions of, and as that term is defined in, the Work Letter Agreement.
Premises B Commencement Date: The earlier to occur of (i) the date upon which Tenant first commences to conduct business in Premises B, and (ii) the date on which the existing tenant of Premises B has fully vacated and the Landlord Work and Tenant Improvements within Premises B are Substantially Complete pursuant to the terms and conditions of, and as that term is defined in, the Work Letter Agreement.
1.11Estimated Commencement Date: The date set forth in the mutually approved Work Schedule (as that term is defined in the Work Letter Agreement) as the estimated date of Substantial Completion of the Landlord Work and Tenant Improvements within the applicable Premises.
1.12Basic Rent:
Basic Rent for Premises A:
| | | | | | | | | | | |
| Months of Initial Term | Basic Rent per Rentable Square Foot ($/mo) | Monthly Installments of Basic Rent ($/mo) | Annual
Basic Rent ($/yr) |
| 1-12* | $4.10 | $132,167.60 | $1,586,011.20 |
| 13-24 | $4.22 | $136,132.63 | $1,633,591.56 |
| 25-36 | $4.35 | $140,216.61 | $1,682,599.32 |
| 37-48 | $4.48 | $144,423.11 | $1,733,077.32 |
| 49-60 | $4.61 | $148,755.80 | $1,785,069.60 |
| 61-72 | $4.75 | $153,218.47 | $1,838,621.64 |
| 73-84 | $4.90 | $157,815.02 | $1,893,780.24 |
| 85-96 | $5.04 | $162,549.47 | $1,950,593.64 |
| 97-108 | $5.19 | $167,425.95 | $2,009,111.40 |
| 109-120 | $5.35 | $172,448.73 | $2,069,384.76 |
| 121-126 | $5.51 | $177,622.19 | $2,131,466.28 |
*Provided that Tenant is not in default under this Lease beyond any applicable notice and cure period, monthly installments of Basic Rent shall be abated for six (6) full months (during months two (2) through the end of month seven (7) of the Initial Term for Premises A) pursuant to the terms and conditions of Section 5.1 below.
Basic Rent for Premises B:
| | | | | | | | | | | |
| Months of Initial Term | Basic Rent per Rentable Square Foot ($/mo) | Monthly Installments of Basic Rent ($/mo) | Annual
Basic Rent ($/yr) |
| 1-12* | $4.10 | $134,357.00 | $1,612,284.00 |
| 13-24 | $4.22 | $138,387.71 | $1,660,652.52 |
| 25-36 | $4.35 | $142,539.34 | $1,710,472.08 |
| 37-48 | $4.48 | $146,815.52 | $1,761,786.24 |
| 49-60 | $4.61 | $151,219.99 | $1,814,639.88 |
| 61-72 | $4.75 | $155,756.59 | $1,869,079.08 |
| 73-84 | $4.90 | $160,429.29 | $1,925,151.48 |
| 85-96 | $5.04 | $165,242.17 | $1,982,906.04 |
| 97-108 | $5.19 | $170,199.44 | $2,042,393.28 |
| 109-120 | $5.35 | $175,305.42 | $2,103,665.04 |
| 121-Expiration Date | $5.51 | $180,564.58 | $2,166,774.96 |
*Provided that Tenant is not in default under this Lease beyond any applicable notice and cure period, monthly installments of Basic Rent for Premises B shall be abated for six (6) full months (during months two (2) through the end of month seven (7) of the Initial Term for Premises B) pursuant to the terms and conditions of Section 5.1 below.
1.13Tenant’s Percentage: (i) As of the Premises A Commencement Date, 49.59%, and (ii) as of the Premises B Commencement Date, 100%.
1.14Security Deposit: $358,186.77.
Guarantor: Maravai Intermediate Holdings, LLC, a Delaware limited liability company
1.15Broker(s): CBRE, Inc. (Ted Jacobs), representing Tenant, and Cushman & Wakefield (Brian Starck and Jack Hughson), representing Landlord.
1.16Permitted Use: Office, laboratory, manufacturing, general office and research and development and any and all uses ancillary thereto, and no other use, subject to compliance with all applicable Laws (defined below).
Article 2
PREMISES AND COMMON AREAS
1.1Premises. Landlord hereby leases to Tenant and Tenant hereby leases from Landlord Premises A and Premises B (collectively, the “Premises”). The Premises consist of the Buildings and the corridor connecting such Buildings, which, together with the Parking Area, is located on the parcel or parcels of real property (“Project Site”) outlined on the Project Site Plan attached hereto, marked as Exhibit “A”, and incorporated herein by this reference (“Project Site Plan”) (all of which, together with the Building Common Areas and the Project Common Areas, as hereinafter defined, are collectively referred to as the “Project”). The Premises are leased in their “AS-IS” condition in accordance with Article 14; provided however, the Premises will be improved by Landlord with the Tenant Improvements described in the Work Letter Agreement, a copy of which is attached hereto, marked as Exhibit “B” and incorporated herein by this reference (“Work Letter Agreement”). The parties hereto agree that this Lease is upon and subject to the terms, covenants and conditions herein set forth. Each of Landlord and Tenant covenants as a material part of the consideration for this Lease to keep and perform each and all of said terms, covenants and conditions by it to be kept and performed.
1.2Rentable Area. Landlord and Tenant stipulate and agree that the Rentable Square Feet contained in the Premises is as specified in Section 1.6. The Basic Rent, Tenant’s Percentage and Security Deposit specified in Sections 1.12, 1.13 and 1.14 above, respectively, are based upon the Rentable Square Feet of the Premises set forth in Section 1.6.
1.3Common Areas. Tenant and its employees, invitees and agents shall have the nonexclusive right to use in common with Landlord and other tenants or occupants of the Project and their respective employees, invitees and agents, subject to the Rules and Regulations referred to in Section 36.1 below and all covenants, conditions and restrictions affecting the Project, any of the following areas which may be appurtenant to the Premises (collectively, “Common Areas”):
1.3.1.any common entrances, lobbies, shared entry lobbies and corridors, shared restrooms, service areas, elevators, stairways, accessways and/or ramps which may be located in the Building, and any common pipes, wires and appurtenant equipment which may be serving the Premises (collectively, “Building Common Areas”); and
1.3.2.the Parking Area and any loading and unloading areas, trash areas, service areas, parking areas, roadways, sidewalks, walkways, plazas, parkways, driveways, landscaped areas and similar areas and facilities from time to time situated within the Project (collectively, “Project Common Areas”).
Notwithstanding anything to the contrary set forth in this Section 2.3, after the Premises B Commencement Date, Tenant and its employees, invitees and agents shall have the exclusive right to use the Common Areas, subject to the terms and conditions of this Lease, including, without limitation, Section 2.4 below and Landlord’s right to perform Landlord’s maintenance and repair obligations as provided in this Lease.
1.4Landlord’s Reservation of Rights. Landlord reserves for itself, and for the owner(s) and operator(s) of the Project or any portion thereof, the right from time to time without material interference with Tenant’s access to the Premises or Tenant’s Permitted Use and without reducing the parking available in the Parking Area below 220 parking spaces (provided, however, the parking available in the Parking Area may be reduced below 220 parking spaces if the reduction is required by applicable governmental authorities):
1.4.1.to install, use, maintain, repair and replace pipes, ducts, conduits, wires and appurtenant meters and equipment for service to other parts of the Building above the ceiling surfaces, below the floor surfaces, within the walls and in the central core areas of the Premises, and to relocate any pipes, ducts, conduits, wires and appurtenant meters and equipment which are located in the Premises or elsewhere, and to expand the Parking Area;
1.4.2.to make changes in its sole but reasonable discretion (but subject to the prior written approval of Tenant, which will not be unreasonably withheld, conditioned or delayed) to the Common Areas, including, without limitation, changes in the location, size, shape and number of driveways, entrances, parking spaces, parking areas, loading and unloading areas, ingress, egress, direction of traffic, landscaped areas and walkways;
1.4.3.to close temporarily any of the Common Areas for maintenance purposes and to avoid claims of prescriptive rights so long as reasonable access to the Premises remains available;
1.4.4.to use the Common Areas while engaged in making additional improvements, repairs or alterations to the Building, the Parking Area or the Project, or any portion thereof; and
1.4.5.to do and perform such other acts and make such other changes in, to or with respect to the Project or any portion thereof as Landlord and/or the owner(s) and/or operator(s) thereof may reasonably deem to be appropriate or as may be required by applicable Laws.
Article 3
TERM
1.1Initial Term. The “Initial Term” of this Lease shall be for the period designated in Section 1.7, commencing on the Premises A Commencement Date with respect to Premises A and commencing on the Premises B Commencement Date with respect to Premises B, and expiring for both Premises on the date which is one hundred twenty-six (126) months after the Premises A Commencement Date, unless sooner terminated as hereinafter provided; provided that if the Commencement Date occurs on a day other than the first day of any calendar month, for purposes of calculating the date (“Expiration Date”) on which the Term is scheduled to expire and the timing of all scheduled increases in Basic Rent during the Term, the Commencement Date shall be deemed to be the first day of the calendar month following the Commencement Date. The Premises A Commencement Date shall be specified in a Memorandum of Lease Terms for Premises A, which shall be in the form of Exhibit “C-1”, attached hereto and incorporated herein by this reference, and shall be executed by Tenant as soon as practicable after the Premises A Commencement Date. The Premises B Commencement Date and the date upon which the Initial Term of this Lease shall end unless sooner terminated pursuant to the provisions hereof shall be specified in a Memorandum of Lease Terms for Premises B, which shall be in the form of Exhibit “C-2”, attached hereto and incorporated herein by this reference, and shall be executed by Tenant as soon as practicable after the Premises B Commencement Date. As used herein, “Term” shall refer to the Initial Term as it may be extended by written agreement of Landlord and Tenant, including, without limitation, as a result of Tenant’s exercise of its Option in accordance with Section 3.2 below.
1.2Option Term. Tenant shall have the right and option (“Option”) to extend the Term of this Lease for one (1) additional period of five (5) years (“Option Term”). The Option Term shall commence on the day immediately succeeding the Expiration Date and shall end on the day immediately preceding the fifth (5th) anniversary of the first day of the Option Term. Notwithstanding any provision of this Section 3.2 to the contrary, the Option shall be personal to the original Tenant named herein (i.e., Trilink Biotechnologies, LLC) (“Original Tenant”) and any Permitted Transferee to whom this Lease is assigned pursuant to a Permitted Transfer (as those terms are defined in Section 27.1.1 below).
1.1.1.Tenant shall exercise the Option by giving written notice to Landlord of its election to do so not earlier than twelve (12) months and not later than nine (9) months prior to the Expiration Date. The giving of such notice of extension by Tenant shall automatically extend the Term of this Lease for the Option Term, and no instrument of renewal or extension need be executed. In the event that Tenant fails to give timely notice to Landlord, this Lease shall automatically terminate at the end of the Initial Term and Tenant shall have no further option to extend the Term of this Lease. The Option shall be exercisable by Tenant only on the express condition that (i) at the time of the exercise, and at all times between the time of such exercise and the commencement of the Option Term, Tenant shall not be in Default under any of the provisions of this Lease, and (ii) Tenant shall not have been ten (10) or more days late in the payment of Monthly Basic Rent more than once during any twelve (12) consecutive month period during the Term.
1.1.2.The Option Term shall be on all the terms and conditions of this Lease, except that: (i) Tenant shall have no further right or option to extend the Term as provided by this Section 3.2; (ii) the Basic Rent for the Option Term shall be equal to the Fair Market Rental Value of the Premises, determined pursuant to Subsection 3.2.3 below; and (iii) Tenant shall not be entitled to any abatement of Basic Rent or any tenant
improvement allowance during the Option Term unless otherwise determined to be included in the Fair Market Rental Value in accordance with Section 3.2.3 below. If Tenant subleases any portion of the Premises or assigns or otherwise transfers any interest under this Lease (other than in connection with a Permitted Transfer), the Option shall lapse. If Tenant subleases any portion of the Premises or assigns or otherwise transfers any interest of Tenant under this Lease (other than in connection with a Permitted Transfer) to any person or entity after the exercise of the Option but prior to the commencement of the Option Term (whether with or without Landlord’s consent), the Option shall lapse and the Term of this Lease shall expire as if the Option was not exercised.
1.1.3.For the purposes hereof, “Fair Market Rental Value” of the Premises shall mean the prevailing annual market rental value (which rental value determination may include increases in Rent during the Option Term) for Class “A” office, manufacturing, and laboratory/research and development space of comparable size, quality and location in comparable first-class office and laboratory/research and development buildings located in the Sorrento Mesa submarket of San Diego, California, as of the date of commencement of the Option Term (“Comparable Transactions”), taking into consideration the amenities offered in or near the Project and the amount, availability and cost of parking; provided, however, that in calculating the Fair Market Rental Value, no consideration shall be given to (1) the fact that Landlord is or is not required to pay a real estate brokerage commission in connection with Tenant’s exercise of its right to lease the Premises during the Option Term or the fact that landlords are or are not paying real estate brokerage commissions in connection with such comparable space, and (2) any period of rental abatement, if any, granted to tenants in comparable transactions in connection with the design, permitting and construction of tenant improvements in such comparable spaces. The Fair Market Rental Value shall additionally include a determination as to whether, and if so to what extent, Tenant must provide Landlord with financial security, such as a letter of credit or security deposit, for Tenant’s Rent obligations in connection with Tenant’s lease of the Premises during the Option Term. Such determination shall be made by reviewing the extent of financial security then generally being imposed in Comparable Transactions from tenants of comparable financial condition and credit history to the then existing financial condition and credit history of Tenant (with appropriate adjustments to account for differences in the then-existing financial condition of Tenant and such other tenants).
1.1.4.Promptly after receiving Tenant’s notice of its election to exercise the Option to extend the Term of this Lease, Landlord shall provide Tenant with Landlord’s good faith estimate of the Fair Market Rental Value of the Premises for the Option Term (“Landlord’s Fair Market Rental Value Notice”). In the event that Tenant objects to Landlord’s determination of the Fair Market Rental Value within fifteen (15) business days following Tenant’s receipt of Landlord’s Fair Market Rental Value Notice, Landlord and Tenant shall attempt to agree upon the Fair Market Rental Value using their best good faith efforts. If Landlord and Tenant fail to reach agreement within ten (10) business days following Tenant’s objection to the Fair Market Rental Value (“Outside Agreement Date”), then each party shall make a separate determination of the Fair Market Rental Value within ten (10) business days after the Outside Agreement Date, and such determinations shall be submitted to arbitration in accordance with Subsection 3.2.4(A) through Subsection 3.2.4(G) below.
(A)Landlord and Tenant shall each appoint one arbitrator who shall be a real estate broker or attorney who shall have been active over the five (5) year period ending on the date of such appointment in the leasing of office and laboratory/research and development properties in the Sorrento Mesa submarket of San Diego, California. The determination of the arbitrators shall be limited solely to the issue of whether Landlord’s or Tenant’s submitted Fair Market Rental Value is the closest to the actual Fair Market Rental Value, as determined by the arbitrators, taking into account the requirements of Subsection 3.2.3. Each such arbitrator shall be appointed within fifteen (15) days after the applicable Outside Agreement Date.
(B)The two (2) arbitrators so appointed shall within ten (10) days of the date of the appointment of the last appointed arbitrator agree upon and appoint a third arbitrator who shall be qualified under the same criteria set forth hereinabove for qualification of the initial two (2) arbitrators.
(C)The three (3) arbitrators shall within thirty (30) days of the appointment of the third arbitrator reach a decision as to whether the parties shall use Landlord’s or Tenant’s submitted Fair Market Rental Value and shall notify Landlord and Tenant thereof.
(D)The decision of the majority of the three (3) arbitrators shall be binding upon Landlord and Tenant.
(E)If either Landlord or Tenant fails to appoint an arbitrator within fifteen (15) days after the applicable Outside Agreement Date, then the arbitrator appointed by one of them shall reach a decision, notify Landlord and Tenant thereof and such arbitrator’s decision shall be binding upon Landlord and Tenant.
(F)If the two (2) arbitrators fail to agree upon and appoint a third arbitrator, or if both parties fail to appoint an arbitrator, then the appointment of the third arbitrator or any arbitrator shall be dismissed and the matter to be decided shall be forthwith submitted to arbitration under the provisions of the American Arbitration Association, but subject to the instruction set forth in this Subsection 3.2.4.
(G)The cost of the arbitration shall be paid by Landlord and Tenant equally.
Article 4
DELIVERY
Landlord will endeavor to tender possession of the applicable Premises to Tenant with the applicable Tenant Improvements Substantially Complete on or before the applicable Estimated Commencement Date; provided, that if the date on which Landlord actually tenders possession of the Premises to Tenant in such condition does not occur on or before the Estimated Commencement Date, this Lease shall not be void or voidable, the Term of this Lease shall not be extended, and Landlord shall not be liable to Tenant for any loss or damage resulting therefrom; provided further that Landlord shall use commercially reasonable efforts to tender to Tenant delivery of possession of the applicable Premises in such condition as soon as reasonably possible after the applicable Estimated Commencement Date. Notwithstanding the foregoing, the Term and Tenant’s obligation to pay Base Rent, Operating Expenses and Real Property Taxes for the applicable Premises shall not begin until the actual Commencement Date for such Premises.
Notwithstanding the foregoing, if Landlord is unable to deliver possession of Premises A to Tenant with the Landlord Work and Tenant Improvements therein Substantially Complete on or before the date which is sixty (60) days after the Estimated Commencement Date for Premises A (“First Outside Delivery Date”), which First Outside Delivery Date shall be extended on a day-for-day basis for any delay caused by an event of Force Majeure (as defined in Section 36.8 below), a Tenant Delay (as that term is defined in the Work Letter Agreement) or similar matters beyond the reasonable control of Landlord, Tenant shall be entitled to receive an abatement of Basic Rent for Premises A on a day-for-day basis for each day between the First Outside Delivery Date and the actual date on which possession of Premises A is delivered to Tenant with the Landlord Work and Tenant Improvements therein Substantially Complete (“Actual Delivery Date for Premises A”), to be applied as a credit toward the Basic Rent next due and payable under this Lease (for example, if the Estimated Commencement Date for Premises A is February 1, 2022, and the Actual Delivery Date for Premises A is February 5, 2022, then Tenant shall receive a credit toward Basic Rent in an amount equal to four (4) days of Basic Rent for Premises A). Notwithstanding the foregoing, if Landlord is unable to deliver possession of Premises A to Tenant with the Landlord Work and Tenant Improvements therein Substantially Complete on or before the date which is one hundred eighty (180) days after the Estimated Commencement Date for Premises A (“Second Outside Delivery Date”), which Second Outside Delivery Date shall be extended on a day-for-day basis for any delay caused by an event of Force Majeure, a Tenant Delay or similar matters beyond the reasonable control of Landlord, then Tenant shall have the right to terminate this Lease by delivering Notice thereof to Landlord no later than five (5) business days after the Second Outside Delivery Date, which termination shall be effective as of the date of such Notice. Tenant’s failure to deliver a Notice of termination within five (5) business days after the Second Outside Delivery Date shall be deemed Tenant’s waiver of its right to terminate this Lease due to a delay in delivery of the Premises.
If Landlord is unable to deliver possession of Premises B to Tenant with the Landlord Work and Tenant Improvements therein Substantially Complete on or before the date which is sixty (60) days after the Estimated Commencement Date for Premises B (“Premises B Outside Delivery Date”), which Premises B Outside Delivery Date shall be extended on a day-for-day basis for any delay caused by an event of Force Majeure, a Tenant Delay or similar matters beyond the reasonable control of Landlord, Tenant shall be entitled to receive an abatement of Basic Rent for Premises B on a day-for-day basis for each day between the Premises B Outside Delivery Date and the actual date on which possession of Premises B is delivered to Tenant with the Landlord Work and Tenant Improvements therein Substantially Complete (“Actual Delivery Date for Premises B”), to be applied as a credit toward the Basic Rent next due and payable under this Lease (for example, if the Estimated Commencement Date for Premises B is June 1, 2022, and the Actual Delivery Date for Premises B is June 5, 2022, then Tenant shall receive a credit toward Basic Rent in an amount equal to four (4) days of Basic Rent for Premises B). Notwithstanding the foregoing, if Landlord is unable to commence the Landlord Work on Premises on or prior to August 1, 2022 (the “Premises B Work Commencement Date”) or (ii) deliver possession of Premises B to Tenant with the Landlord Work and Tenant Improvements therein Substantially Complete on or before the date which is one hundred eighty (180) days after the Estimated Commencement Date for Premises B (“Premises B Second Outside Delivery Date”), which Premises B Work Commencement Date and Premises B Second Outside Delivery Date shall be extended on a day-for-day basis for any delay caused by the existing tenant of Premises B failing to vacate Premises B in a timely manner, an event of Force Majeure, a Tenant Delay or similar matters beyond the reasonable control of Landlord, then Tenant shall have the right to terminate this Lease with respect to Premises B only by delivering Notice thereof to Landlord no later than five (5) business days after the Premises B Work Commencement Date or the Premises B Second Outside Delivery Date (as applicable), which termination shall be effective as of the date of such Notice. Tenant’s failure to deliver a Notice of termination within five (5) business days after the applicable date shall be deemed Tenant’s waiver of its right to terminate this Lease with respect to Premises B due to a delay in delivery of the Premises. The remedies set forth in this Article 4 shall be Tenant’s sole and exclusive remedies at law or equity for the matters described herein.
Article 5
RENT
1.1Basic Rent. Tenant shall pay Landlord as consideration for the use and enjoyment of the Premises the Basic Rent designated in Section 1.12 (subject to proration as hereinafter provided) in equal monthly installments, each in advance on the first day of each calendar month during the Term commencing on the applicable Commencement Date, except that the first month’s Rent for both Premises shall be paid to Landlord upon delivery to Landlord of a copy of this Lease, executed by Tenant. If the Term of this Lease commences on a day other than the first day of a calendar month or ends on a day other than the last day of a calendar month, then the Rent for such period shall be prorated on the basis of a thirty (30) day month. Provided that Tenant is not then in default under this Lease beyond any applicable notice and cure period, monthly installments of Basic Rent for each Premises shall be abated for six (6) full months (months two (2) through seven (7) of the Initial Term for each applicable Premises) (each an “Abatement Period”). All other terms and provisions of this Lease shall apply to the Premises both during the Abatement Period and thereafter, including, without limitation, the obligation to pay all Additional Rent (defined below).
1.2Additional Rent. In addition to the Basic Rent, Tenant agrees to pay as Additional Rent the amount of Rent adjustments and other charges required by this Lease. Other charges to be paid by Tenant hereunder, including, without limitation, payments for Operating Expenses, Real Property Taxes, insurance, insurance deductibles and repairs shall be considered “Additional Rent” for purposes of this Lease. The term “Rent” as used in this Lease shall mean Basic Rent and Additional Rent and all other amounts payable by Tenant pursuant to this Lease. When no other time is stated herein for payment, payment of any amount due from Tenant to Landlord hereunder shall be made within ten (10) business days after Tenant’s receipt of Landlord’s invoice or statement therefor. All Rent shall be paid to Landlord, without prior demand and without any deduction or offset except as specified herein, in lawful money of the United States of America, at the address designated in Section 1.2 hereof or to such other person or at such other place as Landlord may from time to time designate in writing.
1.3Late Payment. If Tenant fails to pay any installment of Rent when due or in the event Tenant fails to make any other payment for which Tenant is obligated under this Lease when due, such late amount shall accrue interest and Tenant shall pay Landlord as Additional Rent interest on such amount at an annual rate (“Default Rate”) equal to the lesser of: (a) the then prevailing prime rate of Bank of America NT & SA (“Prime Rate”) plus six (6) percentage points or (b) the maximum rate permitted by law from the date such amount became due until such amount is paid. If the format or components of the Prime Rate are materially changed, or if the Prime Rate ceases to exist, Landlord shall substitute a prime rate or alternative base rate of interest that is maintained by the Bank of America NT & SA or similar financial institution which Landlord determines in its reasonable business judgment. In addition to said interest, Tenant shall pay to Landlord concurrently with any installment of Rent, or other payment, not paid within five (5) days of the date upon which it is due, and Landlord may demand same from Tenant, as Additional Rent, a late charge equal to eight percent (8%) of the late amount to compensate Landlord for the extra costs incurred as a result of such late payment. THE PARTIES AGREE THAT ANY SUCH LATE PAYMENT MAY CAUSE LANDLORD TO INCUR ADMINISTRATIVE COSTS AND OTHER DAMAGE, THE EXACT AMOUNT OF WHICH WOULD BE IMPRACTICABLE OR EXTREMELY DIFFICULT TO ASCERTAIN, AND THAT SUCH INTEREST AND LATE CHARGE REPRESENT A FAIR AND REASONABLE ESTIMATE OF THE DETRIMENT THAT LANDLORD WILL SUFFER BY REASON OF LATE PAYMENT BY TENANT. Acceptance of any such interest and late charge shall not constitute a waiver of any Tenant Default with respect to the overdue amount, or prevent Landlord from exercising any of the other rights and remedies available to Landlord hereunder or at law.
1.4Additional Late Payment Remedies. If any payment of Rent made by check, draft or money order is returned to Landlord due to insufficient funds, or otherwise, Landlord shall have the right, at any time thereafter and upon Notice (defined below) to Tenant, to require Tenant to make all subsequent payments of Rent by cashier’s or certified check. Any payment returned to Landlord shall be subject to a handling charge of $50.00. If Tenant fails to pay an installment of Basic Rent within ten (10) days following the date the same is due on any three (3) or more occasions during any twelve (12) month period, Landlord shall have the right, in addition to any other rights or remedies it may have hereunder or at law, to require Tenant thereafter to pay installments of Basic Rent quarterly in advance.
Article 6
RENT ADJUSTMENT
1.1Definitions. For the purposes of this Lease, the following terms shall be defined as follows:
1.1.1.Operating Expenses: “Operating Expenses” shall consist of all costs of operation, management, ownership, insurance, maintenance and repair of the Project, including without limitation the Buildings, the Common Areas and all other portions of the Project. Operating Expenses shall include, without
limitation, the following: (a) any and all non-tax assessments payable by Landlord for, or costs or expenses incurred by Landlord in connection with, the Buildings or the Project pursuant to any covenants, conditions or restrictions, reciprocal easement agreements, tenancy-in-common agreements or similar restrictions and agreements affecting the Buildings or the Project; (b) assessments and any taxes or assessments hereafter imposed in lieu thereof; (c) Rent taxes and gross receipts taxes (whether assessed against Landlord or assessed against Tenant and paid by Landlord, or both); (d) water and sewer charges; (e) accounting, legal and other consulting fees incurred by Landlord in connection with the Project or any portion thereof; (f) the net cost and expense of insurance, and any associated insurance deductibles, for which Landlord and/or the owner(s) and/or the operator(s) of the Project is (are) responsible or any first mortgagee with a lien affecting the Premises reasonably deems necessary in connection with the operation of the Buildings or the Project; (g) utilities, including, but not limited to, any and all costs and fees associated with the installation, maintenance, repair, or replacement of intrabuilding network telephone and data cable; (h) janitorial services, security, labor, utilities surcharges or any other costs levied, assessed or imposed by, or at the direction of, or resulting from statutes, including, but not limited to, the Americans with Disabilities Act (42 U.S.C. Section 12101 et seq.), or regulations or interpretations thereof promulgated by, any federal, state, regional, local or municipal governmental authority, agency or subdivision (each, a “Governmental Authority”) in connection with the use or occupancy of the Project or any portion thereof; (i) costs and expenses incurred or suffered by Landlord in connection with transportation or energy management programs; (j) the cost (amortized over such period as is customary under sound institutional real estate property management procedures (“Institutional Owner Practices”), together with interest at a rate (“Interest Rate”) equal to the Prime Rate plus two (2) percentage points on the enumerated balance): (i) of any capital improvements or replacements intended as labor-saving devices or to effect other economies in the maintenance or operation of, or stability of services to, the Buildings (including Building Common Areas) or the Project Common Areas by Landlord or by the owner(s) and/or the operator(s) thereof, or (ii) of replacing any equipment, systems or materials needed to operate the Project or any portion thereof at the same quality levels as prior to the improvement or replacement or as mandated by revisions or governmental interpretations of any applicable Laws (defined below) or (iii) which are designed to reduce Operating Expenses or to comply with Laws; (k) costs incurred in the management of the Project, including supplies, materials, equipment, on-site management office rent, wages and salaries of employees used in the management, operation and maintenance thereof, payroll taxes and similar governmental charges with respect thereto, and a Project management fee (not to exceed three percent (3%) of gross receipts, grossed up to reflect ninety-five percent (95%) occupancy if the Project is less than ninety-five percent (95%) occupied; provided, however, that for each calendar year after the first full calendar year of the Initial Term, such management fee shall not increase by more than six percent (6%) over the maximum permitted management fee for the immediately preceding calendar year, on a cumulative, compounding basis and regardless of the actual gross receipts for such preceding year; provided, however, that if the actual gross receipts and corresponding management fee for any calendar year are greater than the maximum amount permitted to be charged to Tenant hereunder, then the difference shall be added to the management fee for succeeding years of the Term until exhausted); (l) all costs and expenses for air-conditioning, waste disposal, heating, ventilating, elevator repair and maintenance, supplies, materials, equipment, and tools incurred in connection with the Project or any portion thereof (except as the same is payable to Landlord by tenants of the Project under their leases for space in the Project); (m) repair and maintenance of the roof and structural portions of the Buildings and the Common Areas, including the plumbing, heating, ventilating, air conditioning and electrical systems installed or furnished by Landlord; (n) maintenance costs of the Buildings, the Common Areas and the Project or any portion thereof, including utilities and payroll expenses, rent of personal property used in maintenance and all other upkeep; (o) costs and expenses of gardening and landscaping the Project or any portion thereof; (p) maintenance of signs located in or about the Project (other than Tenant’s signs or the signs of other tenants or occupants of the Building who are responsible to maintain their own signs); (q) personal property taxes levied on or attributable to personal property of Landlord or the owner(s) and/or operator(s) of the Project used in connection with the Project; (r) reasonable audit or verification fees incurred in connection with the Project; and (s) the costs and expenses of repairs (including latent defects), resurfacing, maintenance, painting, lighting, cleaning, refuse removal, security and similar items incurred with respect to the Project, including appropriate reserves.
Operating Expenses shall not include: (A) depreciation on the Buildings or equipment therein; (B) Landlord’s executive salaries (above building manager); (C) real estate broker’s commissions; (D) legal fees and disbursements incurred for collection of tenant accounts or negotiation of leases, or relating to disputes between Landlord and other tenants and occupants of the Project; (E) the cost of any capital improvements unless specifically permitted by this Section 6.1.1, parts (a) through (s), inclusive; (F) amounts received by Landlord on account of proceeds of insurance to the extent the proceeds are reimbursement for expenses which were previously included in Operating Expenses; (G) payments of principal and interest on any mortgages upon the Project or Buildings; (H) payments of ground rent pursuant to any ground lease covering the Project or Buildings; (I) the costs of gas, steam or other fuel; operation of elevators and security systems; heating, cooling, air conditioning and ventilating; chilled water, hot and cold domestic water, sewer and other utilities or any other service work or facility, or level or amount thereof, provided to any other tenant or occupant in the Project which either (x) is not required to be supplied or furnished by Landlord to Tenant under the provisions of this Lease or (y) is supplied or furnished to Tenant pursuant to the terms of this Lease with separate or additional charge; (J) the cost of providing any service directly to and paid directly by a single individual lessee, or costs incurred for the benefit of a single lessee; (K) costs of any items to the extent Landlord actually receives reimbursement therefor from insurance proceeds, under warranties, or from a
lessee or other third party, or which are paid out of reserves previously included in Operating Expenses; (L) costs incurred due to Landlord’s breach of a law or ordinance; (M) repairs necessitated by the gross negligence or willful misconduct of Landlord or Landlord’s employees, agents, or contractors; (N) Landlord’s costs of any services provided to lessees or other occupants for which Landlord is actually reimbursed by such lessees or other occupants as an additional charge or rental over and above the basic rent (and escalations thereof) payable under the lease with such lessee or other occupant; (O) costs in connection with services that are provided to another lessee or occupant of the Project, but are not offered to Tenant; (P) salaries of employees of Landlord above those performing property management and facilities management duties for the Project; (Q) costs or expenses incurred in connection with the financing or sale of the Project or any portion thereof; (R) costs of environmental testing, monitoring, removal or remediation of any Hazardous Materials in the Project that are in existence at the Project prior to the Commencement Date except to the extent caused or exacerbated by Tenant; (S) the costs of acquiring investment-grade art; (T) fines, penalties, interest or other amounts imposed in connection with the Landlord’s failure to pay any tax when due; (U) any item that, if included in Operating Expense, would involve a double collection for such item by Landlord; and (V) any cost that is expressly excluded from Operating Expenses in an express provision contained in this Lease.
1.1.2.Real Property Taxes: “Real Property Taxes” shall mean and include any form of assessment, re-assessment, license fee, license tax, business license fee, commercial rent tax, levy, charge, penalty, tax or similar imposition, imposed by any authority having the direct power to tax, including any Governmental Authority, or any school, agricultural, lighting, drainage or other improvement or special assessment district thereof, as against any legal or equitable interest of Landlord in the Buildings, the Premises or the Project, including but not limited to the following:
(A)any tax on Landlord’s “right” to other income from the Project or any portion thereof or as against Landlord’s business of leasing the Project or any portion thereof;
(B)any assessment, tax, fee, levy or charge in substitution, partially or totally, of any assessment, tax, fee, levy or charge previously included within the definition of real estate tax, including but not limited to, any assessments, taxes, fees, levies and charges that may be imposed by any Governmental Authority for such services as fire protection, street, sidewalk or road maintenance, refuse removal and for other governmental services formerly provided without charge to property owners or occupants, it being the intention of Tenant and Landlord that all such new and increased assessments, taxes, fees, levies and charges be included within the definition of “Real Property Taxes” for the purposes of this Lease;
(C)any assessment, tax, fee, levy or charge allocable to or measured by the area of any premises in the Project or the Rent payable hereunder and under any other leases for premises in the Buildings, the Parking Area or the Project, including without limitation any gross income tax or excise tax levied by any Governmental Authority or any political subdivision thereof, with respect to the receipt of such Rent, or upon or with respect to the possession, leasing, operating, management, maintenance, alteration, repair, use or occupancy by tenants of their premises in the Project, or any portion thereof; and
(D)any assessment, tax, fee, levy or charge upon this transaction or any document creating or transferring an interest or an estate in the Project or any portion thereof, or based upon a reassessment of the Project or any portion thereof by virtue of a “change in ownership”, and as a result thereof, and to the extent that in connection therewith, the Buildings are reassessed for real estate tax purposes by the appropriate Governmental Authority pursuant to the terms of Proposition 13 (as adopted by the voters of the State of California in the June, 1978 election, or any successor statute).
Notwithstanding any provision of this Section 6.1.2 expressed or implied to the contrary, “Real Property Taxes” shall not include Landlord’s federal or state income, franchise, inheritance or estate taxes or any fines, penalties or interest arising out of a failure by Landlord to pay any Real Property Taxes when due.
1.1.3.Tenant’s Percentage. “Tenant’s Percentage” means the percentage set forth in Section 1.13.
1.2Calculation Methods and Adjustments.
1.2.1.Subject to the provisions of this Section 6.2, all calculations, determinations, allocations and decisions to be made hereunder with respect to Operating Expenses and Real Property Taxes shall be made on a triple net basis in accordance with the good faith determination of Landlord applying sound accounting and property management principles consistently applied which are consistent with Institutional Owner Practices. Landlord shall have the right to equitably allocate some or all Operating Expenses among particular classes or groups of tenants in the Project or Buildings (for example, retail tenants) to reflect Landlord’s good faith determination that measurably different amounts or types of services, work or benefits associated with Operating Expenses, as applicable, are being provided to or conferred upon such classes or groups. All discounts, reimbursements, rebates, refunds, or credits (collectively, “Reimbursements”) attributable to Operating Expenses or Real Property Taxes received by Landlord
in a particular year shall be deducted from Operating Expenses or Real Property Taxes, as applicable, in the year the same are received; provided, however, if such practice is consistent with Institutional Owner Practices, Landlord may treat Reimbursements generally (or under particular circumstances) on a different basis.
1.2.2.As of the date of this Lease, Tenant shall pay Additional Rent under this Article 6 based on the Operating Expenses and Real Property Taxes for the Project. If the Project at any time contains more than one building, Landlord shall have the right, from time to time, to equitably allocate some or all of the Operating Expenses and/or Real Property Taxes for the buildings comprising the Project among the Buildings and some or all of the other buildings of the Project. In such event, Landlord shall reasonably determine a method of allocating such Operating Expenses and/or Real Property Taxes attributable to the Building and/or such other building(s) of the Project to the Building and/or such other building(s) and Tenant shall be responsible for paying its proportionate share of such expense(s) which are allocated to the Building. Landlord shall also have the right, from time to time, to require Tenant to pay Tenant’s Percentage of Operating Expenses and Real Property Taxes based solely on the Operating Expenses and Real Property Taxes for the Building.
1.2.3.Notwithstanding any provision of this Lease to the contrary, for the sole purpose of calculating Tenant’s Percentage of Operating Expenses, the Controllable Operating Expenses (as defined below) for each calendar year after the first full calendar year of the Term following the Premises B Commencement Date shall not increase by more than six percent (6%) over the maximum permitted Controllable Operating Expenses for the immediately preceding calendar year (i.e., on a cumulative, compounding basis and regardless of the actual Controllable Operating Expenses incurred for such preceding year); provided, however, that if the actual Controllable Operating Expenses for any calendar year are greater than the maximum amount permitted to be charged to Tenant hereunder, then the difference shall be added to the actual Controllable Operating Expenses for succeeding years of the Term until exhausted. The term “Controllable Operating Expenses” mean those Operating Expenses for which increases are reasonably within the control of Landlord, and shall specifically exclude, without limitation, Utility Expenses (as defined below), Insurance Expenses (as defined below), Real Property Taxes, assessments, the cost of any Project amenities, collectively bargained union wages, costs to comply with governmental laws enacted or first applicable after the Commencement Date, trash removal and any services provided by monopolies or where there is otherwise only one (1) provider available to Landlord. For purposes of this Lease, the following definitions shall apply: (i) “Utility Expenses” shall mean the cost of supplying all utilities to the Project; and (ii) “Insurance Expenses” shall mean the cost of insurance required or allowed to be carried by Landlord under this Lease. There shall be no limitation on the amount of increase from year to year on Operating Expenses which are not Controllable Operating Expenses.
1.3Payment of Tenant’s Percentage of Operating Expenses and Real Property Taxes. This shall be a triple net Lease and Basic Rent shall be paid to Landlord absolutely net of all costs and expenses, except as specifically provided to the contrary in this Lease. The provisions for payment of Tenant’s Percentage of Operating Expenses and Tenant’s Percentage of Real Property Taxes are intended to pass on to Tenant, and reimburse Landlord for, all costs and expenses of the nature described in Section 6.1 incurred in connection with the ownership, operation, management, insurance, maintenance and repair of the Project. For each calendar year of the Term, Tenant shall pay Tenant’s Percentage of the Operating Expenses and Tenant’s Percentage of the Real Property Taxes paid or incurred by Landlord for such year as Additional Rent. Tenant shall pay such amounts as follows:
1.3.1.Estimate of Annual Operating Expenses and Real Property Taxes. At the beginning of each calendar year, or as soon thereafter as practicable, Landlord shall deliver to Tenant a reasonable estimate (“Estimated Statement”) of Tenant's Percentage of Operating Expenses and Tenant's Percentage of Real Property Taxes for the then current calendar year. Landlord may revise its estimates of Tenant's Percentage of Operating Expenses and Tenant's Percentage of Real Property Taxes for any year from time to time in its reasonable discretion, and upon receipt of a revised Estimated Statement, Tenant shall begin making payments under this Section 6.3.1 in accordance with such revised estimates. For each calendar year during the Term of this Lease, or portion thereof, Tenant shall pay to Landlord the estimated Tenant’s Percentage of Operating Expenses and the estimated Tenant's Percentage of Real Property Taxes, as specified in the Estimated Statement. These estimated amounts shall be divided into twelve (12) equal monthly installments. Tenant shall pay to Landlord, concurrently with the regular monthly Basic Rent payment next due following the receipt of such an Estimated Statement, an amount equal to one monthly installment multiplied by the number of months from the commencement of the calendar year for which such estimates were prepared to the month of such payment, both months inclusive, less any amounts paid under this Section 6.3.1 after commencement of such calendar year based on the last Estimated Statement delivered by Landlord. Subsequent payments under this Section 6.3.1 shall be payable concurrently with the regular monthly Rent payments for the balance of that calendar year and shall continue until the next Estimated Statement is delivered by Landlord. Failure of Landlord to deliver an Estimated Statement for any calendar year shall not relieve Tenant of its obligation to make estimated payments of Tenant's Percentage of Operating Expenses and Tenant's Percentage of Real Property Taxes under this Section 6.3.1.
1.3.2.Annual Reconciliation. At the end of each calendar year or as soon thereafter as practicable Landlord shall deliver to Tenant a statement (“Annual Reconciliation”) of (a) the actual annual Operating Expenses and Tenant’s Percentage of Operating Expenses for the preceding year, and (b) the actual annual Real Property Taxes and Tenant’s Percentage of Real Property Taxes for the preceding year. If for any year, the sum of Tenant’s Percentage of Operating Expenses and Tenant’s Percentage of Real Property Taxes (as specified in the Annual Reconciliation) is less than the total amount of the estimated payments made by Tenant under Section 6.3.1 above for such year, then any such overpayment, or overpayments, shall be credited toward the monthly Rent next falling due after determination by Landlord of such overpayment or overpayments and shall be paid to Tenant in a lump sum for periods after the expiration of the Term. Similarly, if for any year, the sum of Tenant’s Percentage of Operating Expenses and Tenant’s Percentage of Real Property Taxes (as specified in the Annual Reconciliation) is more than the total amount of the estimated payments made by Tenant under Section 6.3.1 above for such year, then any such underpayment, or underpayments, shall be paid by Tenant to Landlord concurrently with the next regular monthly Basic Rent payment coming due after Tenant’s receipt of the Annual Reconciliation (or if the Term shall have expired or terminated, within thirty (30) days following Tenant’s receipt of such Annual Reconciliation).
1.3.3.Survival of Reconciliation. Even though the Term shall have expired and Tenant shall have vacated the Premises, when the final determination of Tenant’s Percentage of actual annual Operating Expenses, and/or of Tenant’s Percentage of actual annual Real Property Taxes, for the year in which this Lease terminates is delivered to Tenant, (a) Tenant shall immediately pay any amounts payable to Landlord under Section 6.3.2 above (as a result of any underpayments by Tenant under Section 6.3.1 above), and/or (b) conversely, Landlord shall promptly rebate any amounts payable to Tenant under Section 6.3.2 (as a result of any overpayments under Section 6.3.1 above) provided that no Tenant Default existed at the expiration or earlier termination of this Lease.
1.4Review of Annual Reconciliation. Provided that Tenant is not then in default with respect to its obligations under this Lease and provided further that Tenant strictly complies with the provisions of this Section 6.4, Tenant shall have the right, at Tenant’s sole cost and expense and upon thirty (30) days prior Notice (“Review Notice”) to Landlord delivered no later than sixty (60) days after an Annual Reconciliation is delivered to Tenant, to reasonably review or audit Landlord’s supporting books and records (at Landlord’s manager’s corporate offices) for any portion of the Operating Expenses or Real Property Taxes for the particular year covered by such Annual Reconciliation, in accordance with the procedures set forth in this Section 6.4. To the extent that any amounts specified in such Annual Reconciliation were not previously paid, Tenant shall pay all such amounts to Landlord simultaneously with Tenant’s delivery the Review Notice. Except as expressly provided herein, any review or audit of records under this Section 6.4 shall be at the sole expense of Tenant, shall be conducted by independent certified public accountants of national standing which are not compensated on a contingency fee or similar basis relating to the results of such review or audit and shall be completed within sixty (60) days after Landlord provides Tenant with access to Landlord’s supporting books and records. Tenant shall, within thirty (30) days after completion of any such review or audit, deliver Notice to Landlord specifying the items described in the Annual Reconciliation that are claimed to be incorrect by such review or audit (“Dispute Notice”). The right of Tenant under this Section 6.4 may only be exercised once for each year covered by any Annual Reconciliation, and if Tenant fails to deliver a Review Notice within the sixty (60) day period described above or a Dispute Notice within the thirty (30) day period described above, or if Tenant fails to meet any of the other above conditions of exercise of such right, the right of Tenant to review or audit a particular Annual Reconciliation (and all of Tenant’s rights to make any claim relating thereto) under this Section 6.4 shall automatically be deemed waived by Tenant. Tenant acknowledges and agrees that any records of Landlord reviewed or audited under this Section 6.4 (and the information contained therein) constitute confidential information of Landlord, which shall not be disclosed other than to Tenant’s accountants performing the review or audit and principals of Tenant who receive the results of the review or audit. If Landlord disagrees with Tenant’s contention that an error exists with respect to the Annual Reconciliation in dispute, Landlord shall have the right to cause another review or audit of that portion of the Annual Reconciliation to be made by a firm of independent certified public accountants of national standing selected by Landlord (“Landlord’s Accountant”). In the event of a disagreement between the two accounting firms, the review or audit of Landlord’s Accountant shall be deemed to be correct and shall be conclusively binding on both Landlord and Tenant. In the event that it is finally determined pursuant to this Section 6.4 that a particular Annual Reconciliation overstated amounts payable by Tenant under this Article 6 with respect to the applicable year by more than five percent (5%), Landlord shall reimburse Tenant for the reasonable costs of Tenant’s accountant and Landlord shall be liable for the costs of Landlord’s Accountant. In all other cases, Tenant shall reimburse Landlord for the reasonable costs of Landlord’s Accountant.
Article 7
SECURITY DEPOSIT
1.1Security Deposit. Tenant shall deposit with Landlord, upon delivery to Landlord of a copy of this Lease executed by Tenant, the Security Deposit designated in Section 1.14. Said sum shall be held by Landlord as security for the full and faithful performance by Tenant of all of the terms, covenants and conditions of this Lease to
be kept and performed by Tenant during the Term hereof and any extension hereof. Landlord shall have the right, but not the obligation, to apply all or any portion of the Security Deposit for the payment of Rent or any other sum due hereunder, to cure any default by Tenant of its obligations with respect to the restoration and surrender of the Premises or to cure any Tenant Default at any time, in which event Tenant shall be obligated to restore the Security Deposit to its original amount within ten (10) business days, and Tenant’s failure to do so shall be deemed to be a Default under this Lease. Tenant hereby irrevocably waives and relinquishes any and all rights, benefits, or protections, if any, Tenant now has, or in the future may have, under Section 1950.7 of the California Civil Code, any successor statute, and all other provisions of law, now or hereafter in effect, including, but not limited to, any provision of law which: (a) establishes the time frame by which a landlord must refund a security deposit under a lease, or (b) provides that a landlord may claim from a security deposit only those sums reasonably necessary to remedy defaults in the payment of Rent, to repair damage caused by a tenant, or to clean the subject premises. Tenant acknowledges and agrees that: (i) any statutory time frames for the return of a security deposit are superseded by the express period identified in this Section 7.1 and (ii) rather than be so limited, Landlord may claim from the Security Deposit: (A) any and all sums expressly identified in this Section 7.1, and (B) any additional sums reasonably necessary to compensate Landlord for any and all losses or damages caused by Tenant’s default of this Lease, including, but not limited to, all damages or Rent due upon termination of this Lease pursuant to Section 1951.2 of the California Civil Code, as amended and recodified from time to time. Landlord shall not be required to keep the Security Deposit separate from its general funds, and Tenant shall not be entitled to interest on such Security Deposit. Within thirty (30) days following the expiration of the Term, Landlord shall (provided that Tenant is not in Default under this Lease) return the Security Deposit to Tenant, less such portion as Landlord shall have applied in accordance with this Section 7.1. Should Landlord sell its interest in the Premises during the Term hereof and if Landlord deposits with (or gives a credit to) the purchaser thereof the then balance of the Security Deposit held by Landlord, Landlord shall be released from any further liability with respect to the Security Deposit.
1.2Lease Guaranty. As a condition precedent to the effectiveness of this Lease, Tenant shall deliver to Landlord a Lease Guaranty in the form of Exhibit “D” executed by Guarantor concurrently with the execution and delivery of this Lease.
Article 8
USE
1.1General. Tenant shall use the Premises for the Permitted Use set forth in Section 1.16 above, and shall not use or permit the Premises to be used for any other purpose without the prior written consent of Landlord. Nothing contained herein shall be deemed to give Tenant any exclusive right to such use in the Project or any portion thereof (excluding only the Premises).
1.2Laws/CC&R’s.
1.2.1.Tenant shall not use or occupy the Premises in violation of any applicable laws, regulations, rules, orders, statutes or ordinances of any Governmental Authority, office, board or private entity in effect on or after the Effective Date and applicable to the Project or the use or occupancy of the Project, including, without limitation, the rules, regulations and requirements of the Pacific Fire Rating Bureau, and of any similar body, the Americans with Disabilities Act (42 U.S.C. Section 12101 et seq.) (“ADA”) and Hazardous Material Laws (as defined in Section 8.3.7 below) (collectively, “Laws”) or in violation of any government-issued permit for the Buildings or Project or any of the Rules and Regulations (as defined below), and shall, upon Notice from Landlord, discontinue any use of the Premises which is declared by any Governmental Authority having jurisdiction to be a violation of any Laws, or of any government-issued permit for the Buildings or Project. On or before the applicable Commencement Date, Landlord shall cause the Buildings and the Premises to comply with all applicable Laws (including, but not limited to, any obligation imposed pursuant to the ADA). Except as expressly set forth in the immediately preceding sentence, Tenant shall cause the Premises to comply with all applicable Laws and shall comply with any direction of any Governmental Authority having jurisdiction which shall, by reason of the nature of Tenant’s use or occupancy of the Premises, impose any obligation (including, but not limited to, any obligation imposed pursuant to the ADA as a result of Tenant’s particular use of the Premises), upon Tenant or Landlord with respect to the Premises or with respect to the use or occupancy thereof; provided, however, unless resulting from an Alteration performed by Tenant or by Tenant’s specific use of the Premises (as opposed to general office and laboratory/research and development use), Tenant shall not be responsible for any obligation imposed by the ADA after completion of the initial Tenant Improvements with respect to the Common Areas (except its prorata share of compliance costs included in Operating Expenses). Tenant shall comply with all rules, orders, regulations and requirements of the Pacific Fire Rating Bureau or any other organization performing a similar function. Tenant shall not do or permit to be done in or about the Premises anything which causes the insurance on the Premises, the Buildings or the Project or any portion thereof to be canceled or the cost thereof increased. Tenant shall promptly, upon demand, reimburse Landlord for any additional premium charged for any insurance policy by reason of Tenant’s failure to comply with the provisions of this Section 8.2. In determining whether increased premiums are a result of Tenant’s use of the Premises, a schedule issued by the organization computing the insurance rate on the
Project or the Tenant Improvements showing the various components of such rate shall be conclusive evidence of the several items and charges which make up such rate. Tenant shall promptly comply with all reasonable requirements of the insurance authority or any present or future insurer relating to the Premises. Tenant shall not do or permit anything to be done in or about the Premises which will in any way obstruct or interfere with the rights of Landlord or other tenants or occupants of the Buildings, the Parking Area or the Project, or injure or unreasonably annoy them, or use or allow the Premises to be used for any improper, immoral, unlawful or objectionable purpose, nor shall Tenant cause, maintain or permit any legal nuisance in or about the Premises or the Project. Tenant shall comply with all restrictive covenants and obligations created by private contracts that affect the use and operation of the Premises, the Buildings, the Common Areas or any other portion of the Project. Tenant shall not commit or suffer to be committed any waste in or upon the Premises or the Project and shall keep the Premises in first class repair and appearance. If any governmental license or permit shall be required for the proper and lawful conduct of Tenant’s business in the Premises, Tenant, at its expense, shall procure, maintain and comply with the terms and conditions of each such license or permit.
Without limiting the generality of the foregoing:
(A)Landlord and Tenant agree to cooperate, and Tenant shall use its commercially reasonable efforts to participate in governmentally mandated regulations or voluntary traffic management programs applicable to businesses located in the area or to the Project. Neither this Section 8.2.1(A) nor any other provision of this Lease, however, is intended to or shall create any rights or benefits in any other person, firm, company, Governmental Authority or the public. Upon Tenant’s failure to comply with this Section 8.2.1(A), Landlord may suspend Tenant’s parking privileges in addition to taking such other remedies as may be available to a landlord against a defaulting tenant.
(B)Landlord and Tenant agree to cooperate and comply with any and all guidelines or controls imposed upon either Landlord or Tenant by any Governmental Authority or by any energy conservation association to which Landlord is a party concerning energy management
(C)All costs, fees, assessments and other charges paid by Landlord to any Governmental Authority or voluntary association in connection with any program of the types described in Sections 8.2.1(A) and 8.2.1(B) above, and all costs and fees paid by Landlord to any Governmental Authority or third party pursuant to or to effect such program, shall be included in Operating Expenses for the purposes of Article 6, whether or not specifically listed in such Article 6.
(D)Tenant shall be liable for all penalties, noncompliance costs or other losses, costs or expenses incurred by Landlord primarily as a result of Tenant’s failure to comply with any of the provisions of Sections 8.2.1(A) through 8.2.1(C) above. Any such amount shall be payable by Tenant to Landlord within ten (10) business days after Landlord’s demand therefor as Additional Rent. Failure of Tenant to pay any amount due pursuant to this Section 8.2.1(D) when due shall be deemed a Tenant Default pursuant to this Lease.
1.2.2.Tenant shall be responsible for all structural engineering reasonably required to determine structural load for any of Tenant’s furniture, fixtures, equipment, other personal property, Alterations and Tenant Improvements; provided that Landlord reserves the right to prescribe the weight and position of all file cabinets, safes and heavy equipment which Tenant desires to place in the Premises so as to properly distribute the weight thereof. Further, Tenant’s business machines and mechanical equipment which cause vibration or noise that may be transmitted to the Building structures or to any other space in the Buildings shall be so installed, maintained and used by Tenant as to eliminate such vibration or noise.
1.3Hazardous Materials.
1.3.1.Tenant shall not cause or permit any Hazardous Materials (as defined in Section 8.3.7 below) to be brought upon, kept or used in or about the Premises, the Buildings or the Project in violation of applicable Laws by Tenant or any of its employees, agents, representatives, contractors or invitees (collectively with Tenant, each a “Tenant Party”). If (a) Tenant breaches such obligation, (b) the presence of Hazardous Materials as a result of such a breach results in contamination of the Project, any portion thereof, or any adjacent property, (c) contamination of the Premises otherwise occurs during the Term or any extension or renewal hereof or holding over hereunder or (d) contamination of the Project occurs as a result of Hazardous Materials that are placed on or under or are released into the Project by a Tenant Party, then Tenant shall indemnify, save, defend (at Landlord’s option and with counsel reasonably acceptable to Landlord) and hold the Landlord Indemnified Parties (as defined in Section 22.1.2 below) harmless from and against any and all Claims (as defined in Article 20 below) of any kind or nature, including (i) diminution in value of the Project or any portion thereof, (ii) damages for the loss or restriction on use of rentable or usable space or of any amenity of the Project, (iii) damages arising from any adverse impact on marketing of space in the Project or any portion thereof and (iv) sums paid in settlement of Claims that arise before, during or after the Term as a result of such breach or contamination. This indemnification by Tenant includes costs incurred in connection with any investigation of site conditions or any clean-up, remedial, removal or restoration work required by any Governmental Authority because of Hazardous Materials present in the air, soil or
groundwater above, on, under or about the Project. Without limiting the foregoing, if the presence of any Hazardous Materials in, on, under or about the Project, any portion thereof or any adjacent property caused or permitted by any Tenant Party results in any contamination of the Project, any portion thereof or any adjacent property, then Tenant shall promptly take all actions at its sole cost and expense as are necessary to return the Project, any portion thereof or any adjacent property to its respective condition existing prior to the time of such contamination; provided that Landlord’s written approval of such action shall first be obtained, which approval Landlord shall not unreasonably withhold; and provided, further, that it shall be reasonable for Landlord to withhold its consent if such actions could have a material adverse long-term or short-term effect on the Project, any portion thereof or any adjacent property. Tenant’s obligations under this Section shall not be affected, reduced or limited by any limitation on the amount or type of damages, compensation or benefits payable by or for Tenant under workers’ compensation acts, disability benefit acts, employee benefit acts or similar legislation. Notwithstanding anything to the contrary in this Lease, Tenant shall have no liability, responsibility, duty or obligation for any Hazardous Materials conditions existing as of the Commencement Date (including, without limitation, any obligation to indemnify or hold Landlord harmless) to the extent not caused, contributed to or exacerbated by a Tenant Party.
1.3.2.Landlord acknowledges that it is not the intent of this Article to prohibit Tenant from operating its business for the Permitted Use. Tenant may operate its business according to the custom of Tenant’s industry so long as the use or presence of Hazardous Materials is strictly and properly monitored in accordance with applicable Laws. As a material inducement to Landlord to allow Tenant to use Hazardous Materials in connection with its business, Tenant agrees to deliver to Landlord (a) a list identifying each type of Hazardous Material to be present at the Premises that is subject to regulation under any environmental applicable Laws (other than customary quantities of typical office and cleaning supplies, provided no permits or approvals from, and no notice or disclosure to, any Governmental Authorities is required in connection with the presence of such supplies at the Premises), (b) a list of any and all approvals or permits from Governmental Authorities required in connection with the presence of such Hazardous Material at the Premises and (c) correct and complete copies of (i) notices of violations of applicable Laws related to Hazardous Materials and (ii) plans relating to the installation of any storage tanks to be installed in, on, under or about the Project (provided that installation of storage tanks shall only be permitted after Landlord has given Tenant its written consent to do so, which consent Landlord may withhold in its sole and absolute discretion) and closure plans or any other documents required by any and all Governmental Authorities for any storage tanks installed in, on, under or about the Project for the closure of any such storage tanks (collectively, “Hazardous Materials Documents”). Tenant shall deliver to Landlord updated Hazardous Materials Documents, within fourteen (14) days after receipt of a written request therefor from Landlord, not more often than once per year, unless (m) there are any changes to the Hazardous Materials Documents or (n) Tenant initiates any Alterations or changes its business, in either case in a way that involves any material increase in the types or amounts of Hazardous Materials. For each type of Hazardous Material listed, the Hazardous Materials Documents shall include (t) the chemical name, (u) the material state (e.g., solid, liquid, gas or cryogen), (v) the concentration, (w) the storage amount and storage condition (e.g., in cabinets or not in cabinets), (x) the use amount and use condition (e.g., open use or closed use), (y) the location (e.g., room number or other identification) and (z) if known, the chemical abstract service number. Notwithstanding anything in this Section to the contrary, Tenant shall not be required to provide Landlord with any Hazardous Materials Documents containing information of a proprietary nature, which Hazardous Materials Documents, in and of themselves, do not contain a reference to any Hazardous Materials or activities related to Hazardous Materials. Landlord may, at Landlord’s expense, cause the Hazardous Materials Documents to be reviewed by a person or firm qualified to analyze Hazardous Materials to confirm compliance with the provisions of this Lease and with applicable Laws. In the event that a review of the Hazardous Materials Documents indicates non-compliance with this Lease or applicable Laws, Tenant shall, at its expense, diligently take steps to bring its storage and use of Hazardous Materials into compliance. Notwithstanding anything in this Lease to the contrary or Landlord’s review into Tenant’s Hazardous Materials Documents or use or disposal of hazardous materials, however, Landlord shall not have and expressly disclaims any liability related to Tenant’s or other tenants’ use or disposal of Hazardous Materials, it being acknowledged by Tenant that Tenant is best suited to evaluate the safety and efficacy of its Hazardous Materials usage and procedures.
1.3.3.At any time, and from time to time, Landlord shall have the right to conduct appropriate tests of the Project or any portion thereof to demonstrate that Hazardous Materials are present or that contamination has occurred due to the acts or omissions of a Tenant Party. Tenant shall pay all reasonable costs of such tests if such tests reveal that Hazardous Materials exist at the Project in violation of Tenant’s obligations under this Lease.
1.3.4.Tenant shall not install or utilize any underground or other storage tanks storing Hazardous Materials on the Premises without Landlord’s prior written consent, which consent may be withheld in Landlord’s sole and absolute discretion. Subject to the foregoing, if underground or other storage tanks storing Hazardous Materials installed or utilized by Tenant are located on the Premises, or are hereafter placed on the Premises by Tenant (or by any other party, if such storage tanks are utilized by Tenant), then Tenant shall monitor the storage tanks, maintain appropriate records, implement reporting procedures, properly close any underground storage tanks, and take or cause to be taken all other steps necessary or required under the applicable Laws.
1.3.5.Tenant shall promptly report to Landlord any actual or suspected presence of toxic or potentially toxic mold or water intrusion at the Premises of which Tenant has notice.
1.3.6.Tenant’s obligations under this Section 8.3 shall survive the expiration or earlier termination of the Lease. During any period of time needed by Tenant or Landlord after the termination of this Lease to complete the removal from the Premises of any Hazardous Materials, Tenant shall be deemed a holdover tenant and subject to the provisions of Section 8.3.
1.3.7.As used in this Lease, the term “Hazardous Material” means any toxic, explosive, corrosive, flammable, infectious, radioactive, carcinogenic, mutagenic or otherwise hazardous substance, material or waste that is or becomes regulated by applicable Laws or any Governmental Authority, and the term “Hazardous Material Laws” means and includes all now and hereafter existing statutes, laws, ordinances, codes, regulations, rules, rulings, orders, decrees, directives, policies and requirements by any federal, state or local governmental authority regulating, relating to, or imposing liability or standards of conduct concerning public health and safety, the environment or any Hazardous Material, including, without limitation, the Comprehensive Environmental Response, Compensation and Liability Act of 1980, as amended (42 U.S.C. Section 9601 et seq.), Resource Conservation and Recovery Act, as amended (42 U.S.C. Section 6901 et seq.), and California Health and Safety Code (Sections 25100, 25249.5, 25316 and 39000, et seq. in each case).
1.3.8.Notwithstanding anything to the contrary in this Lease, Landlord shall have sole control over the equitable allocation of control areas (as defined in the California Building Standards Code) within the Project for the storage of Hazardous Materials. Without limiting the foregoing, if the use of Hazardous Materials by Tenant is such that Tenant utilizes fire control areas in the Project in excess of Tenant’s Percentage of the Buildings or the Project, as applicable, then Tenant shall, at its sole cost and expense and upon Landlord’s written request, establish and maintain a separate area of the Premises classified by the California Building Standards Code as a “Group H” occupancy area for the use and storage of Hazardous Materials, or take such other action as is necessary to ensure that its share of the fire control areas of the Buildings and the Project is not greater than Tenant’s Percentage of the Buildings or the Project, as applicable. Notwithstanding anything in this Lease to the contrary, Landlord shall not have and expressly disclaims any liability related to Tenant’s or other tenants’ use or disposal of fire control areas, it being acknowledged by Tenant that Tenant and other tenants are best suited to evaluate the safety and efficacy of its Hazardous Materials usage and procedures.
1.4Odors and Exhaust. Tenant acknowledges that Landlord would not enter into this Lease with Tenant unless Tenant assures Landlord that under no circumstances will any other occupants of the Buildings or the Project, if any, be subjected to odors or fumes (whether or not noxious), and that the Buildings and the Project will not be damaged by any exhaust, in each case from Tenant’s operations. Landlord and Tenant therefore agree as follows:
1.4.1.Tenant shall not cause or permit (or conduct any activities that would cause) any release of any odors or fumes of any kind from the Premises.
1.4.2.If the Buildings have a ventilation system that, in Landlord’s judgment, is adequate, suitable, and appropriate to vent the Premises in a manner that does not release odors affecting any indoor or outdoor part of the Project, Tenant shall vent the Premises through such system. If Landlord at any time determines that any existing ventilation system is inadequate, or if no ventilation system exists, Tenant shall in compliance with applicable Laws vent all fumes and odors from the Premises (and remove odors from Tenant’s exhaust stream) as Landlord requires. The placement and configuration of all ventilation exhaust pipes, louvers and other equipment shall be subject to Landlord’s approval. Tenant acknowledges Landlord’s desire to maintain the Project (indoor and outdoor areas) in an odor-free manner, and Landlord may require Tenant to abate and remove all odors in a manner that goes beyond the requirements of applicable Laws.
1.4.3.Tenant shall, at Tenant’s sole cost and expense, provide odor eliminators and other devices (such as filters, air cleaners, scrubbers and whatever other equipment may in Landlord’s judgment be necessary or appropriate from time to time) to completely remove, eliminate and abate any odors, fumes or other substances in Tenant’s exhaust stream that in Landlord’s judgment, emanate from Tenant’s Premises. Any work Tenant performs under this Section shall constitute Alterations.
1.4.4.Tenant’s responsibility to remove, eliminate and abate odors, fumes and exhaust shall continue throughout the Term. Landlord’s construction of the Tenant Improvements shall not preclude Landlord from requiring additional measures to eliminate odors, fumes and other adverse impacts of Tenant’s exhaust stream (as Landlord may designate in Landlord’s discretion). Tenant shall install additional equipment as Landlord requires from time to time under the preceding sentence. Such installations shall constitute Alterations.
1.4.5.If Tenant fails to install satisfactory odor control equipment within ten (10) business days after Landlord’s demand made at any time, then Landlord may, without limiting Landlord’s other rights and remedies, require Tenant to cease and suspend any operations in the Premises that, in Landlord’s determination,
cause odors, fumes or exhaust. For example, if Landlord determines that Tenant’s production of a certain type of product causes odors, fumes or exhaust, and Tenant does not install satisfactory odor control equipment within ten (10) business days after Landlord’s request, then Landlord may require Tenant to stop producing such type of product in the Premises unless and until Tenant has installed odor control equipment satisfactory to Landlord.
1.4.6.Notwithstanding the foregoing provisions of Section 8.4 to the contrary, during any period of time in which Tenant is the sole occupant of the Project, Tenant’s obligations to control and/or mitigate odors, exhaust and fumes as set forth in this Section 8.4 shall be limited to the extent such obligations are required by applicable Laws.
Article 9
MOLD
Tenant agrees to use commercially reasonable efforts to maintain the Premises in a manner that prevents the occurrence of an infestation of toxic, or potentially toxic, mold, mildew, microbial growths, and any associated mycotoxins in the Premises, and shall comply, at a minimum, with the following: (a) Tenant agrees to immediately fix/abate any water intrusion in the Premises to the extent such action is required to be undertaken by Tenant pursuant to its maintenance obligations under this Lease, or, if not within Tenant’s maintenance obligations, then Tenant agrees to immediately notify Landlord of any such water intrusion; (b) Tenant agrees to use all reasonable care to close all windows and other openings in the Premises to prevent outdoor water from penetrating into the interior unit; (c) Tenant agrees to clean and dry any visible moisture on windows, walls, and other surfaces, including personal property, as soon as reasonably possible after discovery; (d) Tenant agrees to keep the Premises free of dirt and debris that could reasonably be expected to harbor mold; (e) Tenant agrees to regularly clean and sanitize kitchens and other surfaces within the Premises where water, moisture condensation, and mold could reasonably be expected to collect; (f) Tenant agrees not to interfere with regular air flow and circulation throughout the Premises; (g) Tenant agrees to limit the indoor watering of plants; (h) Tenant agrees to use commercially reasonable efforts to prevent the overflow or release of water from bathrooms or kitchens, including but not limited to toilets, sinks, kitchen appliances, and other receptacles of water; (i) Tenant agrees not to obstruct fresh air supply to furnace, air conditioner or heater ducts; (j) Tenant agrees to maintain and not obstruct ventilation at all locations in the Premises; (k) Tenant agrees to use commercially reasonable efforts to prevent the clogging of all plumbing within the Premises; (l) Tenant agrees not to engage in any conduct that would reasonably be expected to promote or create mold growth; (m) Tenant agrees to report, within forty-eight (48) hours after Tenant discovery, the following to Landlord: (i) any non-working fan, heater, air conditioner or ventilation system; (ii) plumbing leaks, drips, sweating pipes, wet spots; (iii) overflows from bathroom, kitchen, or other facilities, including, but not limited to, tubs, showers, shower enclosures, toilets, sinks, kitchen appliances, or other receptacles of water, especially in cases where the overflow may have permeated walls, floors, ceilings or fixtures; (iv) water intrusion of any kind; (v) any mold or black or brown spots or moisture on surfaces inside the Premises; (vi) broken plumbing systems or standing water near structures within the Premises; and (vii) any odors consistent with mold growth within the Premises. Tenant agrees not to commence any mold investigation, testing, remediation or repair without first obtaining the prior written consent of Landlord. If Landlord consents to any mold investigation, remediation or repair by Tenant, Tenant agrees to not use any methods of mold investigation, testing, remediation and repair that are speculative and not generally accepted within the scientific community, and Landlord reserves the right to approve any and all third parties retained by Tenant to conduct any such mold investigation, testing, remediation and repair. As of the Effective Date such speculative and generally unaccepted methods of investigation, testing, remediation and repair include: (A) any use of settled dust vacuum sampling; (B) any use of interior wall cavity air sampling; (C) Tenant’s use of do-it-yourself mold investigation kits; and (D) use of any other methods that have not been peer reviewed and generally accepted within the scientific community.
Article 10
NOTICES
1.1Method of Delivery. Any notice, consent, approval or objection required or permitted by this Lease (a “Notice”) shall be in writing and may be delivered: (a) in person (by hand or by messenger or courier service) or (b) by certified or registered mail or United States Postal Service Express Mail, with postage prepaid, or (c) by a nationally recognized overnight delivery service that provides delivery verification, addressed to Tenant at the Premises and to Landlord at each of the addresses designated in Section 1.2, and shall be deemed sufficiently given if served in a manner specified in this Article 10. Either party may specify a different address for Notice purposes by Notice to the other.
1.2Receipt of Notices. Any Notice sent by registered or certified mail, return receipt requested, shall be deemed given on the date of delivery shown on the receipt card, or if no delivery date is shown, the postmark thereon. Notices delivered by United States Postal Service Express Mail or overnight delivery service that guarantees next day delivery shall be deemed given on the next business day after delivery of the same to the United
States Postal Service or overnight delivery service. If any Notice is received on a Saturday, Sunday or legal holiday, it shall be deemed received on the next business day.
1.3Statutory Service of Notice. When a statute permits, or requires, service of a notice in a particular manner, service of that notice (or a similar Notice permitted, or required, by this Lease) in the manner permitted, or required, by this Article 10 shall replace and satisfy the statutory service-of-notice procedures, including, but not limited to, those required by California Code of Civil Procedure Section 1162, or any similar or successor statute.
Article 11
BROKERS
Tenant warrants that it has had no dealings with any real estate broker, finder or agent in connection with the negotiation of this Lease except for the broker(s) whose name(s) is (are) set forth in Section 1.16, whose commission shall be payable by Landlord pursuant to one or more separate agreements, and that it knows of no other real estate broker, finder or agent who is or might be entitled to a commission in connection with this Lease. Tenant shall be solely responsible for the payment of any fee due to any other broker, finder, agent or other party claiming under Tenant, and shall hold Landlord free and harmless against any liability in respect thereto, including attorneys’ fees and costs incurred by Landlord in connection therewith.
Article 12
HOLDING OVER
If Tenant holds over after the expiration or earlier termination of the Term hereof without the express written consent of Landlord, Tenant shall become a Tenant at sufferance, at a Basic Rent equal to one hundred twenty-five percent (125%) of the Rent payable during the last month of the Term, and otherwise subject to the terms, covenants and conditions herein specified, so far as applicable. Acceptance by Landlord of Rent after such expiration or earlier termination without Landlord’s prior written consent shall not waive Landlord’s right to evict Tenant without thirty (30) days prior written notice. The foregoing provisions of this Article 12 are in addition to and do not affect Landlord’s right of reentry or any rights of Landlord hereunder or as otherwise provided by law. If Tenant fails to surrender the Premises upon the expiration or earlier termination of this Lease, Tenant shall indemnify, defend and hold Landlord harmless from all Claims, including, without limitation, any claim made by any succeeding tenant founded on or resulting from such failure to surrender, lost profits and other consequential damages, and any and all attorneys’ fees and costs incurred by Landlord in connection Tenant’s failure to surrender the Premises in accordance with the provisions of this Lease on the expiration or earlier termination of this Lease.
Article 13
TAXES ON TENANT’S PROPERTY
1.1Personal Property and Fixtures. Tenant shall be liable for and shall pay, at least ten (10) days before delinquency, all taxes levied against any of Tenant’s Personal Property (defined below) placed by Tenant or any Tenant Party in or about the Premises. If any such taxes on Tenant’s Personal Property are levied against Landlord or Landlord’s property, or if the assessed value of the Premises, Buildings or Project is increased by the inclusion therein of a value placed upon such Tenant’s Personal Property, and if Landlord, after Notice to Tenant, pays the taxes based upon such increased assessment, which Landlord shall have the right to do regardless of the validity thereof (but only under proper protest if so requested by Tenant), Tenant shall, upon demand, repay to Landlord the taxes so levied against Landlord, or the portion of such taxes resulting from such increase in the assessment.
1.2Tenant Improvements. If the Leasehold Improvements (defined below) in the Premises, whether installed and/or paid for by Landlord or Tenant and whether or not affixed to the real property so as to become a part thereof, are assessed for real property tax purposes at a valuation higher than the valuation at which tenant improvements conforming to Landlord’s “Building Standard Improvements” (as defined in the Work Letter Agreement) for other space in the Buildings are assessed, then the real property taxes and assessments levied against the Buildings or Project by reason of such excess assessed valuation shall be deemed to be taxes levied against Tenant’s Personal Property and shall be governed by the provisions of Section 13.1 above. If the records of the County Assessor are available and sufficiently detailed to serve as a basis for determining whether the Leasehold Improvements are assessed at a higher valuation than Landlord’s “Building Standard Improvements”, such records shall be binding on both Landlord and Tenant. If the records of the County Assessor are not available or sufficiently detailed to serve as a basis for making said determination, the actual cost of construction shall be used.
1.3Additional Taxes. Tenant shall pay to Landlord, within ten (10) days of Landlord’s demand therefor, and in such manner and at such times as Landlord shall direct from time to time by written notice to
Tenant, any excise, sales, privilege or other tax, assessment or other charge (other than income or franchise taxes) imposed, assessed or levied by any Governmental Authority upon Landlord on account of: (a) the Rent payable by Tenant hereunder (or any other benefit received by Landlord hereunder), including, without limitation, any gross receipts tax, license fee or excise tax levied by any Governmental Authority, (b) this Lease, Landlord’s business as a lessor hereunder, and the possession, leasing, operation, management, maintenance, alteration, repair, use or occupancy of any portion of the Premises (including, without limitation, any applicable possessory interest taxes), (c) this transaction or any document to which Tenant is a party creating or transferring an interest or an estate in the Premises, or (d) otherwise in respect of or as a result of the agreement or relationship of Landlord and Tenant hereunder.
Article 14
CONDITION OF PREMISES
1.1As Is. Tenant acknowledges and agrees that, except with respect to the Landlord Work: (a) Tenant has inspected, or has had the opportunity to inspect, the Project, the Buildings and the Premises and, subject to Landlord’s obligations under this Lease, acknowledges that the same are acceptable for Tenant’s intended use and agrees to accept them in their “AS IS, WHERE IS” condition, (b) except as expressly provided in this Lease, neither Landlord nor any agent of Landlord has made any representation or warranty with respect to the Premises, the Buildings, the Parking Area or any other portion of the Project or with respect to the condition thereof or the suitability of the same for the conduct of Tenant’s business, (c) except as expressly provided in the Work Letter Agreement and Section 16.2 below, and subject to the express representations and warranties of Landlord set forth in this Lease, Landlord shall have no obligation to alter, remodel, improve, repair, decorate or paint the Premises or any part thereof, or any portion of the Buildings or Project and (d) except as expressly provided in this Lease, Landlord shall have no obligation to provide Tenant with any allowance, rent credit or abatement in connection with Tenant’s entering into this Lease. The taking of possession of the Premises by Tenant shall conclusively establish that the Project, the Buildings and the Premises were at such time in good order and clean condition and that Landlord shall have discharged all of its obligations under the Work Letter Agreement (subject to any punch list items as set forth in the Work Letter Agreement), and the execution of this Lease by Tenant shall conclusively establish that the Premises, the Buildings, the Project and the Parking Area were in good and sanitary order, condition and repair at such time, except for latent defects, if any. Without limiting the foregoing, Tenant’s execution of the Memorandum of Terms shall constitute a specific acknowledgment and acceptance of the various start-up inconveniences that may be associated with the use of the Buildings, the Parking Area and other portions of the Project, such as certain construction obstacles (e.g., scaffolding), delays in use of freight elevator service, unavailability of certain elevators for Tenant’s use, uneven air-conditioning services and other typical conditions incident to recently constructed (or recently modified) office and laboratory/research and development buildings. Tenant (for itself and all other claiming through Tenant) hereby irrevocably waives and releases its right to terminate this Lease under Section 1932(l) of the California Civil Code. Notwithstanding anything to the contrary in this Lease, Landlord represents, to the actual knowledge of Steve Bollert, without a duty to inquire or investigate, that, as of the Commencement Date, (a) the Premises are not in material violation of any Laws, and (b) there are no damages or defects existing in the Premises that would not be visible upon an in-person inspection of the Premises.
1.2Limited Warranty. Notwithstanding the foregoing provisions of Section 14.1, Landlord hereby warrants that all Building Systems (as that term is defined in Section 15.1 below) servicing the Premises shall be in good working order and condition for a period of twelve (12) months beginning on the Commencement Date. In the event of a failure of the foregoing warranty, provided that Tenant delivers Notice thereof to Landlord within the applicable twelve (12) month period, then Landlord shall cure such failure within a reasonable period of time after receiving such Notice and the cost thereof shall be at Landlord’s sole cost and expense and shall not be included in Operating Expenses, except to the extent such failure is caused by Tenant or any Tenant Party, in which event Tenant shall reimburse Landlord for the cost to cure such failure.
Article 15
ALTERATIONS
1.1Alterations and Major Alterations. Except for Permitted Alterations, Tenant shall make no alterations, additions, or improvements in or to the Premises (collectively, the “Alterations”) without Landlord’s prior written consent, which consent shall not be unreasonably withheld, conditioned or delayed for all Alterations other than Major Alterations (which shall be granted in Landlord’s sole discretion), and then only by licensed contractors or mechanics approved by Landlord in writing, which approval shall not be unreasonably withheld, conditioned or delayed (provided that any contractors performing any Major Alterations shall be subject to approval by Landlord in its sole and absolute discretion). Tenant shall submit to Landlord plans and specifications for any proposed Alterations to the Premises, and may not make such Alterations until Landlord has approved such plans and specifications and the contractor performing any Alterations in writing. Tenant shall construct such Alterations in accordance with the plans and specifications approved by Landlord and in compliance with all applicable Laws,
and shall not amend or modify such plans and specifications without Landlord’s prior written consent. If any proposed Alterations require the consent or approval of any lessor of a superior lease or the holder of a mortgage encumbering the Premises, Tenant acknowledges that such consent or approval must be secured prior to the construction of such Alterations. Tenant agrees not to construct or erect partitions or other obstructions that might interfere with Landlord’s free access to mechanical installations or service facilities of the Buildings or interfere with the moving of Landlord’s equipment to or from the enclosures containing said installations or facilities. All Alterations shall be done at such times and in such manner as Landlord may from time to time reasonably designate. Tenant will pay the entire cost and expense of all Alterations, including, without limitation, for any painting, restoring or repairing of the Premises or the Buildings necessitated by the Alterations, and Landlord’s actual out-of-pocket third party review and Landlord’s supervision fee in an amount equal to three percent (3%) of the cost of the Alterations in question (except for Permitted Alterations in which case Tenant shall not be required to pay Landlord any supervision fee). Tenant will also obtain and/or require: (a) builder’s “all-risk” insurance (or the equivalent thereof) in an amount at least equal to the replacement value of the Alterations; (b) liability insurance insuring Tenant and each of Tenant’s contractors against construction related risks in at least the form, amounts and coverage required of Tenant under Article 22; and (c) if requested by Landlord, demolition (if applicable) and payment and performance bonds in an amount not less than the full cost of the Alterations. The insurance policies described in clause (b) of this Section 15.1 must name Landlord, Landlord’s lender (if any), Bioscience Properties, Inc. (“Property Manager”) and other parties reasonably requested by Landlord as additional insureds, specifically including completed operations. Tenant covenants and agrees that all Alterations done by Tenant shall be performed in full compliance with all Laws. If any Governmental Authority requires any alterations or modifications to the Buildings or the Premises as a result of Tenant’s Permitted Use of the Premises or as a result of any Alteration to the Premises made by or on behalf of Tenant, Tenant will pay the cost of all such alterations or modifications. If any such Alterations involve any modifications to (i) the structural portions of the Buildings, (ii) the mechanical, electrical, plumbing, fire/life safety or heating, ventilating and air conditioning systems of the Buildings (collectively, “Building Systems”) or (iii) any portion of the Buildings outside of the interior of the Premises (a “Major Alteration”), it shall be reasonable for Landlord to withhold its consent to any such Major Alterations and it shall be reasonable for Landlord to condition its consent to any Major Alterations on Landlord making the Major Alterations, provided that Landlord may first require Tenant to deposit with Landlord an amount sufficient to pay the cost of the Major Alterations (including, without limitation, reasonable overhead, administrative costs and profit). Before commencing any work, Tenant shall give Landlord at least ten (10) days’ Notice of the proposed commencement of such work and shall, if required by Landlord, deliver a copy of the completion and payment bond required by Landlord in form, substance and amount satisfactory to Landlord. “Permitted Alterations” means only cosmetic and non-structural Alterations and usual and customary maintenance and repairs of Leasehold Improvements if and to the extent that such cosmetic and non-structural Alterations, maintenance and repairs: (A) are of a type and extent which are customarily permitted to be made without consent by landlords acting consistently with Institutional Owner Practices leasing similar space for similar uses to similar tenants, (B) are in compliance with the Rules and Regulations and all applicable Laws, (C) are not Major Alterations, and (D) do not cost more than Fifty Thousand and 00/100 Dollars ($50,000.00) in each instance.
1.2Removal of Alterations and Tenant’s Personal Property. The Tenant Improvements together with all Alterations upon the Premises made by Tenant after the Commencement Date, including, without limitation, all wall coverings, built-in cabinet work, paneling and the like (collectively, “Leasehold Improvements”), shall, at Landlord’s election given at the time Landlord approves such Alterations, either be removed by Tenant or shall become the property of Landlord and shall remain upon, and be surrendered with, the Premises at the end of the Term hereof; provided, however, that if Landlord, by Notice to Tenant at the time Landlord approves such Alterations, requires Tenant to remove any such Leasehold Improvements, Tenant shall repair all damage resulting from such removal or, at Landlord’s option, shall pay to Landlord the cost of such removal, as reasonably estimated by Landlord, prior to the expiration of the Term of this Lease. Notwithstanding the foregoing, Tenant shall not be required, nor have the right, to remove the Tenant Improvements furnished pursuant to the Work Letter Agreement. All articles of personal property and all business and trade fixtures, cabling, machinery and equipment, furniture and movable partitions owned by Tenant or any other Tenant Party or that are installed by or for Tenant or any other Tenant Party at its expense in the Premises (collectively, “Tenant’s Personal Property”) shall be and remain the property of Tenant and shall be removed by Tenant prior to the expiration of the Term, and Tenant shall repair all damage to the Premises, if any, resulting from such removal. If Tenant shall fail to remove any of the foregoing from the Premises prior to termination of this Lease for any cause whatsoever, Tenant shall be deemed to be holding over in the Premises without the consent of Landlord and Landlord may, at its option, remove the same in any manner that Landlord shall choose, and store the same without liability to Tenant for loss thereof. In such event, Tenant agrees to pay to Landlord upon demand, any and all expenses incurred in such removal (including court costs and attorneys’ fees) and storage charges thereon, for any length of time that the same shall be in Landlord’s possession or control. Landlord may, at its option, without Notice, sell such property, or any of the same, at a private sale and without legal process, for such price as Landlord may obtain and apply the proceeds of such sale to any amounts due under this Lease from Tenant to Landlord and/or to all expenses, including attorneys’ fees and costs, incident to the removal and/or sale thereof.
Article 16
REPAIRS
1.1Tenant Obligations. Tenant shall, when and if needed, at Tenant’s sole cost and expense and subject to Section 14.2 and Article 15 above, make all repairs to the Premises and every part thereof (including all structural and non-structural parts, but excluding the roof membrane) to maintain the Premises in the condition and repair that existed as of the Commencement Date, reasonable wear and tear and casualty damage excepted, and free from any Hazardous Materials. Except as expressly set forth in Section 14.2 above and Section 16.2 below, all Supplemental Equipment and all Building Systems exclusively serving the Premises shall be maintained, repaired and replaced as needed by Tenant at Tenant’s sole cost and expense and Landlord shall have no liability for the operation, repair, maintenance or replacement of any Supplemental Equipment, nor shall Landlord have any liability for the operation, repair or maintenance of any Building Systems exclusively serving the Premises. “Supplemental Equipment” means any items that are installed within the Premises by or at the direction of Tenant or that exclusively serve the Premises (other than standard Building Systems), including, without limitation: (A) any supplemental, specialty or non-Building standard electrical (including lighting), mechanical, plumbing, heating, ventilation and air conditioning systems, fixtures and equipment; (B) any supplemental, specialty or non-Building standard fire, life, safety or security systems, fixtures and equipment; and (C) all video, audio, communications or computer systems, fixtures and equipment (including cabling). Without limiting the foregoing, Tenant shall maintain, at its sole cost and expense, a contract for the regular maintenance and repair of the heating, ventilation and air conditioning systems, fixtures and equipment located in or exclusively serving the Premises.
1.2Landlord Obligations. Landlord shall maintain, repair and replace the structural portions of the Buildings (including, without limitation, the roof structure, roof membrane, slab and exterior walls) outside of the Premises, the Parking Area and the Building Systems, and, except as expressly set forth in Section 14.2 above, the costs incurred by Landlord in performing such maintenance, repairs and replacements shall be included in Operating Expenses to the extent permitted under Section 6.1 above. For purposes of clarification, Landlord shall have no obligation to repair, maintain or replace any part of the Premises, any Building Systems exclusively serving the Premises (except as expressly set forth in Section 14.2 above) or any Supplemental Equipment. Landlord shall not be liable for any failure to make any repairs or replacements or to perform any maintenance to the extent that the need for such repairs, replacements or maintenance is caused by the negligence or willful misconduct of any Tenant Party. Except as provided in Articles 23 and 24 hereof, there shall be no abatement of Rent and no liability of Landlord by reason of any injury to or interference with Tenant’s business arising from the making of any repairs, alterations, improvements or replacements in or to any portion of the Buildings or the Premises or in or to fixtures, appurtenances and equipment therein. Tenant waives the right to make repairs and replacements at Landlord’s expense under any Law now or hereafter in effect including Section 1941 and 1942 of the California Civil Code (as the same may be amended from time to time) and any successor statute and similar Law now or hereafter in effect.
Article 17
LIENS
Tenant shall not cause or permit to be filed against the Premises, the Buildings or the Project or of any portion thereof or against Tenant’s leasehold interest in the Premises any mechanics’, materialmen’s or other liens, including without limitation any state, federal or local “superfund” or Hazardous Materials cleanup lien imposed as a result of the presence of Hazardous Materials in, on or about the Premises, the Buildings or any other portion of the Project. Landlord shall have the right at all reasonable times to post and keep posted on the Premises any notices that it deems necessary for protection from such liens. Tenant shall discharge any lien filed against the Premises or against the Buildings for work claimed to have been done for, or materials claimed to have been furnished to, Tenant, by bond or otherwise, within ten (10) business days after the filing thereof, at the cost and expense of Tenant. If any such liens are filed and Tenant fails to discharge them pursuant to the foregoing sentence, Landlord may, without waiving its rights and remedies based on such breach of Tenant and without releasing Tenant from any of its obligations hereunder, cause such lien(s) to be released by any means it shall deem proper, including payments in satisfaction of the claim giving rise to such lien or by obtaining a corporate statutory mechanic’s lien release bond in an amount equal to one hundred fifty percent (150%) of such lien claim. Tenant shall: (a) pay to Landlord, immediately upon Notice from Landlord, any cost or expense, including, without limitation, attorneys’ fees and costs, incurred by Landlord by reason of Tenant’s failure to discharge any such lien, together with interest thereon at the maximum rate per annum permitted by Law from the date of such payment by Landlord and (b) shall indemnify, defend and hold the Landlord Indemnified Parties harmless from and against any liens.
Article 18
ENTRY BY LANDLORD
Landlord reserves and shall at any and all reasonable times and upon at least twenty-four (24) hours prior notice to Tenant (except in the case of an emergency) and during normal business hours (except in the case of an
emergency) have the right to enter the Premises to supply any service to be provided by Landlord to Tenant hereunder, to inspect the same, to show the Premises to prospective purchasers, lenders, or investors and during the last twelve (12) months of the Term or following a default by Tenant to prospective tenants, to post notices of non-responsibility, to alter, improve or repair the Premises or any other portion of the Buildings and/or the Project, as provided in Section 2.4 above, or for any other reasonable purpose, all without being deemed guilty of any eviction of Tenant and without abatement of Rent. Landlord may, in order to carry out such purposes, erect scaffolding and other necessary structures where reasonably required by the character of the work to be performed, provided that the business of Tenant shall be interfered with as little as is reasonably practicable. Tenant hereby waives any claim for damages for any injury or inconvenience to or interference with Tenant’s business, for any loss of occupancy or quiet enjoyment of the Premises and for any other loss in, upon and about the Premises, the Buildings or the Project on account of Landlord’s entry or work permitted by this Article 18 or by Section 2.4 above, except to the extent caused by Landlord’s gross negligence or willful misconduct; provided, however, that in no event shall Landlord be liable for consequential damages or lost profits. Without limiting the foregoing, Landlord shall use commercially reasonable efforts to minimize any disruption or interference to Tenant’s business during such entry. Landlord shall at all times have and retain a key with which to unlock all doors in the Premises, excluding Tenant’s vaults and safes. Landlord shall have the right to use any and all means that Landlord may deem proper to open said doors in an emergency in order to obtain entry to the Premises. Any entry to the Premises obtained by Landlord by any of said means, or otherwise, shall not be construed or deemed to be a forcible or unlawful entry into the Premises, or an eviction of Tenant from the Premises or any portion thereof, and any damages caused on account thereof shall be paid by Tenant.
Article 19
UTILITIES AND SERVICES
1.1Premises Utilities. Notwithstanding anything to the contrary in this Lease, Tenant shall pay for the cost of all water (including the cost to service, repair and replace reverse osmosis, de-ionized and other treated water), electricity, gas, heating, ventilation and air-conditioning (“HVAC”), light, power, telephone, internet service, cable television, other telecommunications and other utilities supplied to the Premises, together with any fees, surcharges and taxes thereon. All such utilities and services provided to the Premises that are separately metered shall be paid by Tenant directly to the supplier of such utilities or services. If any such utility is not separately metered to Tenant, Tenant shall pay Tenant’s Percentage of all charges of such utility jointly metered with other premises as Additional Rent or, in the alternative, Landlord may, at its option, monitor the usage of such utilities by Tenant and charge Tenant with the cost of purchasing, installing and monitoring such metering equipment, which cost shall be paid by Tenant as Additional Rent. Landlord may base its bills for utilities on reasonable estimates; provided that Landlord adjusts such billings promptly thereafter or as part of the next Annual Reconciliation to reflect the actual cost of providing utilities to the Premises. In the event that the Buildings or Project is less than fully occupied during a calendar year, Tenant acknowledges that Landlord may extrapolate utility usage that varies depending on the occupancy of the Buildings or Project (as applicable) to equal Landlord’s reasonable estimate of what such utility usage would have been had the Buildings or Project, as applicable, been one hundred percent (100%) occupied during such calendar year; provided, however, that Landlord shall not recover more than one hundred percent (100%) of the cost of such utilities. Landlord may, in Landlord’s sole and absolute discretion, at any time and from time to time, contract, or require Tenant to contract, for utility services (including generation, transmission, or delivery of the utility service) with a utility service provider(s) of Landlord’s choosing. Tenant shall fully cooperate with Landlord and any utility service provider selected by Landlord. Tenant shall permit Landlord and the utility service provider to have reasonable access to the Premises and the utility equipment serving the Premises, including lines, feeders, risers, wiring, pipes, and meters. Tenant shall either pay or reimburse Landlord for all costs associated with any change of utility service, including the cost of any new utility equipment, within ten (10) business days after Landlord’s written demand for payment or reimbursement.
1.2Janitorial Service. Tenant, at its sole cost and expense, shall enter into an agreement for regular janitorial services for the Premises with a company which is fully bonded and insured and approved by Landlord in its reasonable discretion. Tenant shall keep the Premises at all times in a clean and orderly condition, at Tenant’s expense and to the reasonable satisfaction of Landlord. Unless otherwise agreed to by Landlord, no one other than persons approved by Landlord shall be permitted to enter the Premises for the purpose of providing janitorial or cleaning service.
1.3Landlord Exculpation. Landlord’s failure to furnish or cause to be furnished any service which Landlord is required or elects to provide hereunder shall not result in any liability to Landlord. Landlord shall not be responsible or liable for any loss, damage, or expense that Tenant may incur as a result of any change of utility service, including any change that makes the utility supplied less suitable for Tenant’s needs, or for any failure, interruption, stoppage, or defect in any utility service. In addition, Tenant shall not be entitled to any abatement or reduction of Rent, no eviction of Tenant shall result from and Tenant shall not be relieved from the performance of any covenant or agreement in this Lease by reason of any such change, failure, interruption, stoppage or defect. In the event of any such failure, interruption, stoppage or defect of a service which Landlord is required to provide
hereunder, Landlord shall diligently attempt to cause service to be resumed promptly. Notwithstanding the foregoing, in the event that Tenant is prevented from using, and does not use, the Premises or any portion thereof, as a result of a failure to provide any services or utilities to the Premises, to the extent within Landlord’s reasonable control, which Landlord is required to provide under this Lease (an “Abatement Event”), then Tenant shall give Landlord written Notice of such Abatement Event, and if such Abatement Event continues for five (5) consecutive business days after Landlord’s receipt of such Notice (“Eligibility Period”) and Landlord does not diligently commence and pursue to completion the remedy of such Abatement Event, then, except to the extent covered by business interruption or similar insurance carried or required to be carried by Tenant hereunder, Basic Rent, Tenant’s Percentage of Operating Expenses and Tenant’s Percentage of Real Property Taxes shall be abated or reduced, as the case may be, after expiration of the Eligibility Period for such time that Tenant continues to be so prevented from using, and does not use, the Premises or a portion thereof, in the proportion that the rentable area of the portion of the Premises that Tenant is prevented from using, and does not use, bears to the total rentable area of the Premises. If, however, Tenant reoccupies any portion of the Premises during such period, the Basic Rent, Tenant’s Percentage of Operating Expenses and Tenant’s Percentage of Real Property Taxes allocable to such reoccupied portion, based on the proportion that the rentable area of such reoccupied portion of the Premises bears to the total rentable area of the Premises, shall be payable by Tenant from the date Tenant reoccupies such portion of the Premises. Such right to abate Basic Rent, Tenant’s Percentage of Operating Expenses and Tenant’s Percentage of Real Property Taxes shall be Tenant’s sole and exclusive remedy at law or in equity for an Abatement Event. Except as expressly provided in this Section 19.3, nothing contained herein shall be interpreted to mean that Tenant is excused from paying Rent due hereunder.
1.4Intentionally Deleted.
1.5Common Area Water. Landlord shall provide water in the Common Area for, to the extent applicable, lavatory and landscaping purposes only, which water shall be from the local municipal or similar source.
1.6Energy Tracking. Within ten (10) business days following Landlord’s written request therefor, Tenant shall deliver to Landlord copies of any invoices for utility services provided to the Premises and related information reasonably requested by Landlord in connection with the requirements of California Public Resources Code Section 25402.10, the corresponding regulations adopted by the California Energy Commission and provided in California Code of Regulations, Title 20, Division 2, Chapter 4, Article 9, Sections 1680-1684, and any supplemental and/or successor statute or regulations concerning the reporting of energy usage and efficiency relative to commercial buildings. Tenant acknowledges that any utility information for the Premises, the Buildings and the Project may be shared with third parties, including Landlord’s consultants and Governmental Authorities. In addition to the foregoing, Tenant shall comply with all applicable Laws related to the disclosure and tracking of energy consumption at the Premises. The provisions of this Section shall survive the expiration or earlier termination of this Lease.
1.7Reservation of Rights. Landlord reserves the right to stop service of the plumbing, ventilation, air conditioning and utility systems, when Landlord deems necessary or desirable, due to accident, emergency or the need to make repairs, alterations or improvements, until such repairs, alterations or improvements shall have been completed, and Landlord shall further have no responsibility or liability for failure to supply plumbing, ventilation, air conditioning or utility service when prevented from doing so by Force Majeure (as defined in Section 36.8 below) or, to the extent permitted by applicable Law, Landlord’s negligence. Without limiting the foregoing, it is expressly understood and agreed that any covenants on Landlord’s part to furnish any service pursuant to any of the terms, covenants, conditions, provisions or agreements of this Lease, or to perform any act or thing for the benefit of Tenant, shall not be deemed breached if Landlord is unable to furnish or perform the same by virtue of Force Majeure or, to the extent permitted by applicable Law, Landlord’s negligence.
Article 20
INDEMNIFICATION AND EXCULPATION OF LANDLORD
Tenant shall indemnify, defend and hold harmless the Landlord Indemnified Parties (as defined in Section 22.1.2 below) from and against any and all claims, demands, penalties, fines, liabilities, actions (including, without limitation, informal proceedings), settlements, judgments, damages, losses, costs and expenses (including attorneys’ fees and costs) of whatever kind or nature, known or unknown, contingent or otherwise, incurred or suffered by or asserted against such Landlord Indemnified Party (collectively, “Claims”) arising from or in connection with, directly or indirectly, (a) any cause whatsoever in the Premises (including, but not limited to, Claims resulting in whole or in part from the negligence of the Landlord Indemnified Party), except to the extent directly caused by the gross negligence or intentional misconduct of such Landlord Indemnified Party, (b) the presence at or use or occupancy of the Premises or Project by a Tenant Party, (c) any act, neglect, fault or omission on the part of any Tenant Party, or (d) a breach or default by Tenant in the performance of any of its obligations hereunder. Payment shall not be a condition precedent to enforcement of the foregoing indemnity. In case any action or proceeding shall be brought against any Landlord Indemnified Party by reason of any such Claim, at such Landlord Indemnified Party’s option, upon Notice from Landlord, Tenant shall defend the same at Tenant’s expense by counsel selected by
Landlord in its sole discretion. Tenant, as a material part of the consideration to Landlord, hereby assumes all risk of damage to property (including, without limitation, any damage to personal property or scientific research, including loss of records kept by Tenant within the Premises (in each case, regardless of whether such damage is foreseeable)) or injury to Tenant or any other Tenant Parties in, upon or about the Premises, the Buildings, the Parking Area or the Project from any cause whatsoever and hereby waives all Claims (including consequential damages and claims for injury to Tenant’s business or loss of income arising out of any loss of use of the Premises, the Buildings, the Parking Area or the Project or any equipment or facilities therein, or relating to any such damage or destruction of personal property as described in this Section) in respect thereof against each Landlord Indemnified Party, except that which is solely caused by, or solely the result of: (i) any Landlord Default (defined below), (ii) the grossly negligent acts of such Landlord Indemnified Party, or (iii) the willful misconduct of such Landlord Indemnified Party. Landlord shall not be liable for any damages arising from any act, omission or neglect of any other tenant in the Project, or of any other third party, except to the extent arising from the gross negligence or willful misconduct of Landlord. Tenant acknowledges that security devices and services, if any, while intended to deter crime, may not in given instances prevent theft or other criminal acts. Landlord shall not be liable for injuries or losses caused by criminal acts of third parties, and Tenant assumes the risk that any security device or service may malfunction or otherwise be circumvented by a criminal. If Tenant desires protection against such criminal acts, then Tenant shall, at Tenant’s sole cost and expense, obtain appropriate insurance coverage. Without limitation on other obligations of Tenant that survive the expiration of the Term, the clauses of this Article 20 shall survive the expiration or earlier termination of this Lease until all Claims against the Landlord Indemnified Parties involving any of the indemnified matters are fully, finally, and absolutely barred by the applicable statutes of limitations.
Subject to the provisions of Section 22.4 below, Landlord shall indemnify, defend and hold harmless Tenant from and against any and all third party claims, demands, penalties, fines, liabilities, actions (including, without limitation, informal proceedings), settlements, judgments, damages, losses, costs and expenses (including reasonable attorneys’ fees and costs) of whatever kind or nature, known or unknown, contingent or otherwise, incurred or suffered by or asserted against Tenant to the extent proximately caused by the gross negligence or willful misconduct of Landlord in connection with Landlord’s activities on or about the Buildings or Project.
Article 21
DAMAGE TO TENANT’S PROPERTY
Notwithstanding the provisions of Article 20 or anything to the contrary in this Lease, no Landlord Indemnified Party shall be liable for: (a) loss or damage to any property by theft or any other cause whatsoever, (b) any injury or damage to persons resulting from fire, storms, earthquakes, explosion, falling plaster, steam, gas, electricity, water or rain which may leak from any part of the Buildings or the Project or from the pipes, appliances or plumbing work therein or from the roof, street or sub-surface or from any other place or resulting from dampness or any other cause whatsoever, except that which is solely caused by, or solely the result of: (i) the grossly negligent acts of such Landlord Indemnified Party, (ii) the willful misconduct of such Landlord Indemnified Party, or (iii) any latent defect in the Premises, the Buildings or any other portion of the Project that is not promptly remedied by Landlord following Landlord's receipt of notice thereof. Tenant shall immediately give Notice to Landlord in case of the occurrence of any fire or accidents in or about the Premises, the Buildings or any other portion of the Project, or the discovery of any defects therein (including, without limitation, any latent defect in the Premises) or in any fixtures or equipment that are the property of Landlord, Tenant or any other tenant or occupant of premises in the Project.
Without limiting the foregoing, Tenant acknowledges that safety and access control devices, services and programs provided by Landlord, if any, while intended to deter crime and ensure safety, may not in given instances prevent theft or other criminal acts, or ensure safety of persons or property. The risk that any safety or access control device, service or program may not be effective, or may malfunction, or be circumvented by a criminal, is assumed by Tenant with respect to Tenant’s property and interests, and Tenant shall obtain insurance coverage to the extent Tenant desires protection against such criminal acts and other losses, as further described in Article 22. Tenant agrees to cooperate in any reasonable safety or security program developed by Landlord or required by Law.
Article 22
INSURANCE
1.1Tenant’s Insurance. Tenant shall, during the Term hereof (and during any period that Tenant may enter, occupy and/or use the Premises prior to the Commencement Date and any holdover period), at its sole cost and expense, keep in full force and effect the following insurance:
1.1.1.Property insurance insuring against any perils included within the classification “All Risk,” including, without limitation, fire, windstorm, cyclone, tornado, hail, earthquake, explosion, riot, riot attending a strike, civil commotion, aircraft, vehicle, smoke damage, vandalism, malicious mischief and sprinkler leakage. Such insurance shall insure all property owned by Tenant or any other Tenant Party, for which Tenant or
any other Tenant Party is legally liable or that was installed at the expense of Tenant or any other Tenant Party, and which is located in the Buildings, including, without limitation, furniture, furnishings, installations, fixtures and equipment, any other personal property, and in addition, all improvements and betterments to the Premises, including all Leasehold Improvements, in an amount not less than one hundred percent (100%) of the full replacement cost thereof. For the purposes of this Section 22.1.1, the Premises shall consist of the floor area shown in the Project Site Plan, consisting of the cubic space spanning from the floor slab to the bottom surface of the floor slab of the floor immediately above the Premises (“Upper Slab”), without any offsets or deductions that are included for the Permitted Use of Tenant. Such cubic space shall include the plenum space which is bounded by the lower surface of the Upper Slab and the suspended ceiling of the Premises. Such policy shall name Landlord, any mortgagees of Landlord and any other additional parties designated by Landlord as loss payees, as their respective interests may appear.
1.1.2.Commercial General Liability Insurance insuring Tenant on the current ISO CG 00 01 occurrence form or any equivalent reasonably acceptable to Landlord against any liability arising out of the lease, use, occupancy or maintenance of the Premises, the Buildings or the Project, or any portion of the foregoing. Such insurance shall be in the following minimum limits: $2,000,000 per occurrence and $3,000,000 in the aggregate and shall cover injury (including mental anguish) to or death of one or more persons and damage to tangible property (including loss of use) including blanket contractual liability, broad form property damage (including coverage for explosion, collapse and underground hazards), and $1,000,000 personal & advertising injury. The policy shall insure the hazards of the Premises and Tenant’s operations thereon, Tenant’s independent contractors and Tenant’s contractual liability (including, without limitation, the indemnity contained in Article 20 hereof to the extent commercially available) and shall: (i) name Landlord (10240 Flanders Investors LLC); SRE SD7, LLC; SB Flanders Investors LLC; the Property Manager; any additional entity Landlord may designate from time to time; and their respective partners, parents, affiliates, divisions and subsidiaries, and each of their respective directors, officers, principals, partners, shareholders, members, managing members, agents, employees, successors and assigns (together with Landlord, collectively, “Landlord Indemnified Parties”) as additional insureds; and (ii) include coverage for cross liability claims between Named Insureds (i.e., “Named Insured vs. Named Insured” Cross Liability Coverage Endorsement if required for coverage and no exclusion for cross liability claims between Named Insureds). Such insurance shall not contain any exclusions or restrictions applicable to operations of the type contemplated by this Lease. In addition to any insurance required of Tenant, Tenant shall secure, pay for and maintain or cause Tenant’s contractors and sub-contractors to secure, pay for and maintain insurance during any construction or work to the Premises performed by or on behalf of Tenant at a minimum equal to the limits of liability required by Tenant. Tenant’s products and completed operations insurance shall be maintained for a minimum period equal to the greater of (i) the period under which a claim can be asserted under any applicable statutes of limitations and/or repose or (ii) three (3) years after Substantial Completion of the Tenant Improvements. Tenant’s contractual liability insurance shall include coverage sufficient to meet the indemnity obligations included herein.
1.1.3.Worker’s Compensation Insurance in compliance with statutory requirements of the state(s) in which the employee resides, is hired and in which this Lease takes place, which insurance shall apply to all persons employed by Tenant, and Employer’s Liability insurance in amounts not less than $1,000,000 per accident, $1,000,000 per disease, and $1,000,000 disease-policy limit.
1.1.4.Business interruption insurance and extra expense coverage on ISO coverage form CP 00 30 or equivalent reasonably acceptable to Landlord, which shall cover Tenant’s monetary obligations under this Lease and any direct or indirect loss of earnings attributable to perils insured against in Section 22.1.1 above for a period of at least twelve (12) months. If Tenant fails to obtain business interruption insurance, it is understood and agreed upon that Tenant is fully responsible for its own business interruption exposure whether insured or not.
1.1.5.Comprehensive Automobile Liability Insurance including coverage for all owned, leased, hired and non-owned vehicles, if any, with a minimum combined single limit of $1,000,000 per occurrence for bodily injury and property damage liability.
1.1.6.Umbrella/Excess Liability Insurance policy with a per occurrence and annual aggregate limit of $5,000,000 per location/project. The limits of liability required in Section 22.1.2 above for Commercial General Liability can be provided in a combination of a Commercial General Liability policy and an Umbrella Liability policy. Coverage shall be in excess of Commercial General Liability, Auto Liability and Employers’ Liability insurance with such coverage being on a follow form basis, concurrent to and not more restrictive than underlying insurance. Tenant shall, by specific endorsement to its Umbrella/Excess Liability policy, cause the coverage afforded to the Landlord Indemnified Parties thereunder to be first tier umbrella/excess coverage above the primary coverage afforded to the Landlord Indemnified Parties as set forth in this Lease and not concurrent with or excess to any other valid and collectible insurance available to the Landlord Indemnified Parties whether provided on a primary or excess basis.
1.1.7.If Tenant sells or dispenses alcoholic beverages, Liquor Liability Insurance with limits of not less than $5,000,000 per occurrence.
1.1.8.Pollution Legal Liability insurance if Tenant stores, handles, generates or treats Hazardous Materials, as determined solely by Landlord, on or about the Premises. Such coverage shall include bodily injury, sickness, disease, death or mental anguish or shock sustained by any person; property damage including physical injury to or destruction of tangible property including the resulting loss of use thereof, clean-up costs, and the loss of use of tangible property that has not been physically injured or destroyed; and defense costs, charges and expenses incurred in the investigation, adjustment or defense of claims for such compensatory damages. Coverage shall apply to both sudden and non-sudden pollution conditions including the discharge, dispersal, release or escape of smoke, vapors, soot, fumes, acids, alkalis, toxic chemicals, liquids or gases, waste materials or other irritants, contaminants or pollutants into or upon land, the atmosphere or any watercourse or body of water. Claims-made coverage is permitted, provided the policy retroactive date is continuously maintained prior to the Commencement Date (or such earlier date that Tenant has access to the Premises), and coverage is continuously maintained during all periods in which Tenant occupies the Premises. Coverage shall be maintained with limits of not less than $1,000,000 per incident with a $2,000,000 policy aggregate.
1.1.9.Any other form or forms of insurance as Tenant or Landlord or any mortgagees of Landlord may reasonably require from time to time in form, in amounts and for insurance risks against which a prudent tenant would protect itself.
1.1.10.Tenant may place all or any of the foregoing insurance coverages under blanket insurance policies carried by Tenant provided that no other loss which may also be insured by such blanket insurance shall affect the insurance coverages required hereby and so long as such policy complies with the amount of coverage required hereunder and otherwise provides the same protection as would a separate policy insuring only Tenant’s insurance obligations in compliance with the provisions of Section 22.1 hereof. In addition, Tenant shall deliver to Landlord a certificate specifically stating that such coverages apply to Landlord, the Premises, the Buildings and the Project.
1.1.11.If Tenant shall hire or bring a vendor or contractor onto the Premises to perform any alterations, work or improvements, Tenant agrees to have a written agreement with such vendor or contractor whereby such vendor or contractor will be required to carry the same insurance coverages for Commercial General Liability, Auto and Worker’s Compensation, Employer’s Liability and Pollution Legal Liability insurance as required of Tenant herein. Tenant shall also require that such vendor’s or contractor’s insurance meet the same additional terms as required of Tenant herein with regards to adding the Landlord Indemnified Parties and all mortgagees as additional insureds, maintaining primary and non-contributory coverage, waiving all rights of recovery and subrogation, and making certificates of insurance available as evidence of all policies during the term of their work and in advance of all applicable renewals. Tenant shall not allow any vendors or contractors to begin work prior to obtaining certificates evidencing all insurance requirements contained herein.
1.2Standard of Insurance. All policies shall be written in a form satisfactory to Landlord, and the Commercial General Liability, Comprehensive Automobile Liability, Umbrella/Excess Liability, Liquor Liability (if applicable) and Pollution Legal Liability policies required under Section 22.1 shall name all Landlord Indemnified Parties as additional insureds on a primary and non-contributory basis. In addition, if Tenant places any such required coverages under a blanket insurance policy as set forth in Section 22.1.11, the blanket policy shall name all Landlord Indemnified Parties as additional insureds on a primary and non-contributory basis via blanket endorsement. All insurance policies required under Section 22.1 shall be issued by companies authorized to do business in the State of California with an A.M. Best’s Rating of at least A-/VIII. No deductibles or Self-Insured Retention (“SIR”) of Tenant shall exceed $25,000 (provided, however, the flood insurance may have a deductible of $100,000 and the EQSL insurance may have a deductible of $50,000) without Landlord’s prior written approval. All deductibles and SIR are the responsibility of Tenant and must be shown on the certificate of insurance. On or before the date which is ten (10) days after the execution of this Lease, and prior to or on the renewal of such policies thereafter, Tenant shall deliver to Landlord certificates evidencing the existence of the amounts and forms of coverage satisfactory to Landlord. No such policy shall be cancelable or reducible in coverage except after written notice to Landlord per the policy conditions. Any insurance limits required by this Lease are minimum limits only and not intended to restrict the liability imposed on any Tenant for liability under this Lease. Tenant shall, prior to the expiration of such policies, furnish Landlord with renewals or “binders” thereof, or Landlord may order such insurance and charge the cost thereof to Tenant as Additional Rent. If Landlord obtains any insurance that is the responsibility of Tenant under this Article 22, Landlord shall deliver to Tenant a written statement setting forth the cost of any such insurance and showing in reasonable detail the manner in which it has been computed and Tenant shall remit said amount to Landlord within ten (10) business days.
1.3Landlord Insurance.
1.3.1.During the Term of this Lease, Landlord shall insure the Buildings and the Parking Areas (to the extent Landlord is the owner thereof) (excluding any property which Tenant is obligated to insure under Sections 22.1 and 22.2 hereof) against damage with All-Risk insurance (which may, but shall not be required to, insure against earthquake damage) and public liability insurance, all in such amounts and with such deductibles as Landlord and Tenant jointly and reasonably consider appropriate. Landlord may, but shall not be obligated to,
obtain and carry any other form or forms of insurance as Landlord or Landlord’s mortgagees may determine advisable. Notwithstanding any contribution by Tenant to the cost of insurance premiums, as provided herein, Tenant acknowledges that it has no right to receive any proceeds from any insurance policies carried by Landlord.
1.3.2.If any of Landlord’s insurance policies shall be canceled or cancellation shall be threatened or the coverage thereunder reduced or threatened to be reduced in any way because of Tenant’s specific use of the Premises or any part thereof by Tenant or any assignee or subtenant of Tenant or by anyone Tenant permits on the Premises and, if Tenant fails to remedy the condition giving rise to such cancellation, threatened cancellation, reduction of coverage, threatened reduction of coverage, increase in premiums, or threatened increase in premiums, within three (3)days after Notice thereof, Landlord may, at its option, but without any obligation so to do, enter upon the Premises and attempt to remedy such condition, and Tenant shall promptly pay the cost thereof to Landlord as Additional Rent.
1.4Subrogation Waivers.
1.4.1.Subrogation Waiver – Policies Other than Property Insurance. Tenant hereby waives all rights against the Landlord Indemnified Parties, Landlord’s contractors (and their subcontractors of every tier), and their respective employees and agents, for any claims that arise from Tenant’s work or activities and for recovery of damages under Tenant’s insurance policies required under Section 22.1 or any other insurance policy carried by Tenant related to the Premises or this Lease (excluding Tenant’s property insurance, which is addressed hereunder in Section 22.4.2). Tenant shall obtain an endorsement effecting the foregoing waiver with respect to its workers compensation and employers liability insurance. If any other policy implicated by the waiver in this Section 22.4.1 does not allow the applicable insured party to waive rights of recovery against others prior to a loss, the applicable insured shall obtain an endorsement effecting the applicable waiver.
1.4.2.Subrogation Waiver – Property Insurance. Landlord and Tenant waive all rights against each other for damages caused by fire or other causes of loss occurring on and after the date on which this Lease is executed to the extent such damages are covered (or are required to be covered) by any property insurance required under this Article 22 (including business income and loss of rent insurance) or otherwise carried by such party in relation to the Premises, the Buildings or the Project, regardless of whether such insurance is specifically required under this Lease. Tenant’s waiver in this Section 22.4.2 also extends to the Landlord Indemnified Parties. Each party shall obtain an endorsement pursuant to which its insurers waive their subrogation rights against the parties specified in this Section 22.4.2. The waivers in this Section 22.4.2 will be effective as to a person or entity even though that person or entity would otherwise have a duty of indemnification, did not pay the insurance premium directly or indirectly, or did not have an insurable interest in the property damaged. To the extent that either party self-insures for its insurance obligations under this Lease (e.g., maintains a deductible amount), such party shall be treated as an independent insurer with full waiver of subrogation.
1.5Exclusions. Landlord shall not be liable for any injury or damage to persons or property resulting from fire, explosion, falling plaster, steam, gas, electricity, electrical or electronic emanations or disturbance, water, rain or leaks from any part of the Buildings or from the pipes, or caused by dampness, vandalism, malicious mischief or by any other cause of whatever nature, unless caused by or due to the gross negligence of Landlord, its agents, servants or employees, and then only after (i) reasonable prior notice to Landlord of the condition claimed to constitute negligence, if applicable, and (ii) the expiration of a reasonable time after such notice has been received by Landlord without Landlord having taken all reasonable and practicable means to cure or correct such condition; and pending such cure or correction by Landlord, Tenant shall take all reasonably prudent temporary measures and safeguards to prevent any injury, loss or damage to persons or property. In no event shall Landlord be liable for any loss, the risk of which is covered by Tenant’s insurance or is required to be so covered by this Lease; nor shall Landlord or its agents be liable for any such damage caused by other persons in the Buildings or caused by operations in construction of any private, public, or quasi-public work; nor shall Landlord be liable for any latent defect in the Premises or in the Buildings.
Article 23
DAMAGE OR DESTRUCTION
1.1Damages. If the Premises are damaged by fire or other perils covered by Landlord’s insurance, Landlord shall:
1.1.1.In the event of one hundred percent (100%) destruction of the Premises (“Total Destruction”), at Landlord’s option, as soon as reasonably possible thereafter, commence repair, reconstruction and restoration of the Premises and prosecute the same diligently to completion, in which event this Lease shall remain in full force and effect; provided, however, that if within ninety (90) days after the occurrence of such damage, Landlord shall by Notice to Tenant elect not to so repair, reconstruct or restore the Premises, this Lease shall terminate as of the date of such Total Destruction.
1.1.2.In the event of a partial destruction of the Premises and if the damage thereto is such that the Premises is capable of being repaired, reconstructed or restored within a period of ninety (90) days from the date of Landlord’s discovery of such damage, and if Landlord will receive insurance proceeds sufficient to cover the total cost of such repairs, reconstruction or restoration, Landlord shall commence and proceed diligently with the work of repairs, reconstruction and restoration of the Premises and this Lease shall continue in full force and effect. If such work of repair, reconstruction and restoration shall require a period longer than ninety (90) days or exceeds twenty-five percent (25%) of the full replacement cost of the Premises, or if insurance proceeds will not be sufficient to cover the cost of such repairs, reconstruction and restoration, then Landlord either may elect to so repair, reconstruct or restore and this Lease shall continue in full force and effect or may elect not to repair, reconstruct or restore and this Lease shall then terminate as of the date of such partial destruction. Under any of the conditions of this Section 23.1.2, Landlord shall give Notice to Tenant of its intention regarding repairs, including Landlord’s good faith estimate of the time required to substantially complete such repairs (“Landlord’s Repair Notice”), within said ninety (90) day period. Notwithstanding anything to the contrary set forth herein, if the partial destruction (or if there is 100% destruction referenced in Section 23.1.1) causes more than fifty percent (50%) of the Rentable Square Feet of the Premises to be unusable for the Permitted Use and Landlord’s Repair Notice estimates that the repairs will take more than one hundred eighty (180) days after the occurrence thereof to substantially complete, and if Landlord does not elect to terminate this Lease, then Tenant shall have the right to terminate this Lease by Notice to Landlord delivered within ten (10) business days following delivery of Landlord’s Repair Notice, which termination shall be effective no earlier than thirty (30) days after delivery of such Notice to Landlord. If damage is due to any cause other than fire or other peril covered by extended coverage insurance, Landlord may elect to terminate this Lease.
1.1.3.In any case where Landlord elects to repair, restore or reconstruct the Premises following the occurrence of any damage to which this Article 23 applies, then Tenant shall assign to Landlord the proceeds of its property insurance attributable to the Leasehold Improvements. If the cost of restoring the Leasehold Improvements exceeds the amount of the proceeds of Tenant’s property insurance that are received by Landlord, Tenant shall promptly pay the amount of such deficiency to Landlord upon demand.
1.2Termination of Lease. Upon any termination of this Lease under any of the provisions of this Article 23, the parties shall be released without further obligation to the other from the date possession of the Premises is surrendered to Landlord except for items which have therefore accrued and/or are then unpaid or items which expressly survive the expiration or sooner termination of this Lease.
1.3Rent Abatement. In the event of any casualty, the Rent payable under this Lease shall be abated proportionately with the degree to which Tenant’s Permitted Use of the Premises is impaired either during the period of such repair, reconstruction or restoration or until termination of the Lease pursuant to this Article 23, but only to the extent that Landlord is compensated for such loss by the insurance carried or required to be carried pursuant to Section 22.1.4 above. Notwithstanding the foregoing, there shall be no abatement of Rent if such damage is caused primarily by the negligence or intentional wrongdoing of Tenant or any Tenant Party. Tenant shall not be entitled to any compensation or damages for loss in the use of the whole or any part of the Premises and/or any inconvenience or annoyance occasioned by such damage, repair, reconstruction or restoration. If Landlord is obligated to or elects to repair or restore as herein provided, Landlord shall be obligated to repair or restore only those portions of the Premises which were originally provided at Landlord’s expense, and the repair and restoration of items not provided at Landlord’s expense shall be the obligation of Tenant.
1.4Damage Near End of Term. Notwithstanding anything to the contrary contained in this Article 23, if material damage to the Premises occurs during the last twelve (12) months of the Term, either party may elect, no earlier than sixty (60) days after the date of the damage and not later than ninety (90) days after the date of such damage, to terminate this Lease by written notice to the other effective as of the date specified in the notice, which date shall not be less than ten (10) business days nor more than sixty (60) days after the date such notice is given.
1.5Waiver of Statute. In the event of damage to the Premises, Tenant shall not be released from any of its obligations under this Lease except to the extent and upon the conditions expressly stated in this Article 23. Tenant hereby waives the provisions of California Civil Code Section 1932, Subsection 2, and Section 1933, Subsection 4, and any other statute or court decision relating to the abatement or termination of a lease upon destruction of the Premises and the provisions of this Article 23 shall govern in case of such destruction.
Article 24
EMINENT DOMAIN
1.1Permanent Taking. If all of the Premises, or such part thereof as shall substantially interfere with Tenant’s Permitted Use and occupancy thereof, shall be taken for any public or quasi-public purpose by any lawful power or authority by exercise of the right of appropriation, condemnation or eminent domain, or sold to prevent such taking (a “Taking”), either party shall have the right to terminate this Lease by Notice to the other effective as of the date possession is required to be surrendered to said authority. Tenant shall not assert any claim against Landlord or the taking authority for any compensation because of such Taking, and Landlord shall be entitled to
receive the entire amount of any award without deduction for any estate or interest of Tenant. If the amount of property or the type of estate taken shall not substantially interfere with the conduct of Tenant’s business, Landlord shall be entitled to the entire amount of the award without deduction for any estate or interest of Tenant, Landlord shall restore the Premises to substantially their same condition prior to such partial Taking, and Basic Rent shall be reduced, effective as of the date the condemning authority takes possession, in the same proportion which the Rentable Square Feet of the portion of the Premises so taken bears to the Rentable Square Feet of the entire Premises before the Taking. Nothing contained in this Section 24.1 shall be deemed to give Landlord any interest in any award made to Tenant for the taking of personal property and fixtures belonging to Tenant or for relocation costs and expenses.
1.2Temporary Taking. Notwithstanding anything to the contrary in Section 24.1 above, in the event of Taking of the Premises or any part thereof for temporary use, (a) this Lease shall be and remain unaffected thereby and Rent shall not abate, and (b) Tenant shall be entitled to receive for itself such portion or portions of any award made for such use with respect to the period of the Taking which is within the Term, provided that if such Taking shall remain in force at the expiration or earlier termination of this Lease, Tenant shall then pay to Landlord a sum equal to the reasonable cost of performing Tenant’s obligations under Section 15.2 above and Article 31 below with respect to surrender of the Premises and, upon such payment, shall be excused from such obligations. For purpose of this Article 24, a “temporary” Taking shall be defined as a Taking for a period of two hundred seventy (270) days or less and a “permanent” Taking shall be defined as a Taking for a period of more than two hundred seventy (270) days.
1.3Waiver of Statute. Tenant (for itself and all others claiming through Tenant) hereby irrevocably waives and releases its rights under Section 1265.130 of the California Code of Civil Procedure.
Article 25
DEFAULTS AND REMEDIES
1.1Tenant Default. The occurrence of any one or more of the following events, upon the expiration of any applicable time period, shall constitute a default hereunder by Tenant (“Tenant Default”):
1.1.1.Abandonment of the Premises by Tenant. Notwithstanding the provisions of California Civil Code Section 1951.3, “Abandonment” is defined to include, but not limited to, any absence by Tenant from the Premises for thirty (30) days or longer while in default pursuant to any other provision of this Section 25.1;
1.1.2.The failure by Tenant to make any payment of Rent or any other payment required to be made by Tenant hereunder, as and when due, where such failure shall continue for a period of three (3) business days after Landlord’s delivery of Notice thereof;
1.1.3.The failure by Tenant to obtain and keep in force at all times any insurance Tenant is required to obtain and keep in force under Article 22 where such failure is not cured within five (5) business days after Landlord’s delivery of Notice of such failure;
1.1.4.Hypothecation, assignment or other transfer of this Lease or subletting of the Premises, or attempts of such actions in violation of Article 27 of this Lease;
1.1.5.The failure by Tenant to deliver any certificate, instrument or statement that is required to be delivered by Tenant under Article 28, Article 29 or Section 36.16 within the time frames required in Article 28, Article 29 or Section 36.16, as applicable, which Tenant fails to cure within five (5) business days after Landlord’s delivery of Notice thereof;
1.1.6.The failure by Tenant to observe or perform any of the express or implied covenants or provisions of this Lease to be observed or performed by Tenant, other than as specified in Sections 25.1.1 – 25.1.5 above or Section 25.1.7 below, where such failure shall continue for a period of thirty (30) days after Landlord’s delivery of Notice thereof; provided that if the nature of any such failure is such that more than thirty (30) days are reasonably required for its cure, then no Tenant Default shall be deemed to occur if (and for so long as) Tenant commences the cure of such failure within said thirty (30) day period and thereafter diligently prosecutes such cure to completion within ninety (90) days after Landlord’s delivery of Notice thereof; or
1.1.7.The (a) making by Tenant of any general assignment for the benefit of creditors; (b) filing by or against Tenant of a petition to have Tenant adjudged a bankrupt or a petition for reorganization or arrangement under any Law relating to bankruptcy; (c) appointment of a trustee or receiver to take possession of substantially all of Tenant’s assets located at the Premises or of Tenant’s interest in this Lease; (d) attachment, execution or other judicial seizure of substantially all of Tenant’s assets located at the Premises or of Tenant’s interest in this Lease; or (f) Tenant’s convening of a meeting of its creditors or any class thereof for the purpose of effecting a moratorium upon or composition of its debts, or any class thereof; provided that no Tenant Default will be deemed to occur under this Section 25.1.7 if (i) any petition described in clause (a) above that filed against (rather than by) Tenant, is
dismissed within thirty (30) days) after filing, (ii) in the event any trustee or receiver shall take possession of substantially all of Tenant’s assets located at the Premises or Tenant’s interest in this Lease, possession of the same is restored to Tenant within thirty (30) days or (iii) any attachment, execution or other judicial seizure described in clause (d) above is discharged within thirty (30) days.
Any Notice from Landlord required hereby shall be in lieu of, and not in addition to, any Notice required under California Code of Civil Procedure Section 1161 regarding unlawful detainer actions or any similar successor statute. Accordingly, Tenant (for itself and all others claiming through Tenant) hereby expressly and irrevocably waives the notice requirements of California Code of Civil Procedure Section 1162 that would otherwise govern notices required under Section 1161, and agrees that any notice provided pursuant to this Section 25.1 shall replace and satisfy any such requirements of Section 1162.
1.2Landlord Remedies. In the event of any such Tenant Default, in addition to any other remedies available to Landlord at law or in equity, including, without limitation, the remedies available under California Civil Code Section 1951.2 and any successor statute, Landlord shall have the immediate option to terminate this Lease and all rights of Tenant hereunder. In the event that Landlord shall elect to so terminate this Lease then Landlord may recover from Tenant:
1.2.1.The worth at the time of award of any unpaid Rent which had been earned at the time of such termination; plus
1.2.2.the worth at the time of award of the amount by which the unpaid Rent which would have been earned after termination until the time of award exceeds the amount of such Rent loss that Tenant proves could have been reasonably avoided; plus
1.2.3.the worth at the time of award of the amount by which the unpaid Rent for the balance of the Term after the time of award exceeds the amount of such Rent loss that Tenant proves could be reasonably avoided; plus
1.2.4.any other amount necessary to compensate Landlord for all the detriment proximately caused by Tenant’s failure to perform Tenant’s obligations under this Lease or which in the ordinary course of things would be likely to result therefrom, including but not limited to the cost of recovering possession of the Premises, expenses of reletting, including necessary repair, renovation and alteration of the Premises, reasonable attorneys’ fees and any other reasonable costs; and
1.2.5.at Landlord’s election, such other amounts in addition to or in lieu of the foregoing as may be permitted from time to time by applicable Law.
As used in Sections 25.2.1 and 25.2.2 above, the “worth at the time of award” is computed by allowing interest at the Default Rate. As used in Section 25.2.3 above, the “worth at the time of award” is computed by discounting such amount at the discount rate of the Federal Reserve Bank of San Francisco (“Discount Rate”) at the time of award plus one percent (1%). If the format or components of the Discount Rate are materially changed, or if the Discount Rate ceases to exist, Landlord shall substitute a discount rate which is maintained by the Federal Reserve Bank of San Francisco or similar financial institution and which is most nearly equivalent to the Discount Rate.
1.3Additional Remedies. If any such Tenant Default occurs, Landlord may utilize the remedy described in California Civil Code Section 1951.4 (which provides landlord may continue the lease in effect after a tenant’s breach and abandonment and recover Rent as it becomes due, if tenant has the right to sublet or assign subject to reasonable limitations). Accordingly, in the event of any Tenant Default and abandonment of the Premises by Tenant, if Landlord does not elect to terminate this Lease on account of such Tenant Default, then Landlord may from time-to-time, without terminating this Lease, enforce all of its rights and remedies under this Lease, including the right to recover all Rent as it becomes due. In the event of the Abandonment of the Premises by Tenant or in the event that Landlord utilizes the remedy described in this Section 25.3 above or shall take possession of the Premises pursuant to legal proceeding or pursuant to any notice provided by Law, then if Landlord does not elect to terminate this Lease as provided above, Landlord may from time to time, without terminating this Lease, either recover all Rent as it becomes due or relet the Premises or any part thereof for the Term of this Lease on terms and conditions as Landlord in its sole discretion may deem advisable with the right to make alterations and repairs to the Premises.
If Landlord shall elect to so relet, such reletting shall not relieve Tenant of any obligation hereunder, except that the rents received by Landlord from such reletting shall be applied as follows: (a) first, to the payment of any indebtedness other than Rent due hereunder from Tenant to Landlord; (b) second, to the payment of any cost of such reletting; (c) third, to the payment of the cost of any alterations and repairs to the Premises; (d) fourth, to the payment of Rent due and unpaid hereunder and (e) the residue, if any, shall be held by Landlord and applied to payment of future Rent as the same may become due and payable hereunder. Should that portion of such rents
received from such reletting during any month, which is applied to the payment of Rent hereunder, be less than the Rent payable during that month by Tenant hereunder, then Tenant shall pay such deficiency to Landlord immediately upon demand therefor by Landlord. Such deficiency shall be calculated and paid monthly. Tenant shall also pay to Landlord, as soon as ascertained, any costs and expenses, including attorneys’ fees, incurred by Landlord in such reletting or in making such alterations and repairs not covered by the rents received from such reletting. During the continuance of a Tenant Default, Landlord shall have the right to market the Premises to potential new tenants and may show the Premises to such potential new tenants during normal business hours.
1.4Notice of Default. Tenant hereby acknowledges that default by Tenant hereunder, and Landlord’s election to prepare and serve a Notice of any such default hereunder (a “Notice of Default”), will cause Landlord to incur costs not contemplated by this Lease, and costs in addition to any costs which may be reimbursed to Landlord by any provision which may be contained herein relative to the payment of interest or late charges on amounts due hereunder. Accordingly, Landlord shall be entitled to reasonable attorneys’ fees and all other costs and expenses incurred in the preparation and service of a Notice of Default and consultations in connection therewith, with respect to which Landlord and Tenant agree that Five Hundred Dollars ($500.00) is a reasonable minimum sum per such occurrence, whether or not legal action is subsequently commenced in connection with any such default. It is further hereby specifically agreed by and between Landlord and Tenant that any and all such fees and costs shall be deemed Additional Rent hereunder, and may, at the option of Landlord, be included in any Notice of Default hereunder.
1.5Landlord’s Right to Cure. If Tenant should fail to make any payment or perform any of its other obligations hereunder, Landlord, without being under any obligation to do so and without thereby waiving such default, may make such payment and/or remedy such other default for the account of Tenant (and enter the Premises for such purpose): (a) immediately and without notice in the case: (i) of emergency, (ii) of a default by Tenant of its obligations under Section 8.3, Section 15.2 and/or Article 31, (iii) where such default unreasonably interferes with any other tenant in the Project, (iv) a failure to satisfy or otherwise discharge any lien, or (v) where such default will result in the violation of Law or the cancellation of any insurance policy maintained by Landlord and (b) in any other case if such default continues beyond the applicable notice and cure period specified in Section 25.1 above, and thereupon Tenant shall be obligated to, and hereby agrees to pay Landlord, upon demand, all costs, expenses, and disbursements incurred by Landlord in taking such remedial action, together with an amount equal to five percent (5%) thereof for Landlord’s overhead and administrative expenses, and the sum of such costs, together with interest thereon at the rate described in Section 5.3 from the date of Landlord’s payment thereof, shall be deemed Additional Rent.
1.6Waiver of Redemption. Tenant (for itself and all others claiming through Tenant) hereby irrevocably waives and releases its rights to redemption and reinstatement under any present or future case law or statutory provision (including, without limitation, Sections 473, 1174 and 1179 of the California Code of Civil Procedure and Section 3275 of the California Civil Code) in the event that Tenant is dispossessed from the Premises for any reason.
1.7Landlord’s Default. Landlord’s failure to perform or observe any of its obligations under this Lease shall constitute a default by Landlord under this Lease (a “Landlord Default”) only if Landlord, or the Holder (defined below) of any Security Instrument (defined below) covering the Premises, fails to perform obligations required of Landlord within thirty (30) days after Notice by Tenant to Landlord (and to each Holder pursuant to Section 36.5 below), specifying wherein Landlord has failed to perform such obligations in reasonable detail; provided, however, that if the nature of Landlord’s obligation is such that more than thirty (30) days are required for performance, then no Landlord Default shall occur if Landlord commences performance within such thirty (30) day period and thereafter diligently prosecutes the same to completion (or if any Holder of any Security Instrument commences and prosecutes the cure pursuant to Section 36.5 below). In no event shall Tenant be entitled to terminate this Lease by reason of any Landlord Default, and Tenant’s remedies shall be limited to an action for monetary damages at law. Without limiting the foregoing, in recognition that Landlord must receive timely payments of Rent and operate the Project, Tenant shall have no right of self-help to perform repairs or any other obligation of Landlord and, except as expressly provided in Articles 23 and 24, shall have no right to withhold, set-off, or abate Rent.
Article 26
NO WAIVER
All rights, options and remedies of Landlord contained in this Lease shall be construed and held to be cumulative, and not one of them shall be exclusive of the other, and Landlord shall have the right to pursue any one or all of such remedies or any other remedy or relief which may be provided by Law, whether or not stated in this Lease. The waiver by Landlord of any breach of any term, covenant or condition herein contained shall not be deemed to be a waiver of any subsequent breach of the same or any other term, covenant or condition herein contained, nor shall any custom or practice which may grow up between the parties in the administration of the terms hereof be deemed a waiver of or in any way affect the right of Landlord to insist upon the performance by Tenant in strict accordance with said terms. The subsequent acceptance of Rent hereunder by Landlord shall not be
deemed to be a waiver of any preceding breach by Tenant of any term, covenant or condition of this Lease, other than the failure of Tenant to pay the particular Rent so accepted, regardless of Landlord’s knowledge of such preceding breach at the time of acceptance of such Rent. No acceptance by Landlord of a lesser sum than the Basic Rent and Additional Rent or other sum then due shall be deemed to be other than on account of the earliest installment of such Rent or other amount due, nor shall any endorsement or statement on any check or any letter accompanying any check be deemed an accord and satisfaction, and Landlord may accept such check or payment without prejudice to Landlord’s right to recover the balance of such installment or other amount or pursue any other remedy provided in this Lease. Without limiting the foregoing, Tenant (for itself and all others claiming through Tenant) acknowledges that this Article 26 imparts actual notice to Tenant, pursuant to California Code of Civil Procedure Section 1161.1(c), that Landlord’s acceptance of partial payment of Rent shall not constitute a waiver of any rights available under this Lease or at law or equity, including, without limitation, the right to recover possession of the Premises.
Article 27
ASSIGNMENT AND SUBLETTING
1.1Transfer. Tenant shall not voluntarily or by operation of law: (a) sublease all or any part of the Premises (“Sublease”), (b) assign this Lease (“Assignment”), or (c) enter into any other agreement or arrangement: (i) that permits a third party (other than Tenant’s employees and occasional guests) to enter, occupy or use any portion of the Premises or remove any of Tenant’s Personal Property therefrom or (ii) otherwise assigns, transfers, mortgages, pledges, hypothecates, encumbers or permits a lien to attach to Tenant’s interest under this Lease or in the Premises (each of the foregoing (a), (b) and (c), a “Transfer”), without first obtaining Landlord’s prior written consent in accordance with this Article 27. In addition, for purposes of this Lease a “Transfer” (which shall be subject to the provisions of this Article 27) shall also include the dissolution of the entity that constitutes Tenant without its immediate reconstitution. No consent to an assignment, encumbrance or sublease shall constitute a waiver of any provision of this Article 27 or consent to any future assignment, encumbrance or transfer. Any Transfer without Landlord’s prior written consent shall be voidable at Landlord’s election and shall constitute a Tenant Default.
1.2Transfer Procedure. If Tenant desires to make any Transfer, then at least thirty (30) days prior to the date when Tenant desires the Transfer to be effective (“Transfer Date”) Tenant shall give Landlord a Notice (“Transfer Notice”) setting forth: (a) the name, address and business of the person or entity to which the Transfer is proposed (“Proposed Transferee”); (b) information (including references) concerning the character, ownership and financial condition of the Proposed Transferee; (c) the proposed Transfer Date (which shall not be later than 90 days following the Transfer Notice); (d) any ownership or commercial relationship between Tenant and the Proposed Transferee; and (e) the consideration and all other material terms and conditions of the proposed Transfer, all in such detail as Landlord shall reasonably require. If Landlord reasonably requests additional detail (including, without limitation, financial statements of the proposed Transferee or a current estoppel certificate from Tenant), the Transfer Notice shall not be deemed to have been received until Landlord receives such additional detail, and Landlord may withhold consent to any proposed Transfer until such information is provided to it.
1.3Recapture. Within thirty (30) days of Landlord’s receipt of a Transfer Notice, and all information specified in Section 27.2 above, Landlord may, at its option, in its sole and absolute discretion, by Notice to Tenant (“Recapture Notice”), elect to: (a) in the case of a proposed Sublease, sublease the Premises or the portion thereof proposed to be sublet by Tenant at a rental rate per square foot equal to the lesser of the per square foot rental rate under this Lease or the proposed Sublease; (b) in the case of a proposed Assignment, take an assignment of this Lease upon the same terms as those offered to the proposed assignee; or (c) terminate this Lease in its entirety or as to the portion of the Premises subject to the proposed Transfer. Tenant shall have the right, by Notice to Landlord no later than five (5) business days following Landlord’s delivery of a Recapture Notice, to withdraw the subject Transfer Notice, in which event such Recapture Notice shall have no force or effect. Tenant’s failure to deliver such Notice to withdraw the subject Transfer Notice within such five (5) business day period shall be deemed a waiver of its right to withdraw such Transfer Notice and the parties shall proceed pursuant to the Recapture Notice. If Landlord elects to proceed pursuant to clause (a) or (b) above (and Tenant does not timely withdraw the subject Transfer Notice), any payment by Landlord to Tenant pursuant to such clause shall not exceed the amount which Tenant would have received pursuant to Section 27.5.2 below if Landlord had elected to consent to the proposed Sublease or Assignment. If this Lease shall be terminated with respect to the entire Premises, the Term shall end on the Transfer Date as if that date had been originally fixed in this Lease for the expiration of the Term. If Landlord recaptures only a portion of the Premises, the Rent during the unexpired Term and Tenant’s Percentage shall be adjusted proportionately based on the Rentable Square Feet remaining in the Premises after such recapture. Tenant shall, at Tenant’s own cost and expense, discharge in full any commissions which may be due and owing as a result of any proposed assignment or subletting, whether or not the Premises (or portion thereof) are recaptured pursuant to this Section 27.3 and rented by Landlord to the proposed tenant or any other tenant. Notwithstanding the foregoing, Landlord’s rights set forth in this Section 27.3 shall not apply to a Sublease of a portion of the Premises to a party
who is acquiring a business segment located in such subleased portion of the Premises from Tenant or an affiliate of Tenant.
1.4Landlord’s Consent; Consent Standards; No Release.
1.4.1.Unless Landlord elects to exercise any of its rights under Section 27.3 above, Landlord shall, by Notice to Tenant, elect to: (a) consent to such proposed Transfer upon the terms and to the Proposed Transferee; or (b) refuse to give its consent to the proposed Transfer. Landlord shall not unreasonably withhold its consent to any Proposed Transfer; provided that, without limiting other situations in which it may be reasonable for Landlord to withhold its consent to any proposed Transfer, it shall be deemed reasonable for Landlord to withhold its consent to any proposed Transfer if Landlord determines in its sole discretion that: (i) the Proposed Transferee, in the case of an Assignment of this Lease or a Sublease of more than fifty percent (50%) of the Rentable Square Feet of the Premises, does not have sufficient financial strength or stability to perform all obligations under this Lease, and to perform them without any higher risk of default than Tenant; (ii) the intended use of the Premises (or the applicable portion thereof) by the Proposed Transferee is inconsistent or incompatible with other uses in the Project; (iii) the intended use of the Premises (or the applicable portion thereof) by the Proposed Transferee will require alteration of the Premises; (iv) the intended use of the Premises (or the applicable portion thereof) by the Proposed Transferee will violate this Lease or any Laws governing the Premises or the Project; (v) the Proposed Transferee has the power of eminent domain, is a Governmental Authority or an agency or subdivision of a foreign government; (vi) either the Proposed Transferee, or any person which directly or indirectly controls, is controlled by, or is under common control with the Proposed Transferee: (A) occupies space in the Project or has negotiated with Landlord or any of its affiliates within the preceding one hundred eighty (180) days (or is currently negotiating with Landlord or any of its affiliates) to lease space in the Project or (B) does not intend to occupy the Premises or the applicable portion thereof; (vii) at the time Tenant delivers the Transfer Notice, there exists an uncured Tenant Default; (viii) the proposed Transfer would cause Landlord to be in violation of another lease or agreement to which Landlord is a party or would give an occupant of the Project a right to cancel or modify its lease; (ix) any ground lessor or mortgagee whose consent to such Transfer is required fails to consent thereto; (x) the use of the Premises (or the applicable portion thereof) or the Project by the Proposed Transferee would, in Landlord’s judgment, significantly increase pedestrian traffic in and out of the Project, generate increased loitering in Common Areas, increase security risk, or require any alterations to the Project to comply with applicable Laws; (xi) the Proposed Transferee would be a competitor to another tenant in the Project in violation of such other tenant’s lease; (xii) the Proposed Transferee has been required by any prior landlord, lender or Governmental Authority to take material remedial action in connection with Hazardous Materials contaminating a property, which contamination resulted from Proposed Transferee’s action or omission or use of the property in question; or (xiii) the Proposed Transferee is subject to a material enforcement order issued by any Governmental Authority in connection with the use, disposal or storage of Hazardous Materials.
1.4.2.Tenant further agrees that Landlord may condition its consent to any proposed Transfer upon satisfaction of any of the following conditions: (a) delivery to Landlord of a true copy of a fully executed sublease, assignment of lease or other instrument pursuant to which the applicable Transfer is made (“Transfer Instrument”); (b) delivery to Landlord of original executed copies (by Tenant and the Transferee (defined below)) of Landlord’s form of Consent to Sublease (in the case of a Sublease) or Assignment and Assumption of Lease and Consent (in the case of an Assignment) or other instrument under which Landlord grants consent to the applicable Transfer (“Consent Instrument”) and (c) receipt by Landlord of all sums and amounts to which Landlord is entitled under Section 27.5 below. Tenant acknowledges and agrees that any Consent Instrument may, without limitation: (i) in the case of a Sublease or Assignment, require the person or entity to which the Transfer is made (“Transferee”) to be bound by all of the terms and provisions of this Lease and to perform all of the obligations of Tenant hereunder applicable to the Premises, or the portion thereof that is the subject of the applicable Transfer; (ii) in the case of an Assignment, include waivers by Tenant of all applicable suretyship defenses, including, but not limited to, those contained in Sections 2787 to 2855, inclusive, of the California Civil Code; and (iii) in the case of a Sublease: (A) provide that such Sublease is subject and subordinate to this Lease to all Security Instruments encumbering the Buildings or the Project, (B) require the Transferee to, upon demand by Landlord following the occurrence of any Tenant Default, remit directly to Landlord, all monies payable from such Transferee to Tenant in connection with such Sublease and (C) provide that in the event of termination of this Lease for any reason, including without limitation a voluntary surrender by Tenant, or in the event of any reentry or repossession of the Premises by Landlord, Landlord may, at its option, either: (x) terminate the sublease or (y) take over all of the right, title and interest of Tenant, as sublessor, under such sublease, in which case such sublessee shall attorn to Landlord, but that nevertheless Landlord shall not: (1) be liable for any previous act or omission of Tenant under such sublease, (2) be subject to any defense or offset previously accrued in favor of the sublessee against Tenant, or (3) be bound by any previous modification of any sublease made without Landlord’s written consent, or by any previous prepayment by sublessee of more than one month’s rent.
1.4.3.If Landlord grants its consent to any proposed Transfer described in any Transfer Notice, Tenant may during the thirty (30) days thereafter consummate such Transfer with the Proposed Transferee upon the terms and conditions described in the applicable Transfer Notice; provided, however, that any material change in
such terms shall be subject to Landlord’s consent as provided in this Article 27. No Assignment or Sublease or other Transfer (whether with or without Landlord’s consent) shall relieve Tenant or any assignee or sublessee from any obligation under this Lease whether or not accrued as of the date of the Assignment or Sublease (and, to the extent such Tenant is deemed a surety of an assignee, Tenant hereby waives all applicable suretyship defenses, including, but not limited to, those contained in Sections 2787 to 2855, inclusive, of the California Civil Code.
1.5Landlord’s Costs; Transfer Premiums.
1.5.1.If Tenant requests Landlord’s consent to a proposed Transfer under the provisions of this Article 27, Tenant shall, upon demand, reimburse all of Landlord’s reasonable expenses, costs and attorneys’ fees incurred in connection with processing such request for consent, whether or not Landlord grants consent to such proposed Transfer.
1.5.2.If Landlord consents to a Transfer, Tenant shall pay to Landlord fifty percent (50%) of any rent or other consideration realized by Tenant pursuant to such Transfer in excess of (i) the Rent payable by Tenant under this Lease, (ii) any reasonable tenant improvement allowance or other economic concession (e.g., space planning allowance, moving expenses, free or reduced rent periods, etc.) actually incurred by Tenant in connection with such Transfer, (iii) any reasonable advertising costs and brokerage commissions actually incurred by Tenant in connection with such Transfer, and (iv) any reasonable legal fees actually incurred by Tenant in connection with such Transfer.
1.6Rights Not Transferable. All: (a) options to extend or renew the Term and/or to expand the Premises, if any, contained in this Lease or any addendum or amendment hereto or letter of agreement; (b) all rights to any signage at the Project in any location outside of the Premises, if any, contained in this Lease or any addendum or amendment hereto or letter of agreement; (c) all rights to above standard (or discounted) parking at the Project, if any, contained in this Lease or any addendum or amendment hereto or letter of agreement; and (d) all rights to receive any above standard services or utilities, if any, contained in this Lease or any addendum or amendment hereto or letter of agreement, are personal to the Original Tenant (or a Permitted Transferee), and may not be transferred in connection with any Transfer or exercised by any Transferee. Consent by Landlord to any Transfer shall not include consent to the assignment or transfer of any such options, rights or privileges (and such options, rights, or privileges shall terminate upon such assignment or subletting), unless Landlord, in its sole and absolute discretion, specifically grants in writing such options, rights, privileges or services to such assignee or subtenant.
1.7Affiliate Transfers. Notwithstanding anything to the contrary contained in this Article 27, (a) any Transfer to an affiliate of Tenant (an entity which is controlled by, controls, or is under common control with Tenant), (b) any Transfer to an entity which acquires all or substantially all of the assets or interests (partnership, stock or other) of Tenant, or (c) any Transfer to an entity which is the resulting entity of a merger or consolidation of Tenant, shall not be deemed a Transfer under this Article 27, provided that (i) in the case of an assignment of this Lease or a sublease of more than fifty percent (50%) of the Rentable Square Feet of the Premises, the financial credit of such transferee entity is, in Landlord’s reasonable judgment, the same or greater than that of Tenant both as of the Effective Date of this Lease and as of the date of the proposed transfer; (ii) Tenant notifies Landlord of such transfer within thirty (30) days thereof and promptly thereafter supplies Landlord with any documents or information reasonably requested by Landlord regarding such transfer or such affiliate; and (iii) such transfer is not a subterfuge by Tenant or Guarantor to avoid its obligations under this Lease or otherwise effectuate any “release” by Tenant or Guarantor of such obligations. A transfer made in accordance with this Section 27.7 shall be referred to as a “Permitted Transfer” and the transferee shall be referred to as a “Permitted Transferee.” “Control,” as used in this Section 27.7, shall mean the ownership, directly or indirectly, of more than fifty percent (50%) of the voting securities of, or possession of the right to vote, in the ordinary direction of its affairs, of more than fifty percent (50%) of the voting interest in, any person or entity. No assignment or sublease under this Section 27.7 shall relieve Tenant from any of its obligations under this Lease whether or not accrued as of the date of such assignment or sublease.
Article 28
SUBORDINATION
Without the necessity of any additional documents being executed by Tenant for the purpose of effecting a subordination, and at the election of Landlord, or any current or future mortgagee or holder of deed of trust with a lien on the Buildings or the Project or any ground lessor with respect to the Buildings or the Project (each, a “Holder”), this Lease shall be subject and subordinate at all times to: (a) all ground leases or underlying leases which may now exist or hereafter be executed affecting the Building, the Project, or the land upon which the Buildings and the Project are situated, or both; and (b) the lien of any mortgage or deed of trust which may now exist or hereafter be executed in any amount for which the Buildings, the Project, the land upon which the Buildings and the Project are situated, ground leases or underlying leases, or Landlord’s interest or estate in any of said items is specified as security (collectively, “Security Instruments”). With respect to any Security Instrument existing as of the Commencement Date, Landlord shall use commercially reasonable efforts to cause the Holder thereof to enter
into a subornation, non-disturbance and attornment agreement concurrently with the execution of this Lease. Notwithstanding the foregoing, Landlord shall have the right to subordinate or cause to be subordinated such ground leases or any such liens to this Lease. In the event that any ground lease or underlying lease terminates for any reason or any mortgage or deed of trust is foreclosed or a conveyance in lieu of foreclosure is made for any reason, Tenant shall, notwithstanding any subordination, attorn to and become the tenant of the successor-in-interest to Landlord, at the option of such successor-in-interest to Landlord provided that such successor-in-interest to Landlord agrees to not disturb the tenancy of Tenant pursuant and subject to the terms and conditions of this Lease. Tenant covenants and agrees to execute and deliver, within ten (10) business days after demand by Landlord therefor, any additional commercially reasonable documents evidencing the priority or subordination of this Lease with respect to any such Security Instruments.
Article 29
ESTOPPEL CERTIFICATE
1.1Tenant Estoppel Certificate. Within ten (10) business days following any written request which Landlord may make from time to time, Tenant shall execute and deliver to Landlord a statement, in a form substantially similar to the form of Exhibit “E” attached hereto, and incorporated herein by this reference (a “Tenant Estoppel Certificate”) certifying: (a) the Commencement Date of this Lease; (b) that this Lease is unmodified and in full force and effect (or, if there have been modifications hereto, that this Lease is in full force and effect, and stating the date and nature of such modifications); (c) the date to which the Rent and other sums payable under this Lease have been paid; (d) that to the best of Tenant’s knowledge, there are no then current defaults under this Lease by Landlord except as specified in Tenant’s statement; (e) that Tenant has received no written notice that there are any then current defaults under this Lease by Tenant except as specified in Tenant’s statement; and (f) such other matters as are reasonably included in such statement by Landlord. Landlord and Tenant intend that any statement delivered pursuant to this Article 29 may be relied upon by any mortgagee, lessor, beneficiary, purchaser or prospective purchaser of the Buildings or the Project or any interest therein.
1.2Failure to Deliver. Tenant’s failure to deliver such statement within such time shall be conclusive upon Tenant: (a) that this Lease is in full force and effect, without modification except as may be represented by Landlord, (b) that there are no uncured defaults in Landlord’s performance, (c) that not more than one (1) month’s Rent has been paid in advance and (d) that the statements included in the Tenant Estoppel Certificate are true and correct, without exception. Additionally, any such failure to timely deliver a Tenant Estoppel Certificate shall constitute an immediate Tenant Default hereunder.
Article 30
INTENTIONALLY OMITTED
Article 31
SURRENDER OF PREMISES
Upon the expiration or earlier termination of the Term hereof, Tenant shall peaceably surrender the Premises and all Leasehold Improvements therein, excepting only any of the same that are required to be removed in accordance with Section 15.2 above, to Landlord broom-clean, in good order, repair and condition (reasonable wear and tear and casualty damage excepted), with all of Tenant’s Personal Property removed and free of any Hazardous Materials (excluding any Hazardous Materials in existence as of the applicable Commencement Date except to the extent caused or exacerbated by Tenant), and shall otherwise comply with all of the requirements of Section 15.2 above and Section 41.1 below. The voluntary or other surrender of this Lease by Tenant, or a mutual cancellation thereof, shall not work a merger, and shall, at the option of Landlord, operate as an assignment to it of any or all subleases or subtenancies. The delivery of keys to any employee of Landlord or to Landlord’s agent or any employee thereof shall not be sufficient to constitute a termination of this Lease or a surrender of the Premises.
Article 32
PERFORMANCE BY TENANT
All covenants and agreements to be performed by Tenant under any of the terms of this Lease shall be timely performed by Tenant at Tenant’s sole cost and expense and without any abatement of Rent except as expressly provided in this Lease. If Tenant shall fail to pay any sum of money owed to any party other than Landlord, for which it is liable hereunder, or if Tenant shall fail to timely perform any other act on its part to be performed hereunder Landlord may, without waiving or releasing Tenant from obligations of Tenant, but shall not be obligated to, make any such payment or perform any such other act to be made or performed by Tenant pursuant to Section 25.5 above.
Article 33
PARKING
Beginning on the Commencement Date, Tenant and Tenant’s business visitors (“Tenant’s Parking Invitees”) shall be entitled to use the number of parking spaces set forth in Section 1.8 during the Term, which parking spaces shall be located in the surface parking area of the Project (“Parking Area”). There shall be no direct charge attributable to Tenant’s use of the Parking Area, other than any taxes imposed by any governmental authority in connection with the renting of parking spaces by Tenant or the use of the Parking Area by Tenant. Tenant’s continued right to use the Parking Area is conditioned upon Tenant abiding by the Parking Rules and Regulations set forth on Exhibit “G” as amended from time to time for the orderly operation and use of the Parking Area, including any sticker, parking pass or other identification system established by Landlord, Tenant’s cooperation in seeing that Tenant’s employees and visitors also comply with the Parking Rules and Regulations and Tenant not being in default under this Lease (beyond any applicable notice and cure periods). Landlord specifically reserves the right to change the size, configuration, design, layout and all other aspects of the Parking Area at any time and Tenant acknowledges and agrees that Landlord may, from time to time, close-off or restrict access to the Parking Area for purposes of permitting or facilitating any such construction, alteration or improvements; provided, however, in connection with any such access restrictions, the same shall be without incurring any liability to Tenant and without any abatement of Rent under this Lease to the extent Landlord provides any reasonably required temporary, alternate parking. Landlord may delegate its responsibilities hereunder to a parking operator in which case such parking operator shall have all the rights of control attributed hereby to Landlord. Any parking passes issued to Tenant pursuant to this Article 33 shall be provided to Tenant solely for use by Tenant’s own personnel and such passes may not be transferred, assigned, subleased or otherwise alienated by Tenant without Landlord’s prior approval. Tenant may validate visitor parking by such method or methods as Landlord may establish, at the validation rate from time to time generally applicable to visitor parking.
Article 34
LIMITATION ON LIABILITY
1.1Landlord’s Liability. In consideration of the benefits accruing hereunder, Tenant and all of its successors and assigns covenant and agree that, in the event of any actual or alleged failure, breach or default hereunder by Landlord:
1.1.1.The sole and exclusive remedy shall be against Landlord’s interest in the Project;
1.1.2.Only Landlord shall be sued or named as a party in any suit or action;
1.1.3.No writ of attachment, execution, possession, or sale, will ever be levied against the assets of Landlord, except the Buildings;
1.1.4.The obligations under this Lease do not constitute personal obligations of any Landlord Indemnified Party (other than Landlord), and Tenant shall not seek recourse against any Landlord Indemnified Party (other than Landlord) or any of their personal assets (other than Landlord's interest in the Project) for satisfaction of any liability in respect to this Lease (and, without limiting the foregoing, neither the negative capital account of any Landlord Indemnified Party, nor any obligation of any Landlord Indemnified Party to restore a negative capital account or to contribute capital to Landlord, shall at any time be deemed to be the property or an asset of Landlord, and neither Tenant nor any of its successors or assigns shall have any right to collect, enforce or proceed against or with respect to any such negative capital account of an Landlord Indemnified Party’s obligation to restore or contribute); and
1.1.5.These covenants and agreements are enforceable by Landlord and the other Landlord Indemnified Parties.
Article 35
CONFIDENTIALITY
Tenant agrees that the terms and conditions of this Lease and any documents or information delivered hereunder are confidential and constitute proprietary information. Disclosure of the terms and conditions hereof or any documents or information delivered hereunder could adversely affect the ability of Landlord to negotiate with other tenants or potential tenants of the Project. Tenant and its partners, officers, members, managers, directors, employees, agents, advisors, representatives and attorneys, shall not disclose the terms and conditions of this Lease or any documents or information delivered hereunder to any other person without the prior written consent of Landlord except (a) pursuant to an order of a court of competent jurisdiction, (b) to its investors, lenders or prospective lenders, (c) to accountants who audit its financial statements or prepare its tax returns, (d) to its attorneys, insurers, to any Governmental Authority or person to whom disclosure is required by applicable Law, (e)
to Permitted Tranferees, affiliates of Tenant and prospective assignees and/or subtenants and (f) in connection with any action brought to enforce the terms of this Lease on account of the breach or alleged breach hereof. In the event that Tenant concludes that it is obligated by Law to disclose the terms of this Lease (e.g., pursuant to a filing with the Securities and Exchange Commission (“SEC”) or the NASDAQ), Tenant shall provide written notice to Landlord before any public disclosure, and the parties shall use their commercially reasonable efforts to cause a mutually agreeable release or announcement to be issued. The foregoing shall not preclude communications or disclosures by Tenant necessary to implement the provisions of this Lease or to comply with the accounting and disclosure obligations of the SEC or the rules of the New York Stock Exchange. If Tenant determines that it is required to file this Lease, a summary thereof, or a notification thereof, and/or descriptions related thereto, to comply with the requirements of an applicable stock exchange, SEC regulation, or any Governmental Authority, including the SEC, Tenant shall use its best efforts to provide the maximum amount of advance written notice of any such required disclosure to Landlord with a minimum advance notice period of five (5) business days. Tenant will provide Landlord with a copy of this Lease marked to show provisions for which Tenant intends to seek confidential treatment. Tenant shall reasonably consider and incorporate Landlord’s comments thereon to the extent consistent with the legal requirements governing redaction of information from material agreements that must be publicly filed. Notwithstanding anything to the contrary contained in this Lease, neither Landlord nor Tenant (nor either of their affiliates) shall (orally or in writing) publicly disclose, issue any press release or make any other public statement, or otherwise communicate with the media, concerning the existence of this Lease or the subject matter hereof, without the prior written approval of the other.
Article 36
MISCELLANEOUS
1.1Rules and Regulations. Tenant shall faithfully observe and comply with the “Rules and Regulations”, a copy of which is attached hereto, marked Exhibit “F”, and incorporated herein by this reference (“Rules and Regulations”) and all reasonable modifications thereof and reasonable additions thereto made from time to time by Landlord; provided, however any modifications and/or additions to the Rules and Regulations after the Premises B Commencement Date that are not required by applicable law shall be subject to the prior written approval of Tenant, which approval will not be unreasonably withheld, conditioned or delayed. Landlord shall not be responsible to Tenant for the violation or nonperformance by any other tenant or occupant of the Project of any of said Rules and Regulations.
1.2Conflict of Laws. This Lease shall be governed by and construed pursuant to the Laws of the State of California (without reference to its conflicts of laws rules or principles).
1.3Successors and Assigns. Except as otherwise provided in this Lease, all of the covenants, conditions and provisions of this Lease shall be binding upon and inure to the benefit of the parties hereto and their respective heirs, personal representatives, successors and assigns (subject to the restrictions on Tenant’s right to assign, sublet or transfer contained in Article 27).
1.4Professional Fees. If (a) Landlord should bring suit for possession of the Premises, for the recovery of any sum due under this Lease, or because of the breach of any provisions of this Lease, or for any other relief against Tenant hereunder, or (b) any other litigation is commenced between the parties with respect to this Lease, then all reasonable costs and reasonable expenses, including, without limitation, actual professional fees such as appraisers’, accountants’, and attorneys’ fees, incurred by the prevailing party therein shall be paid by the other party, which obligation on the part of the other party shall be deemed to have accrued on the date of the commencement of such action and shall be enforceable whether or not the action is prosecuted to judgment.
1.5Mortgagee Protection. Tenant shall give Notice to any beneficiary of a deed of trust or mortgage covering the Premises whose address shall have been furnished to Tenant of any default on the part of Landlord under this Lease, and shall offer such beneficiary or mortgagee a reasonable opportunity to cure the default, in no event less than sixty (60) days, including time to obtain possession of the Premises by power of sale or a judicial foreclosure if necessary to effect a cure.
1.6Definition of Landlord. The term “Landlord”, as used in this Lease, so far as covenants or obligations on the part of Landlord are concerned, shall be limited to mean and include only the owner or owners, at the time in question, of the fee title of the Premises or the lessees under any ground lease, if any. In the event of any transfer, assignment or other conveyance or transfers of any such title, the original landlord herein named (and in case of any subsequent transfers or conveyances, the then grantor) shall be automatically freed and relieved from and after the date of such transfer, assignment or conveyance of all liability as respects the performance of any covenants or obligations on the part of Landlord contained in this Lease thereafter to be performed. Without further agreement, the transferee of such title shall be deemed to have assumed and agreed to observe and perform any and all obligations of Landlord hereunder, during its ownership of the Premises. Landlord may transfer its interest in the Premises without the consent of Tenant and such transfer or any subsequent transfer shall not be deemed a violation on Landlord’s part of any of the terms and conditions of this Lease.
1.7Identification of Tenant. If more than one entity executes this Lease as Tenant: (a) each of them shall be jointly and severally liable for observing and performing all of the terms, covenants, conditions, provisions and agreements of this Lease to be observed and performed by Tenant, and (b) the term “Tenant” as used in this Lease shall mean and include each of them jointly and severally. The act of or Notice from, or Notice or refund to, or the signature of any one or more of them, with respect to the tenancy of this Lease, including but not limited to any renewal, extension, expiration, termination or modification of this Lease, shall be binding upon each and all of the entities executing this Lease as Tenant with the same force and effect as if each and all of them had so acted, so given or received such Notice or refund, or so signed.
1.8Force Majeure. Each party shall have no liability whatsoever to the other party on account of any of the following (“Force Majeure”): (a) the inability of such party to fulfill, or any delay in fulfilling, any of its obligations under this Lease by reason of strike, other labor trouble, governmental preemption or priorities or other controls in connection with a national or other public emergency, or shortages of fuel, supplies or labor resulting therefrom, inclement weather, casualty, earthquake, war, riot, civil commotion, terrorism, pandemic, epidemic, or any other cause, whether similar or dissimilar to the above, beyond such party’s reasonable control (financial condition excepted); or (b) any failure or defect in the supply, quantity, character, or maintenance of electricity, water, intrabuilding network telephone and data cable service, or other service furnished to the Premises by reason of any requirement, act or omission of the public utility or others furnishing the Buildings with such service, or for any other reason, whether similar or dissimilar to the above, beyond such party’s reasonable control. If this Lease specifies a time period for performance of an obligation of such party, that time period shall be extended by the period of any delay in such party’s performance caused by any of the events of Force Majeure described above. Notwithstanding the foregoing, nothing in this Section 36.8 shall relieve Tenant from the obligation to pay any Rent or extend the time for payment of any Rent.
1.9Terms and Headings. The words “Landlord” and “Tenant” as used herein shall include the plural as well as the singular. Words used in any gender include other genders. The Article and Section headings of this Lease are not a part of this Lease and shall have no effect upon the construction or interpretation of any part hereof.
1.10Examination of Lease. Submission of this instrument for examination or signature by Tenant does not constitute a reservation of or option for lease, and it is not effective as a lease or otherwise until execution by and delivery to both Landlord and Tenant.
1.11Time. Time is of the essence with respect to the performance of every provision of this Lease in which time is a factor.
1.12Prior Agreement; Amendments. This Lease contains all of the agreements of the parties hereto with respect to any matter covered or mentioned in this Lease, and no prior agreement or understanding pertaining to any such matter, written or verbal, shall be effective for any purpose. No provisions of this Lease may be amended or added to except by an agreement in writing signed by the parties hereto or their respective successors-in-interest.
1.13Severability. Any provision of this Lease which shall prove to be invalid, void or illegal shall in no way affect, impair or invalidate any other provision hereof, and such other provisions shall remain in full force and effect.
1.14Recording. Tenant shall not record this Lease or a short form memorandum hereof without the consent of Landlord (in its sole and absolute discretion), which consent may be conditioned upon Tenant’s delivery to Landlord of a fully executed quitclaim releasing Tenant’s interest in the Premises, the Project or any portion thereof.
1.15Modification for Lenders. If, in connection with obtaining construction, interim or permanent financing for the Project the lender shall request reasonable modifications in this Lease as a condition to such financing, Tenant will not unreasonably withhold, delay or condition its consent thereto, provided that such modifications do not materially increase the obligations or costs of Tenant hereunder or materially adversely affect the leasehold interest hereby created or Tenant’s rights hereunder.
1.16Financial Statements. At any time during the Term of this Lease, Tenant shall, upon ten (10) business days’ Notice from Landlord, provide Landlord with its current financial statements and financial statements of the two (2) years prior to the year in which Landlord’s Notice was given (together with, if Tenant's obligations under this Lease are guaranteed, the guarantor's current financial statements and financial statements of the two (2) years prior to the year in which Landlord’s Notice was given); provided Tenant shall not be required to provide such financial statements more than once in each consecutive twelve (12) month period during the Term unless (a) Tenant is in default under this Lease, or (b) requested in connection with a proposed sale, transfer, financing or refinancing of the Project or any portion thereof. If Tenant’s compliance with the immediately preceding sentence would result in Tenant providing the same financial statements that were previously provided to Landlord, then Tenant shall be responsible for providing only those financial statements, if any, that have not previously been delivered. Such statements shall be prepared in accordance with generally accepted accounting principles and, if such is the normal
practice of Tenant, shall be audited by an independent certified public accountant. All financial statements shall be certified as true and correct by Tenant’s chief financial officer and Tenant agrees that Landlord may share such financial statements with prospective lenders and purchasers of the Property provided that such lenders and purchasers execute a non-disclosure agreement in a form reasonably acceptable to Tenant if such financial statements are not publicly available. Notwithstanding anything to the contrary set forth in this Section 36.16, Guarantor shall not be required to comply with the terms of this Section 36.16 if Guarantor’s stock is traded on a public exchange and such entity is in compliance with the disclosure requirements as a public company, and Tenant shall not be required to comply with the terms of this Section 36.16 if Guarantor’s publicly available financial statements include Tenant’s financial statements on a consolidated basis.
1.17Quiet Enjoyment. Landlord covenants and agrees with Tenant that, upon Tenant paying the Rent required under this Lease and performing all of the covenants and provisions on Tenant’s part to be observed and performed under this Lease, Tenant shall during the Term, peaceably and quietly have, hold and enjoy the Premises in accordance with this Lease without interference by any persons lawfully claiming by or through Landlord. The foregoing covenant is in lieu of any other covenant express or implied.
1.18Tenant as Corporation, Partnership or Limited Liability Company. If Tenant is a corporation, partnership or limited liability company, Tenant represents and warrants that it is an entity duly qualified to do business in California and that the individuals executing this Lease on Tenant’s behalf are duly authorized to execute and deliver this Lease on its behalf, in the case of a corporation, in accordance with its by-laws and with a duly adopted resolution of the board of directors of Tenant, a copy of which shall be delivered to Landlord upon execution hereof by Tenant, in the case of a partnership, in accordance with the partnership agreement and the most current amendments thereto, if any, copies of which shall be delivered to Landlord upon execution hereof by Tenant, and, in the case of a limited liability company, in accordance with its governing documents and any documents required thereby, copies of which shall be delivered to Landlord upon execution hereof by Tenant, and that this Lease is binding upon Tenant in accordance with its terms.
1.19CASp Disclosure. For purposes of Section 1938 of the California Civil Code, Landlord hereby discloses to Tenant that the Building Common Areas, Project Common Areas and Premises, as of the date of this Lease, have not been inspected by a Certified Access Specialist (CASp), as that term is defined in California Civil Code Section 55.52. In accordance with subsection (e) of Section 1938 of the California Civil Code, Tenant is further notified as follows:
A Certified Access Specialist (CASp) can inspect the subject premises and determine whether the subject premises comply with all of the applicable construction-related accessibility standards under state law. Although state law does not require a CASp inspection of the subject premises, the commercial property owner or lessor may not prohibit the lessee or tenant from obtaining a CASp inspection of the subject premises for the occupancy or potential occupancy of the lessee or tenant, if requested by the lessee or tenant. The parties shall mutually agree on the arrangements for the time and manner of the CASp inspection, the payment of the fee for the CASp inspection, and the cost of making any repairs necessary to correct violations of construction-related accessibility standards within the premises.
Article 37
SIGNAGE
Landlord retains absolute control over the exterior appearance of the Buildings and the Project and the exterior appearance of the Premises as viewed from the Building Common Areas and Project Common Areas. Tenant will not, without Landlord’s prior written consent, install, or permit to be installed, any drapes, furnishings, signs, lettering, designs, advertising or any items that will in any way alter the exterior appearance of the Buildings, the Project or the exterior appearance of the Premises as viewed from the Building Common Areas and Project Common Areas. Any sign, advertising, design, or lettering installed by Tenant shall be considered an Alteration and shall be subject to the provisions of Article 15; provided that Landlord shall have the right to withhold its consent to the same in its sole and absolute discretion. Landlord, at its sole cost and expense, shall install suite identification signage bearing Tenant’s name and logo adjacent to the entrance to the Premises. In addition, Tenant, at its sole cost and expense, shall have the exclusive right, without obligation, to have its name (including its logo) displayed on (a) one (1) building-top sign on one of the Buildings, and (b) on the Project’s monument sign, in each case in a location designated by Landlord (collectively, “Tenant’s Signage”), subject to the terms and conditions set forth in this Article 37. Tenant hereby acknowledges that, as of the Effective Date, Landlord has not received approval from the City of San Diego (or any other authority with jurisdiction over the Project) for any exterior signage for the Project and, accordingly, Tenant’s right to any such signage (including, without limitation, Tenant’s Signage set forth above) is contingent on such approval. In addition, the specifications of Tenant’s Signage (including, without limitation, the dimensions and configuration thereof) shall be subject to the prior written approval of Landlord,
which approval shall not be unreasonably withheld, provided that such specifications are consistent with Landlord’s sign program for the Project and all applicable Laws. The construction and installation of Tenant’s Signage shall be performed by Tenant (upon Landlord’s approval thereof), at Tenant’s sole cost and expense. Prior to installation, Tenant shall deliver to Landlord a drawing depicting the design, size, location, specifications, graphics, materials and colors of Tenant’s Signage, all of which shall be consistent with Landlord’s sign program and the Rules and Regulations. Tenant’s Signage shall be subject to any applicable review and approval by the City of San Diego and any other authorities with jurisdiction over the Project, and Tenant shall obtain all applicable permits and authorizations by Governmental Authorities prior to installation of Tenant’s Signage. After installation, Tenant shall maintain Tenant’s Signage in good condition and repair at all times through the Term. Tenant shall remove Tenant’s Signage upon the expiration or earlier termination of this Lease and shall repair any damage caused thereby. The maintenance and removal of Tenant’s Signage shall be performed at Tenant’s sole cost and expense. All signage rights granted to Tenant under this Lease are personal to the original Tenant named herein, and, except in connection with a Permitted Transfer to a Permitted Transferee, may not be assigned or transferred without Landlord’s prior written consent, which consent Landlord may withhold in its sole and absolute discretion.
Article 38
EXECUTIVE ORDER 13224
Tenant hereby represents and warrants to Landlord that Tenant is not: (a) in violation of any Anti-Terrorism Law (defined below); (b) conducting any business or engaging in any transaction or dealing with any Prohibited Person (defined below), including, without limitation, the making or receiving of any contribution of funds, goods or services to or for the benefit of any Prohibited Person; (c) dealing in, or otherwise engaging in any transaction relating to, any property or interest in property blocked pursuant to Executive Order No. 13224; (d) engaging in or conspiring to engage in any transaction that evades or avoids, or has the purpose of evading or avoiding, or attempts to violate any of the prohibitions set forth in any Anti-Terrorism Law; or (e) a Prohibited Person, nor are any of its partners, members, managers, officers or directors a Prohibited Person. As used herein, “Anti-Terrorism Law” is defined as any Law relating to terrorism, anti-terrorism, money laundering or anti-money laundering activities, including, without limitation, Executive Order No. 13224 and Title 3 of the USA Patriot Act. As used herein “Executive Order No. 13224” is defined as Executive Order No. 13224 on Terrorist Financing effective September 24, 2001, and relating to “Blocking Property and Prohibiting Transactions With Persons Who Commit, or Support Terrorism.” “Prohibited Person” is defined as: (i) a person or entity that is listed in the Annex to Executive Order No. 13224; (ii) a person or entity with whom Tenant or Landlord is prohibited from dealing or otherwise engaging in any transaction by any Anti-Terrorism Law; or (iii) a person or entity that is named as a “specially designated national and blocked person” on the most current list published by the U.S. Treasury Department Office of Foreign Assets Control at its official website, http://www.treas.gov/ofac/t11sdn.pdf or at any replacement website or other official publication of such list. “USA Patriot Act” is defined as the “Uniting and Strengthening America by Providing Appropriate Tools Required to Intercept and Obstruct Terrorism Act of 2001” (Public Law 107-56).
Article 39
WAIVER OF JURY TRIAL
TO THE EXTENT PERMITTED BY LAW, LANDLORD AND TENANT WAIVE THE RIGHT TO A TRIAL BY JURY.
Article 40
TENANT REPRESENTATIONS
Tenant represents and warrants to Landlord as of the date hereof and continuing thereafter as follows:
(a) The execution and delivery of this Lease by Tenant will not result in a breach of the terms or provisions of, or constitute a default (or a condition that, upon notice or lapse of time, or both, would constitute a default) under its organizational documents or any indenture, agreement, or obligation by which Tenant is bound, and will not constitute a violation of any Law applicable to Tenant.
(b) The person executing this Lease on Tenant’s behalf is duly authorized to so act; that Tenant is duly organized, is qualified to do business in the jurisdiction in which the Project is located, is in good standing under the Laws of the state of its organization and the Laws of the jurisdiction in which the Project is located, and has the power and authority to enter into this Lease; and that all action required to authorize Tenant and such person to enter into this Lease has been duly taken.
(c) Any financial statements provided by Tenant are true, correct and complete in all material respects and do not omit to state a fact that would be material to Tenant’s financial condition. There has been no material adverse change in Tenant’s financial condition since Tenant provided such financial statements.
(d) Tenant is in compliance with all applicable anti-money laundering Laws, including, without limitation, the USA Patriot Act, and the Laws administered by the United States Treasury Department’s Office of Foreign Assets Control, including, without limitation, Executive Order No. 13224. Tenant is not owned or controlled directly or indirectly by any person or entity, on the SDN List published by the United States Treasury Department’s Office of Foreign Assets Control and Tenant is not a person otherwise identified by any Governmental Authority as a person with whom a U.S. Person is prohibited from transacting business. As of the date hereof, a list of such designations and the text of Executive Order No. 13224 are published under the internet website address www.ustreas.gov/offices/enforcement/ofac.
Article 41
ADDITIONAL PROVISIONS
1.1Early Access. Following twenty-four (24) hours’ notice to Landlord and upon receipt of written approval by Landlord (which shall not be unreasonably withheld or delayed), Landlord shall permit Tenant and its agents to enter the applicable Premises up to sixty (60) days prior to the applicable Commencement Date (“Early Access Period”) for the sole purpose of installing, at Tenant’s sole cost and expense, its furniture, fixtures, equipment and cabling in the applicable Premises; provided, however, that in no event shall such early access, regardless of when provided, extend or otherwise affect the Commencement Date. Tenant, at its sole cost and expense, may enter the Premises prior to the Early Access Period to conduct indoor air samples, subject to Landlord’s prior written consent, which shall not be unreasonably withheld or delayed, and provided that Tenant uses Weis Environmental (or if Weis Environmental is not available, another environmental consultant selected by Tenant, subject to Landlord’s reasonable approval) to gather and analyze any such air samples; any such entry by Tenant shall be subject to all other terms and conditions of this Section 41.1. Any such entry shall be in a manner and upon terms and conditions and at times satisfactory to Landlord’s representative. The foregoing license to enter the Premises prior to the Commencement Date is, however, conditioned upon Tenant’s contractors and their subcontractors and employees working in harmony and not interfering with the work being performed by Landlord. If at any time such entry shall cause disharmony or interfere with the work being performed by Landlord, this license may be withdrawn by Landlord upon twenty-four (24) hours written notice to Tenant. Tenant shall be liable for any damages caused by Tenant’s activities at the Premises. Such license is further conditioned upon the compliance by Tenant’s contractors with all requirements imposed by Landlord on third party contractors, including, without limitation, the maintenance by Tenant and its contractors and subcontractors of workers’ compensation and public liability and property damage insurance in amounts and with companies and on forms satisfactory to Landlord, with certificates of such insurance being furnished to Landlord prior to proceeding with any such entry. The entry shall be deemed to be under all of the provisions of this Lease except as expressly set forth in this Section 41.1. During the Early Access Period, Tenant shall have no obligation to pay Basic Rent, Operating Expenses or Real Property Taxes. Landlord shall not be liable in any way for any injury, loss or damage which may occur to any such work being performed by Tenant, the same being solely at Tenant’s risk. All costs and expenses in connection with or arising out of the performance of any work by Tenant during such early entry shall be borne by Tenant, and all payments therefor shall be made by Tenant promptly as they become due. Tenant shall, at its sole cost and expense, comply with all applicable laws, ordinances, regulations and policies governing its work. Tenant shall defend, indemnify and hold Landlord and its members, agents, employees, partners, and their respective employees, partners, officers, directors, agents, representatives, successors and assigns, harmless from and against any and all suits, claims, actions, losses, costs, liabilities or expenses (including reasonable attorneys’ fees and claims for workers’ compensation) to the extent arising out of or in connection with any and all work by Tenant or on Tenant’s behalf, during such early entry (including, but not limited to, claims for breach of warranty, personal injury or property damage). Landlord shall have the right, in Landlord’s sole and absolute discretion, to settle, compromise, or otherwise dispose of any and all suits, claims, and actions against any of the indemnified parties arising out of or in connection with the work performed by Tenant during any early entry. Tenant shall coordinate such entry with Landlord’s building manager and, except as expressly set forth in this Section 41.1, such entry shall be made in compliance with all terms and conditions of this Lease and the Rules and Regulations attached hereto.
1.2Environmental Assessments. Tenant hereby acknowledges receipt of the Phase I Environmental Site Assessment dated August 26, 2020, prepared by Partner Engineering and Science, Inc. regarding the Project (“Original Phase I Assessment”); a copy of the summary of the Original Phase I Assessment is attached hereto as Exhibit “H”. The Original Phase I Assessment identifies a controlled recognized environmental condition that affects Premises A and Premises B (the “CREC”). Tenant may elect for Landlord to remediate the CREC by delivering Landlord written notice thereof any time before Landlord commences the Tenant Improvements (a “Remediation Notice”). If Tenant elects for Landlord to remediate the CREC pursuant to the immediately preceding sentence, Landlord may, at Landlord’s sole cost, either (a) install a vapor intrusion mitigation system and a sub-slab depressurization system as identified in the letter dated April 22, 2021 from Weis Environmental or (b) remove the
solvents and volatile compounds identified in the CREC from the soils of the Premises, (as applicable, the “Remediation Work”). The Original Phase I Assessment shall serve as the “baseline” for determining the environmental condition of the Project prior to Tenant’s occupancy thereof. In addition to the surrender obligations set forth elsewhere in this Lease (including, without limitation, Section 15.2 and Article 31), upon the expiration or earlier termination of this Lease, Tenant, at its sole cost and expense, shall (a) cause a Phase I environmental assessment (or similar non-invasive assessment) of the Project (“Phase I Surrender Assessment”) to be performed and deliver the results thereof to Landlord no later than thirty (30) days following such expiration or earlier termination (but in no event shall the Phase I Surrender Assessment be dated more than ten (10) days prior to such expiration or earlier termination); and (b) if and to the extent recommended by the Phase I Surrender Assessment and consented to by Landlord in writing, cause a Phase II environmental assessment (or similar additional assessment) of the Project (“Phase II Surrender Assessment”) to be performed and deliver the results thereof to Landlord no later than thirty (30) days following the date of the Phase I Surrender Assessment. In addition, Landlord shall have the right, in its sole and absolute discretion, to hire, or to cause Tenant to hire, an environmental consultant to conduct a physical inspection of the Project (“Environmental Inspection”) upon the expiration or earlier termination of this Lease, which inspection shall be at Tenant’s sole cost and expense. The Phase I Surrender Assessment and any Phase II Surrender Assessment and/or Environmental Inspection, as the same compare to the Original Phase I Assessment, shall be used to, among other things, determine the extent of Tenant’s compliance (or noncompliance) with Section 8.3 above.
1.3Generator. Tenant shall have the right to install one (1) standalone emergency generator serving the Premises (“Generator”) in a location reasonably approved by Landlord. All costs and expenses to install and maintain the Generator shall be the sole responsibility of Tenant, except that Tenant may elect, prior to the commencement of the Tenant Improvements, to apply a portion of the Tenant Improvement Allowance toward the cost of the Generator. The installation of the Generator shall be deemed an Alteration subject to the terms and conditions of Article 15 above.
1.4Security System. Tenant shall have the right to install an integrated security system within the Premises (“Security System”). All costs and expenses to install and maintain the Security System shall be the sole responsibility of Tenant. The installation of the Security System shall be deemed an Alteration subject to the terms and conditions of Article 15 above.
1.5Roof Rights and Telecommunications Equipment. Tenant shall have the non-exclusive right to install on the roof of the Buildings, at its sole cost and expense but at no charge due to Landlord, one (1) satellite dish or one (1) antenna, together with related cables and appurtenances for Tenant’s data transmission network, provided that (i) the dish or antenna, all appurtenant equipment, and all connecting wires and cables on the roof and within the Building risers shall be depicted on plans and specifications showing the size and configuration thereof and such plans and specifications shall be delivered to Landlord for its prior approval, which shall not be unreasonably withheld or delayed; (ii) any such installation shall be completed in full compliance with any and all rules established for the location of communications installations established by Landlord, all applicable laws, municipal or governmental rules, regulations, permits and approvals, any covenants, conditions or restrictions governing the Premises and/or the Project in effect at the time Tenant desires to install such equipment, and any applicable owners’ association rules, regulations, permits and approvals; (iii) any such installation and the use thereof shall not interfere with the operation of any third party communications installations existing on the roof of the Buildings at the time Tenant installs its equipment; and (iv) any such installation shall not be visible from view from the ground level or any other lot within the Project to Landlord’s reasonable satisfaction. Subject to the foregoing, Tenant shall have the right to vertical access to the roof at no additional cost for the purpose of installing and maintaining its rooftop installation. Tenant shall pay all costs and expenses incurred in connection with the installation, maintenance and, upon the expiration of the Term or any earlier termination of this Lease, the removal of all of Tenant’s equipment, wires and cables from the roof and all other portions of the Buildings. Furthermore, Tenant shall obtain Landlord’s consent, which shall not be unreasonably withheld or delayed, prior to each occasion on which Tenant or its agents access the roof. Tenant shall contract solely with Landlord’s roof contractor for any and all rooftop installation and removal.
[signatures on following page]
IN WITNESS WHEREOF, the parties have executed this Lease as of the Effective Date.
| | | | | |
| LANDLORD: | 10240 FLANDERES INVESTORS LLC, |
| a Delaware limited liability company |
| |
| By: SRE SD7, LLC, a Delaware limited liability company, its sole member
By: SB Flanders Investors LLC, a California limited liability company its manager
By: /s/ Steve Bollert Name: Steve Bollert Its: Authorized Signatory |
| |
| TENANT: | TRILINK BIOTECHNOLOGIES, LLC, |
| a Delaware limited liability company |
| |
| By: /s/ Kevin Herde Name: Kevin Herde Its: CFO
By: /s/ Brian Neel Name: Brian Neel Its: COO |
INDEX OF DEFINED TERMS
Abandonment, 31
Abatement Event, 24
Actual Delivery Date for Premises A, 6
Actual Delivery Date for Premises B, 7
ADA, 13
Additional Rent, 7
Alterations, 20
Annual Reconciliation, 12
Anti-Terrorism Law, 43
Assignment, 34
Basic Rent, 2
Broker(s), 3
Building Common Areas, 3
Building Standard Improvements. See Exhibit B
Building Systems, 21
Buildings, 1
Claims, 25
Common Areas, 3
Comparable Transactions, 5
Consent Instrument, 36
Construction Documents. See Exhibit B
Controllable Operating Expenses, 11
CREC, 45
Debts. See Exhibit D
Default Rate, 8
Discount Rate, 33
Dispute Notice, 12
Early Access Period, 44
Effective Date, 1
Eligibility Period, 24
Environmental Inspection, 46
Estimated Commencement Date, 2
Estimated Statement, 11
Excess Costs. See Exhibit B
Executive Order No. 13224, 44
Expiration Date, 4
Fair Market Rental Value, 5
First Outside Delivery Date, 6
Force Majeure, 41
Generator, 46
Governmental Authority, 8
Guaranteed Obligations. See Exhibit D
Guarantor, 3
Hazardous Material, 16
Hazardous Material Laws, 16
Holder, 37
HVAC, 23
Initial Term, 1, 4
Insolvent. See Exhibit D
Institutional Owner Practices, 9
Insurance Expenses, 11
Interest Rate, 9
Landlord, 1
Landlord Default, 34
Landlord Indemnified Parties, 27
Landlord Work. See Exhibit B
Landlord’s Accountant, 12
Landlord’s Address, 1
Landlord’s Fair Market Rental Value Notice, 5
Landlord’s Repair Notice, 30
Laws, 13
Lease, 1
Leasehold Improvements, 21
LW Contractor. See Exhibit B
Major Alteration, 21
Notice, 18
Notice of Default, 33
Operating Expenses, 8
Option, 4
Option Term, 4
Original Phase I Assessment, 45
Original Tenant, 5
Outside Agreement Date, 5
Parking Area, 39
Permitted Alterations, 21
Permitted Transfer, 37
Permitted Transferee, 37
Permitted Use, 3
Phase I Surrender Assessment, 45
Phase II Surrender Assessment, 45
Premises, 1
Premises A, 1
Premises A Commencement Date, 2
Premises B, 1
Premises B Commencement Date, 2
Premises B Outside Delivery Date, 7
Premises B Second Outside Delivery Date, 7
Premises B Work Commencement Date, 7
Prime Rate, 8
Prohibited Person, 44
Project, 3
Project Common Areas, 4
Project Site, 3
Project Site Plan, 3
Property Manager, 21
Proposed Transferee, 35
Real Property Taxes, 10
Reimbursements, 10
Remediation Notice, 45
Remediation Work, 45
Rent, 7
Rentable Square Feet, 1
Review Notice, 12
Rules and Regulations, 40
SEC, 40
Second Outside Delivery Date, 7
Security Deposit, 3
Security Instruments, 37
Security System, 46
SIR, 28
Space Plan. See Exhibit B
Sublease, 34
Substantial Completion. See Exhibit B
Substantially Complete. See Exhibit B
Supplemental Equipment, 22
Taking, 31
Tenant, 1
Tenant Default, 31
Tenant Delay. See Exhibit B
Tenant Estoppel Certificate, 38
Tenant Improvement Allowance, 2
Tenant Improvements. See Exhibit B
Tenant Party, 15
Tenant’s Address, 1
Tenant’s Parking Invitees, 39
Tenant’s Percentage, 3, 10
Tenant’s Personal Property, 21
Term, 4
TI Contractor. See Exhibit B
Total Destruction, 30
Transfer, 34
Transfer Date, 35
Transfer Instrument, 36
Transfer Notice, 35
Transferee, 36
Upper Slab, 26
USA Patriot Act, 44
Utility Expenses, 11
Waste Products. See Exhibit F
Waste Regulations. See Exhibit F
Work Letter Agreement, 3
Work Schedule. See Exhibit B
DocumentExhibit 10.16
FIRST AMENDMENT TO LEASE AGREEMENT
This FIRST AMENDMENT TO LEASE AGREEMENT (“Amendment”) is entered into as of October 14, 2021 (“Effective Date”), by and between 10240 FLANDERS fNVESTORS LLC, a Delaware limited liability company (“Landlord”), and TRILINK BIOTECHNOLOGIES, LLC, a Delaware limited liability company (“Tenant”), with reference to the facts set forth in the Recitals below.
RECITALS
A.Landlord and Tenant are parties to that certain Lease Agreement dated August 6, 2021 (“Lease”), for certain Premises consisting of approximately 32,236 Rentable Square Feet within the Building known as 10240 Flanders Court, San Diego, California (“Premises A”), and approximately 32,770 Rentable Square Feet within the Building known as 10247 Flanders Court, San Diego, California (“Premises B”), as more specifically described in the Lease.
B.Landlord and Tenant have agreed to reduce the Abatement Period for each Premises, as more fully set forth below.
AMENDMENT
NOW, THEREFORE, in consideration of the Recitals above, the mutual covenants and conditions below, and other good and valuable consideration, the receipt and sufficiency of which are hereby acknowledged, the parties agree as follows:
1.Abatement Period. Notwithstanding any provision of the Lease to the contrary, provided that Tenant is not then in default under the Lease beyond any applicable notice and cure period, the abatement of monthly installments of Basic Rent for the Premises shall be reduced from six (6) months to five and one-half (5½) months (month two (2) through the first one half of month seven (7) of the Initial Term for each Premises) (each an “Abatement Period”).
2.Brokers. Tenant represents and warrants to Landlord that no patty is entitled to a commission or fee by or through Tenant in connection with this Amendment. Tenant shall hold Landlord free and harmless from any and all claims, demands, losses, liabilities, lawsuits, judgments, costs and expenses (including, without limitation, reasonable attorneys' fees) with respect to any leasing commission or equivalent compensation alleged to be owing on account of any dealings with any party claiming by or through Tenant.
3.Defined Terms. Unless otherwise specifically defined in this Amendment, terms with initial capital letters in this Amendment shall have the same meaning as such terms have in the Lease.
4.No Construction Against Party Drafting Amendment. Landlord and Tenant acknowledge and agree that each of them, and their respective professional advisors, have reviewed this Amendment and that the provisions of this Amendment shall not be construed against either party. The rule of construction that ambiguities are to be construed against the party drafting the agreement shall not apply to the interpretation of this Amendment and is waived.
5.Counterpart Execution. This Amendment may be executed in multiple counterparts, each of which when so executed and delivered shall be deemed to be an original and all of which together shall constitute one instrument.
6.Continued Effect. Except as specifically modified by this Amendment, all of the terms, conditions and provisions of the Lease shall remain in full force and effect.
[signatures on following page]
IN WITNESS WHEREOF, the parties have executed this First Amendment to Lease Agreement as of the Effective Date.
“LANDLORD” 10240 FLANDERS INVESTORS LLC,
a Delaware limited liability company
By: SRE SD7, LLC,
a Delaware limited liability company, its sole member
By: SB Flanders Investors LLC,
a California limited liability company, its manager
By: /s/ Steve Bollert
Name: Steve Bollert
Title: Authorized Signatory
“TENANT” TRILINK BIOTECHNOLOGJES, LLC,
a Delaware limited liability company
By: /s/ Brian Neel
Name: Brian Neel
Its: COO
ACKNOWLEDGMENT OF GUARANTOR
The undersigned, as Guarantor under that certain Lease Guaranty dated August 6, 2021, does hereby acknowledge and agree that the provisions of said Lease Guaranty shall remain in full force and effect as to the obligations of Tenant under the Lease, as amended herein. The undersigned Guarantor acknowledges that Landlord would not have executed this Amendment without this acknowledgment and agreement on the part of the undersigned Guarantor.
GUARANTOR:
MARAVAI INTERMEDIATE HOLDINGS, LLC,
a Delaware limited liability company
By: /s/ Kevin Herde
Name: Kevin Herde
Title: CFO
DocumentExhibit 10.17
SECOND AMENDMENT TO LEASE AGREEMENT
This SECOND AMENDMENT TO LEASE AGREEMENT (“Amendment”) is entered into as of October 1, 2022 (“Effective Date”), by and between 10240 FLANDERS INVESTORS LLC, a Delaware limited liability company (“Landlord”), and TRILINK BIOTECHNOLOGIES, LLC, a Delaware limited liability company (“Tenant”), with reference to the facts set forth in the Recitals below.
RECITALS
A.Landlord and Tenant are parties to that certain Lease Agreement dated August 6, 2021, as amended by that certain First Amendment to Lease Agreement dated October 14, 2021 (“Lease”), for certain Premises consisting of approximately 32,236 Rentable Square Feet within the Building known as 10240 Flanders Court, San Diego, California (“Premises A”), and approximately 32,770 Rentable Square Feet within the Building known as 10247 Flanders Court, San Diego, California b(“Premises B”), as more specifically described in the Lease.
B.Tenant has received a grant of certain funds for the construction of a portion the Tenant Improvements from the United States Department of Defense (“DoD”) pursuant to a certain Award/Contract No. W58P052220010 dated as of May 16, 2022 (“DoD Grant”). In connection with The DOD Grant, the DoD has or will impose certain conditions and requirements relative to the construction of the Tenant Improvements and other work on and within the Building including, without limitation, certain required contract provisions and clauses, bond and security requirements, procurement standards, prohibitions on certain telecommunications equipment, security disclosures, audit requirements and the use of labor, as more fully provided in 2 CFR 1122, et seq., 32 CFR 21, et seq. and 48 CFR 31 and 231 and in the regulations cited therein (collectively, “DoD Requirements”).
C.Landlord and Tenant have agreed that (i) any and all increases to the cost of the Tenant Improvements and/or the Landlord Work resulting from or related to compliance with the DoD Requirements shall be paid by Tenant to Landlord as Excess Costs in accordance with the Work Letter Agreement; and (ii) Tenant shall indemnify Landlord from and against any and all damages, losses and/or claims for liability to the extent resulting from or related to the DoD Requirements, as more fully set forth below.
AMENDMENT
NOW, THEREFORE, in consideration of the Recitals above, the mutual covenants and conditions below, and other good and valuable consideration, the receipt and sufficiency of which are hereby acknowledged, the parties agree as follows:
1.Excess Costs. Notwithstanding any provision of the Lease to the contrary, any and all increases to the cost of the Tenant Improvements and/or the Landlord Work (including, without limitation, reasonable attorneys’ fees and costs) resulting from the DoD Requirements, directly or indirectly, shall be deemed “Excess Costs” as provided in the Work Letter Agreement.
2.Tenant Delay. Notwithstanding any provision of the Lease to the contrary, any delay to the scheduled date of Substantial Completion of the Tenant Improvements (i.e., September 1, 2022), to the extent arising from the implementation of the DoD Requirements, directly or indirectly, shall be deemed a “Tenant Delay” as provided in the Work Letter Agreement.
3.Indemnity. Tenant’s indemnification obligations provided in Article 20 of the Lease shall apply to Claims arising from the enforcement of the DoD Requirements; provided, however, that Tenant shall have no such indemnification obligations with respect to any Claim that results from the negligence or willful misconduct of a Landlord Indemnified Party.
4.Defined Terms. Unless otherwise specifically defined in this Amendment, terms with initial capital letters in this Amendment shall have the same meaning as such terms have in the Lease.
5.No Construction Against Party Drafting Amendment. Landlord and Tenant acknowledge and agree that each of them, and their respective professional advisors, have reviewed this Amendment and that the provisions of this Amendment shall not be construed against either party. The rule of construction that ambiguities are to be construed against the party drafting the agreement shall not apply to the interpretation of this Amendment and is waived.
6.Counterpart Execution. This Amendment may be executed in multiple counterparts, each of which when so executed and delivered shall be deemed to be an original and all of which together shall constitute one instrument.
7.Continued Effect. Except as specifically modified by this Amendment, all of the terms, conditions and provisions of the Lease shall remain in full force and effect.
[signatures on following page]
IN WITNESS WHEREOF, the parties have executed this Second Amendment to Lease Agreement as of the Effective Date.
“LANDLORD” 10240 FLANDERS INVESTORS LLC,
a Delaware limited liability company
By: SRE SD7, LLC,
a Delaware limited liability company, its sole member
By: SB Flanders Investors LLC,
a California limited liability company, its manager
By: /s/ Steve Bollert
Name: Steve Bollert
Title: Authorized Signatory
“TENTANT” TRILINK BIOTECHNOLOGIES, LLC,
a Delaware limited liability company
By: /s/ Kurt Oreshack
Name: Kurt Oreshack
Title: EVP, General Counsel
By: /s/ Kevin Herde
Name: Kevin Herde
Title: CFO
ACKNOWLEDGMENT OF GUARANTOR
The undersigned, as Guarantor under that certain Lease Guaranty dated August 6, 2021, does hereby acknowledge and agree that the provisions of said Lease Guaranty shall remain in full force and effect as to the obligations of Tenant under the Lease, as amended herein. The undersigned Guarantor acknowledges that Landlord would not have executed this Amendment without this acknowledgment and agreement on the part of the undersigned Guarantor.
GUARANTOR:
MARAVAI INTERMEDIATE HOLDINGS, LLC,
a Delaware limited liability company
By: /s/ Kevin Herde
Name: Kevin Herde
Title: CFO
Document
LEASE AGREEMENT
BETWEEN
CIGG, LLC
Landlord
AND
CYGNUS TECHNOLOGIES, LLC
Tenant
DATED: June 11, 2021
LEASE
THIS LEASE (this “Lease”) is entered into this June 11, 2021 (the date of execution of this Lease by authorized parties of Landlord and Tenant shall be the “Effective Date”), by and between CIGG, LLC, a North Carolina limited liability company, having its principal place of business at 3514 South College Road, Wilmington, North Carolina, as landlord (“Landlord”), and CYGNUS TECHNOLOGIES, LLC, a Delaware limited liability company, authorized to conduct business in North Carolina, 4332 Southport Supply Rd, SE, Southport; NC 28461, as tenant (“Tenant”).
1.DEMISED PREMISES. Landlord covenants and warrants that prior to the Completion Date (as defined below), it will own lawful fee simple title to those certain premises located within the City of Leland, County of Brunswick, State of North Carolina, and more particularly described by the legal description attached hereto and made a part hereof as Exhibit “A” (the “Land”), and as shown on the ALTA survey attached hereto and made a part hereof as Exhibit “B”.
Landlord hereby leases to Tenant the Land, and entire Building and other improvements to be constructed on the Land pursuant to the terms herein, together with all the rights, easements and interests appurtenant thereto (collectively, the “Demised Premises”), on the terms and conditions contained herein. This Lease is intended as, and shall constitute, an agreement of lease, and shall not be construed as conveying to Tenant any right, title or interest in or to the Demised Premises other than a leasehold estate.
The rentable area (“Rentable Area”) of the Demised Premises shall be determined by Landlord’s architect measured from the outside walls. Landlord’s architect shall deliver to Landlord and Tenant a certificate stating such Rentable Area calculated in accordance with the immediately preceding sentence as soon as practicable following the Completion Date. Within thirty (30) days after the Completion Date, Tenant shall have the right, but not the obligation, to have its architect or contractor measure the Rentable Area of the Demised Premises in accordance with the abovementioned standard of measurement. If Tenant’s architect or contractor determines that the Rentable Area of the Demised Premises is less than the Rentable Area of same as determined by Landlord’s architect, then the parties shall seek to resolve the difference, provided that either Tenant or Landlord shall have the right to submit any such dispute to arbitration in accordance with this Lease. Upon the final determination of the Rentable Area, the Rent, the Tenant Improvement Allowance and any other item of this Lease calculated based on the Rentable Area shall be appropriately adjusted.
2.CONSTRUCTION OF IMPROVEMENTS BY LANDLORD. Landlord covenants that it shall Complete on or prior to the Fixed Completion Date (as defined below), at its sole cost and
expense, in a diligent and good working manner and in compliance with all Laws, Permitted Encumbrances and the Tenant Final Approved Plans, the construction of (i) an approximately 45,156-square foot, two-story office building to Warm Vanilla Shell condition (as defined in Section 6 below), including, without limitation, construction and installation of core and shell, base building electrical, mechanical, plumbing, heating, ventilation, air. conditioning and life safety systems (the “Building”), (ii) all site improvements, including, without limitation, any and all roads, sidewalks, curb cuts, paved parking areas equipped with two electric vehicle charging stations and sufficient number of parking spaces to comply with Laws, ingress and egress drive lanes, utilities, landscaping and make all other improvements required by Laws and the Tenant Final Approved Plans (collectively, the “Site Improvements”) and (iii) the Required Tenant Improvements (as defined in Section 6 below), (such work under (i), (ii) and (iii) is collectively referred to herein as “Landlord’s Work”). Landlord shall post all bonds, secure and pay for all permits, and pay all inspection fees for Completion (as defined below) of Landlord’s Work. Tenant shall at all reasonable times have access to and the right to inspect construction of Landlord’s Work and the elements thereof, wherever and by whomever same take place.
The preliminary site plan attached hereto as Exhibit “C” (the “Preliminary Site Plan”) identifies the general location of the improvements comprising Landlord’s Work and is subject to modification as provided in Section 3. The Tenant Final Approved Plans shall contain the final approved site plan.
The term “Laws” shall mean all laws, statutes, ordinances, rules and regulations, orders and other governmental requirements applicable to the Demised Premises of any municipal, county, state, federal, or other government agency or authority having jurisdiction over the parties to this Lease or the Demised Premises, or both.
3.CONSTRUCTION PLANS.
(A)Landlord’s Proposed Plans. Within sixty (60) days after the Effective Date, Landlord shall furnish Tenant a complete set of design drawings and specifications for Landlord’s Work, which will include a site plan, topographical data, a landscape plan, and set of construction drawings and budget for the Required Tenant Improvements (“Landlord’s Proposed Plans”) of Landlord’s Work, without material deviation from the Preliminary Site Plan. Tenant shall (i) approve, or (ii) approve subject to specified modifications, or (iii) disapprove Landlord’s Proposed Plans, in writing within fifteen (15) days of Tenant’s receipt of same. Landlord shall make those changes which are requested by Tenant and shall within ten (10) days of its receipt of such request submit the revised portion of the Landlord’s Proposed Plans. Tenant shall notify Landlord in writing whether it approves of the resubmitted
Landlord’s Proposed Plans within five (5) business day after its receipt thereof. This process shall be repeated until the Landlord’s Proposed Plans have been finally approved by Landlord and Tenant.
(B)Tenant’s Approval of Landlord’s Proposed Plans. Landlord’s Proposed Plans as approved by Landlord and Tenant shall be referred to as the “Tenant Initial Approved Plans”. Upon Tenant’s approval of Landlord’s Proposed Plans, Tenant shall complete and execute Schedule I, attached hereto, which shall identify the Tenant Initial Approved Plans with specificity.
(C)Governmental Approved Plans. Landlord shall promptly present the Tenant Initial Approved Plans for approval to all governmental agencies having jurisdiction over Landlord’s Work and whose approval is required for construction of same (the “Applicable Governmental Agencies”). The Tenant Initial Approved Plans as approved by the Applicable Governmental Agencies shall be referred to as the “Governmental Approved Plans”. Landlord shall promptly deliver to Tenant the Governmental Approved Plans together with (a) a summary of changes, or statement that no changes were made, from the Tenant Initial Approved Plans, and (b) evidence of governmental approval. If the Tenant Initial Approved Plans were approved by Applicable Governmental Agencies without change or revision, Tenant shall accept the plans as the Tenant Final Approved Plans (as defined below). If Governmental Approved Plans materially differ from the Tenant Initial Approved Plans, Tenant shall notify Landlord, in writing within fifteen (15) days of its receipt of Governmental Approved Plans: either (a) that Tenant accepts the Governmental Approved Plans as the Tenant Final Approved Plans; or, (b) that Tenant rejects the Governmental Approved Plans and identifying the reasons for Tenant’s rejection. In the event that Tenant rejects the Governmental Approved Plans, Landlord shall, for a period not to exceed one hundred twenty (120) days following the date of Tenant’s rejection notice (the “Modification Period”), diligently pursue modifications to satisfy both Laws and Tenant. If during the Modification Period, plans cannot be prepared which meet the requirements of Laws and Tenant, then Landlord shall notify Tenant in writing and Tenant shall have thirty (30) days after the receipt of Landlord’s notice to either accept the Governmental Approved Plans or terminate this Lease, in which case neither party shall have further liability or obligation to the other.
(D)Tenant Final Approved Plans. “Tenant Final Approved Plans” shall mean those plans approved by the Applicable Governmental Agencies and which are accepted by Tenant. The Tenant Final Approved Plans shall be incorporated into this Lease by reference. The Tenant Final Approved Plans, as same may be modified pursuant to Section 4, shall be the controlling documents for the construction of Landlord’s Work. Any substitutions or variations from the Tenant Final Approved Plans must be approved in writing by Tenant.
4.CHANGE ORDERS. Tenant may, at any time before the Completion Date, order additions to, deletions from, or alterations of Landlord’s Work (a “Change Order”). Before any Change Order is made, Tenant shall specify the Change Order in detail and in writing. Promptly after receipt of the written specifications for the Change Order, Landlord shall submit to Tenant a detailed change order memorandum (a “Change Order Memorandum”) itemizing the estimated Actual Cost (as defined below) of the proposed Change Order, and the impact to the Construction Commencement Date and Fixed Completion Date, if any, necessary to implement the Change Order. Tenant shall promptly either execute and return such Change Order Memorandum or notify Landlord in writing that it retracts the Change Order request. Only a fully executed Change Order Memorandum shall be binding on Tenant and such Change Order Memorandum shall be incorporated by reference into the Tenant Final Approved Plans upon full execution of same.
The cost or savings of change orders shall be equal to the Actual Cost to Landlord of the Change Order. As used herein, “Actual Cost” shall mean design fees, if any, and the cost of direct labor, materials, equipment, supervision fee (not to exceed seven percent (7%) of the cost of Landlord’s Work), permits and any additional fees associated with the Change Order, if any. Upon Tenant’s execution and delivery of the Change Order Memorandum, Landlord shall deliver to Tenant a detailed accounting for such change order showing the Actual Cost, together with supporting invoices and other documentation therefor, and Tenant shall pay such amounts within thirty (30) days after receipt thereof.
5.ZONING AND APPROVALS. Landlord represents and warrants to Tenant that, as of the Rent Commencement Date, all of Landlord’s Work shall be Completed in compliance with all applicable Laws and Permitted Encumbrances and Tenant’s intended use of the Demised Premises as an office and medical laboratory facility (collectively, “Tenant’s Intended Use”) shall be permitted by and in compliance with all Laws and Permitted Encumbrances.
(A)Zoning. Landlord shall secure, at Landlord’s cost and expense, any zoning approvals required for Tenant’s Intended Use and shall, prior to commencing construction, deliver to Tenant a zoning letter from the Town of Leland, Planning and Zoning Department confirming that the zoning designation of the Demised Premises permits Tenant’s Intended Use (the “Zoning Letter”).
(B)Approvals. Landlord shall secure and pay for all approvals required for Tenant’s occupancy of the Demised Premises; provided, however, Tenant shall obtain all licenses required to operate its business. In the event a loss is suffered due to Landlord’s failure to obtain any approval required by this Lease, Landlord shall indemnify and hold the Tenant Indemnitees (as defined in Section 24) harmless from and against any and all claims, expenses (including, but not limited to, attorneys’ fees),
costs, liabilities, damages, causes of action or judgments which may arise or come about by Landlord’s failure to obtain any approvals that were required for tenant’s occupancy of the Demised Premises or use of the Demised Premises for Tenant’s Intended Use.
(C)Mutual Cooperation. Landlord and Tenant shall, upon the request of the other and at no cost to the requested party, cooperate with the requesting party in securing any variance(s) or approval(s) sought by the requesting party in connection with the development of the Land as required under this Lease and the occupancy and use of the Demised Premises or the operation of business thereon. Such cooperation may include but shall not be limited to signing any application and/or appearing at any hearing or before any board or commission as may be required.
6.CONSTRUCTION COMMENCEMENT AND CONSTRUCTION COMPLETION.
(A)Commencement. Landlord shall acquire fee simple title to the Land and commence construction of Landlord’s Work no later than thirty (30) days after the issuance of a building permit by the Building Inspection department with jurisdiction over same, and provided that Tenant provides all information necessary for Landlord to complete the building plans within thirty (30) days of the Effective Date, then construction shall commence no later than the date that is one hundred fifty (150) days after the Effective Date (the “Construction Commencement Date”). If Landlord has not acquired fee simple title to the Land and commenced construction of Landlord’s Work by the Construction Commencement Date, except for delays caused by a Force Majeure Event (as defined in Section 36 below) Tenant may terminate this Lease by providing written notice to Landlord, such termination to be effective on the date that is sixty (60) days following the date of such notice unless Landlord provides Tenant with reasonably sufficient evidence in writing that Landlord has acquired fee simple title to the Land and construction of Landlord’s Work has commenced prior to the expiration of such sixty (60) day period following the date of Tenant’s notice, in which case Tenant’s termination notice shall become null and void and shall be of no further force and effect. Commencement of construction shall be deemed to have occurred when Landlord makes its first physical alteration to the Land provided Landlord continues to pursue meaningful progress on the construction of Landlord’s Work.
(B)Definitions. Landlord’s Work shall be deemed “Complete”, “Completed” or to have achieved “Completion” (or any other grammatical variation thereof) when Landlord’s Work has been constructed in accordance with all Laws, Permitted Encumbrances and the Tenant Final Approved Plans, subject only to the completion of punch-list items which can be corrected or completed without interference with Tenant’s use or occupancy of the Demised Premises (as determined by Tenant), as evidenced by receipt of (i) an AIA certificate of substantial completion from Landlord’s architect for
Landlord’s Work confirming its substantial completion in accordance with the Tenant Final Approved Plans and (ii) a Certificate of Compliance and Certificate of Occupancy, as applicable, or its equivalent required by applicable Laws for occupancy and use of all Landlord’s Work. That date Landlord Completes Landlord’s Work shall be referred to herein as the “Completion Date”. If Tenant initially occupies the Demised Premises following receipt of a Temporary Certificate of Occupancy, Landlord agrees to diligently pursue a final Certificate of Occupancy as soon as reasonably practicable thereafter.
(C)Completion Deadlines. Landlord shall provide to Tenant written notice no fewer than thirty (30) days prior to the date that Landlord anticipates that Landlord’s Work will be Complete (the “30-Day Notice”). Landlord covenants that Landlord’s Work shall be Completed by the date identified in the 30-Day Notice, but not later than three hundred sixty (360) days after the Effective Date (the “Fixed Completion Date”). If the Completion Date does not actually occur on or before the Fixed Completion Date, Tenant may terminate this Lease by written notice to Landlord and effective as of the date of such notice, this Lease shall be of no further force and effect. In addition and notwithstanding the generality of the foregoing, in the event that Landlord’s Work is not Completed within thirty (30) days after the Fixed Completion Date, Tenant shall receive a credit against the Base Rent due hereunder equal to one (1) day of Base Rent with respect to the Demised Premises for each day that elapses between the Fixed Completion Date, and the Completion Date, which credit(s) shall be applied to the Base Rent payments due following the Rent Commencement Date.
(D)Tenant Specific Upfit. Landlord shall be solely responsible for the cost of constructing the Building and the Demised Premises to “Wann Vanilla Shell” condition as part of the construction of the Building, as set forth above in the definition of “Landlord’s Work”. As used in this Lease, the term “Wann Vanilla Shell” condition shall mean the completed construction of the Building shell with sheet rock and electrical outlets on all four perimeter walls, dropped ceilings with lighting, plumbing/restrooms, flooring treatment, elevator and stairs. Tenant agrees that the Tenant’s upfits to the Building in excess of “Warm Vanilla Shell” condition are unique to Tenant’s use of the Building, and require specialized design and materials (the “Required Tenant Improvements”). Accordingly, Landlord shall provide Tenant a construction allowance relative to the construction of the Required Tenant Improvements (i.e., those improvements to be made to the Demised Premises as part of Landlord’s Work in excess of Warm Vanilla Shell condition of the Building) in an amount not to exceed EIGHTY DOLLARS ($80.00) per Rentable Area of the Demised Premises (the “Tenant Improvement Allowance”). Promptly following the date hereof, Tenant will work with Landlord’s space planner to develop a space plan for the Required Tenant Improvements that is reasonably acceptable to Landlord (the “Space Plan”). Following mutual approval of the Space Plan, Landlord shall submit to Tenant, as part of the Landlord Proposed Plans, a. set of construction drawings, a cost budget for the Required
Tenant Improvements consistent with the approved Space Plan, and bids, contracts or estimates supporting the cost budget. Tenant may propose changes to the scope of Required Tenant Improvements in accordance with the procedure relative to the review and approval of the Landlord’s Proposed Plan pursuant to Section 3 above. If the estimated budget of actual costs to construct the Required Tenant Improvements are expected to exceed the Tenant Improvement Allowance the Tenant shall be allowed to notify Landlord of any items in the Tenant Final Approved Plans that Tenant desires to change within five (5) business days after Landlord’s notice thereof to Tenant. Tenant agrees that upon completion of the Required Tenant Improvements and delivery of the Demised Premises to Tenant by Landlord, Tenant shall reimburse Landlord for the Actual Costs incurred by Landlord in completing the Required Tenant Improvements that exceed the Tenant Improvement Allowance. Landlord agrees to provide Tenant a detailed itemization of the Required Tenant Improvements installed by Landlord together with receipted paid bills therefor, it being understood and agreed that Tenant is responsible for all costs and expenses incurred by Landlord which exceeds the Tenant Improvement Allowance.
(E)Punchlist. Within seven (7) days after the Completion Date, Landlord’s representative and Tenant’s representative shall conduct a walk-through of the Demised Premises and identify any necessary touch-up work, repairs and minor completion items that are necessary for final completion of the Landlord’s Work. Landlord shall complete all punch-list items and shall provide Tenant with evidence of the completed punch-list within thirty (30) days after the Completion Date.
(F)Early Access. If and to the extent permitted by Laws, Tenant shall have the right to enter the Demised Premises after receipt of the 30-Day Notice in order to move and install furniture, fixtures and equipment and otherwise prepare the Demised Premises for operation of business. During any such early entry in the Demised Premises, Tenant shall comply with all terms of this Lease except for the obligation to pay Rent until the occurrence of the Rent Commencement Date. Any such early entry shall not interfere with Landlord’s construction activities, nor shall it impair Landlord’s ability to timely procure a Certificate of Occupancy or Certificate of Compliance from the governmental agency with jurisdiction of same.
7.CONSTRUCTION WARRANTIES.
(A)One-Year Warranties. Landlord shall remedy, at its sole cost and expense, any defects due to defective workmanship, materials or equipment and pay for any damage to other portions of Landlord’s Work resulting from such defects and/or the remedying thereof, which shall be discovered within one year following the Completion Date (“Landlord’s One-Year Warranty”). In addition to and not in lieu of Landlord’s One-Year Warranty, Landlord shall furnish to Tenant, without demand, no later
than sixty (60) days after the Completion Date, a written warranty from Landlord’s general contractor, for the benefit of Tenant, of all workmanship, materials and equipment provided to construct the Building, Landlord’s Work and the Required Tenant Improvements for a period of one (1) year commencing on the Completion Date (“Contractor’s One-Year Warranty”). Nothing contained in this Section shall be construed to establish a period of limitation with respect to other obligations which Landlord may have under the Tenant Final Approved Plans. Establishment of the Landlord’s One-Year Warranty and the Contractor’s One-Year Warranty relate only to the specific obligation to correct defective work, and has no relationship to the time within which the obligation to comply with the Tenant Final Approved Plans may be sought to be enforced, nor to the time within which proceedings may be commenced to establish Landlord’s liability with respect to Landlord obligations other than specifically to correct defective work. Tenant shall give Landlord notice of observed defects with reasonable promptness. Landlord shall replace or remedy the defective work within thirty (30) days after the date of Tenant’s written notice to Landlord. Should Landlord fail to replace or remedy the defective work within such thirty (30) day period, Tenant shall have the right to make such corrections and Landlord expressly authorizes Tenant to offset the cost thereof against future rent payments.
(B)Equipment Warranties. Landlord shall secure (i) the manufacturer’s warranty for the roof, including materials and labor, for a period of at least fifteen (15) years from the Completion Date, and (ii) the manufacturer’s warranty for the HVAC compressors and heat exchangers for a period of at least five (5) years from the Completion Date.
8.TERM. The initial term of this Lease shall commence on the Rent Commencement Date and expire on the last day of the calendar month that is ten (10) years following the Rent Commencement Date (the “Initial Term”) unless sooner terminated as permitted herein. Provided Tenant is not then in default of this Lease beyond any applicable notice and cure periods and Tenant or its Permitted Assignee has continuously leased the Demised Premises in good faith throughout the Term of this Lease, Tenant shall have the right to extend the term of this Lease for four (4) successive periods of five (5) years each (each an “Extension Term”), upon the same terms and conditions as herein set forth, except as to number of Extension Terms remaining and Rent payable for such Extension Term. The Initial Term together with any Extension Term, properly exercised, shall be referred to as the “Term”. Tenant may extend the Term of this Lease for an Extension Term only by giving Landlord written notice thereof not less than one hundred eighty. (180) days prior to the expiration of the Initial Tenn, or of any Extension Tenn, as applicable. In the event Tenant does not give notice exercising its right to extend the Tenn of this Lease, Tenant waives its right to all subsequent Extension Tenns.
9.RENT.
(A)Rent. The rent provided in this Lease shall commence upon the date the Landlord delivers the Demised Premises to Tenant with Landlord’s Work Completed (the “Rent Commencement Date”). Promptly following the Rent Commencement Date, Tenant shall execute the acknowledgement of the Rent Commencement Date in substantially the form attached to the Lease as Exhibit “F” and made a part thereof, acknowledging, among other things, that Tenant has accepted the Demised Premises. The base rent (“Base Rent”) during the first Lease Year (as defined below) of the Tenn shall be TWENTY-FIVE DOLLARS ($25.00) per square foot of Rentable Area of the Demised Premises. Thereafter, Rent shall increase annually during the Tenn (including, without limitation, any properly exercised Extension Term) by two and one-half percent (2.5%) over the Base Rent of the prior year. By way of example, the Base Rent in the second Lease Year shall increase to TWENTY-FIVE AND 63/l00THS DOLLARS ($25.63) per square foot; Base Rent in the third Lease Year shall increase to TWENTY SIX AND 27/l00THS DOLLARS ($26.27) per square foot, and so forth. The term “Lease Year” shall mean each 12-month period of the Term commencing on the Rent Commencement Date and every anniversary thereof, provided that if the Rent Commencement Date is on a date other than the first day of the month, the first Lease Year shall be extended by the number of days from the Rent Commencement Date until the first day of the next succeeding month. It is the intention of the parties hereto that the Rent provided for herein shall be received and enjoyed by the Landlord as a net sum free from all imposition, charge, fee, utility bill, maintenance expense, premium, or other expense or cost, except state, or federal income taxes assessed against the Landlord or state, or federal capital levy, franchise, estate, succession, inheritance, or transfer taxes of the Landlord.
(B)Rent Payment. Commencing on the Rent Commencement Date, Tenant shall pay its Rent monthly in advance on or before the first day of each month without offset or demand. All Rent provided for in this Lease shall be prorated for any partial month at the beginning or end of the Initial Term or any Extension Term, as the case may be. Tenant’s pro rata rent obligation shall be in the ratio of the number of days in the partial month for which Tenant has a rent obligation divided by the number of days in the month and multiplied by the applicable monthly rent as set out above. Tenant shall make Rent checks payable to CIGG, LLC, and mail them to the following address: 3415 South College Road, Wilmington, NC 28412, or such other location as may be designated by Landlord to Tenant in writing from time to time in accordance with Section 33 of this Lease.
(C)Electronic Fund Transfer. In the event that the financial institution of the payee identified under Subsection C above accepts the electronic transfer of funds, Tenant may, at its election, make payments of Rent and all other sums which may come due hereunder from time to time by electronic fund transfer. Landlord shall complete all applications and do all other things as may be reasonably required to authorize such electronic fund transfers.
(D)Additional Rent. Any amount required to be paid by Tenant hereunder (in addition to Base Rent) and any charges or expenses incurred by Landlord on behalf of Tenant under the terms of this Lease shall be considered “Additional Rent” payable in the same manner and upon the same terms and conditions as the Base Rent reserved hereunder, except as set forth herein to the contrary. Base Rent and Additional Rent are collective referred to herein as “Rent”. In addition to the Base Rent specified in this Lease, commencing as of the Rent Commencement Date, Tenant shall pay to Landlord as Additional Rent for the Demised Premises, in each full or partial Lease Year during the Term, an amount equal to the Operating Expenses for a particular Lease Year (the “Annual Operating Expense Adjustment”). Landlord and Tenant acknowledge and agree that so long as Tenant is leasing all of the rentable square footage of the Building, Tenant shall be responsible for 100% of the Operating Expenses incurred by Landlord, but that such amount shall be reduced proportionately in the event of any assignment or sublease permitted by this Lease or other reduction of the Demised Premises as may be agreed by Landlord or Tenant. As used herein “Operating Expenses” shall mean the amount of all of Landlord’s costs and expenses paid in connection with utilities (if any), taxes and governmental assessments payable pursuant to Section 21 below, trash removal service, drainage system expense, landscaping maintenance, management fees, professional service fees and expenses (including reasonable attorneys’ fees and accounting fees), parking area maintenance costs, insurance premiums for any insurance coverage requirements under this Lease, any and all assessments imposed by any Property Owners Association with jurisdiction over the Demised Premises, and any and all other costs and expenses attendant with the Landlord’s obligations with respect to the operation, maintenance and repair of the Demised Premises. Operating Expenses shall not include (i) replacement of or structural repairs to the roof structure or the exterior walls; (ii) repairs to the extent covered by insurance proceeds that are actually received by Landlord, or paid by Tenant or other third parties; (iii) alterations solely attributable to third parties other than Tenant; (iv) marketing expenses; (v) any cost or expense associated with compliance with any Laws if such condition existed prior to the Rent Commencement Date; (vi) amortization of principal or interest on account of any indebtedness; (vii) any legal expenses arising out of any misconduct or negligence of Landlord or any person for which Landlord is responsible or arising out of dealings between any principals constituting Landlord or arising .out of any leasing, sale or financing of the Building or the Land or any part of either of them. Within sixty (60) days after the end of Lease year, Landlord and Tenant agree to calculate the actual Operating Expense for the prior twelve month period, and if such calculation results in the Tenant having underpaid the actual Operating Expense for such period, within thirty (30) days after demand is made by Landlord, Tenant shall pay any deficiency. Additionally, Landlord shall estimate the Annual Operating Expense Adjustment annually, and written notice thereof shall be given to Tenant prior to the beginning of each calendar year. In no event may any such Annual Operating Expense Adjustment exceed, by more five percent (5%), the actual
Operating Expenses for the immediately preceding Lease Year. Tenant shall pay to Landlord each month, at the same time the Base Rent is due, an amount equal to one-twelfth (1/12) of the estimated Annual Operating Expense Adjustment. If Landlord does not furnish any such Annual Operating Expense Adjustment subsequent to the commencement thereof, then until the first day of the second month following the month in which such estimate is furnished to Tenant, Tenant shall pay to Landlord on the first day of each month an amount equal to the monthly sum payable by Tenant to Landlord under this Section in respect of the last month of the preceding Lease Year, and after such estimate is furnished to Tenant, Landlord shall refund Tenant any overpayment or if there is a deficiency, Tenant shall pay any deficient amount to Landlord thirty (30) days after demand therefor.
(E)Audit. Tenant, upon notice given at any time, may elect to have Tenant’s designated representative (in such notice) examine such of Landlord’s books and records (collectively “Records”) as are reasonably relevant to review Operating Expenses. “Records” include sales tax reports and sales tax returns, receipts, bank and check books and records supporting data maintained by Landlord and related to Operating Expenses. Tenant’s representative shall be permitted to examine the Records at the office of Landlord or Landlord’s managing agent at such time or times during normal business hours as Landlord shall reasonably designate and Tenant shall be entitled, at its expense, to make copies of Records reasonably relevant to the Operating Expenses for the Lease Year in question. Tenant and Tenant’s employees, accountants and agents shall treat all Records as confidential, and, upon request by Landlord, shall confirm such confidentiality obligation in writing prior to such examination. In no event shall the preceding sentence be deemed to limit Tenant’s rights of discovery and disclosure in any action or proceeding, or be construed so as to prohibit Tenant from complying with the directive of any court or arbitrator. Within one hundred twenty (120) days after the date on which the Records are made available to Tenant in response to a request by Tenant, Tenant may send a notice (“Tenant’s Statement”) to Landlord that Tenant disagrees with the applicable Annual Operating Expense Adjustment, specifying in reasonable detail the basis for Tenant’s disagreement and the amount of the Operating Expenses that Landlord claims is due from Tenant for such Lease Year. Landlord and Tenant shall thereafter meet and attempt in good faith to resolve such disagreement. If the parties fail to resolve such disagreement within ninety (90) days after the date when Tenant gives Tenant’s Statement to Landlord, then either Landlord or Tenant may notify the other that such party desires to have such disagreement determined by arbitration in accordance with the terms of this Lease. Pending resolution of such disagreement, (A) Tenant shall pay the undisputed portion(s) of the Annual Operating Expense Adjustment in accordance with the Annual Operating Expense Adjustment in question and (B) Tenant shall not be deemed to be in default under this Lease for withholding payment of the disputed portion(s). If the arbitrator shall determine that any disputed portion of such Annual Operating Expense Adjustment was required to be paid by Tenant,
Tenant shall pay to Landlord within, thirty (30) days after such determination, such disputed amount. If it is determined as a result of any examination by Tenant of Landlord’s Records that the sums payable by Tenant in respect of Operating Expenses for any Lease Year exceeded the actual payment of Operating Expenses for such Lease Year by more than five percent (5%), then, in addition to amounts payable by Landlord, Landlord shall reimburse Tenant for the out-of-pocket cost of Tenant’s examination. Landlord shall retain all Records relating to payment of Operating Expenses until Tenant no longer has the right to examine such Records. Tenant’s payment of any Operating Expenses shall not preclude Tenant from later disputing the correctness of any Operating Expenses within the time periods set forth above. The foregoing right of audit shall only apply to Operating Expenses paid by Tenant during any twelve-month period for up to three (3) years prior to the date of the Tenant’s audit demand. Landlord shall not be required to retain any Records for any period exceeding three (3) years.
10.SECURITY DEPOSIT. Tenant and Landlord have previously entered into that certain Build to Suit Development Agreement dated January 29, 2021 (the “Development Agreement”), whereby Tenant has paid to Landlord the sum of ONE HUNDRED FIFTY THOUSAND and NO/lO0THS DOLLARS ($150,000.00) to secure payment of certain architectural and design fees incurred by Landlord on Tenants behalf in designing the Building to Tenant’s specifications. Upon Tenant’s occupancy of the Demised Premises and opening for its business therein, Landlord shall deposit the full amount of such payment ($150,000.00) into an Escrow Account at First Bank located in Wilmington, North Carolina to be held as the Security Deposit under this Lease (the “Security Deposit”). The Security Deposit shall be held during the Term to secure the faithful performance of Tenant’s promises and duties contained herein. Landlord shall not be required to pay any interest to Tenant upon the Security Deposit. Upon any termination of the tenancy herein created, including any month to month tenancy created from a holdover, Landlord may deduct from the Security Deposit amounts sufficient to pay (1) any damages sustained by Landlord as a result of Tenant’s nonpayment of Rent or nonfulfillment of the term of this Lease or breach of any other term or condition of this Lease beyond any applicable notice and cure periods; (2) any damages to the Demised Premises for which Tenant is responsible and has failed to timely repair beyond any. applicable notice and cure periods; (3) any unpaid bills which become a lien against the Demised Premises due to Tenant’s default under this Lease beyond any applicable notice and cure period; (4) any costs of re renting the Demised Premises after the termination of this Lease due to the default of Tenant beyond any applicable notice or cure period; and (5) any court costs incurred by Landlord in connection with terminating the tenancy due to Tenant’s default of this Lease beyond any applicable notice and cure period. After having deducted the above amount, Landlord shall refund to Tenant the balance of the Security Deposit along with an itemized statement of any deductions no later than sixty (60) days after the expiration or earlier termination of this Lease.
11.HOLD OVER. Any holding over by Tenant beyond the Initial Tenn of this Lease or any Extension Tenn thereof shall be on the same terms and conditions as contained herein, except for Rent which shall be at the rate of one hundred fifteen percent (115%) of the last monthly Rent specified in this Lease and shall be a month-to-month tenancy terminable by either party upon thirty (30) days’ prior written notice to the other party.
12.USE: BUSINESS OPERATION: AND QUIET POSSESSION.
(A)Use. Except with respect to the Prohibited Uses and to the extent not prohibited by the Permitted Encumbrances, Tenant may use the Demised Premises for any lawful purpose. Without limiting the foregoing, Landlord acknowledges that Tenant shall have the right, from time to time, to use any portion of the Demised Premises for offices and medical laboratories. The “Prohibited Uses” shall mean those uses prohibited by that Master Declaration Of Protective Covenants For Waterford Commercial Property recorded in Book 1813, Page 786 of the Brunswick County, North Carolina, Registry, and any Amendments or Supplements thereto including but not limited to those recorded in Book 1823, Page 1313; Book 4091, Page 1307; Book 4631, Page 139; and any and all amendments and supplements thereto; all in the Brunswick County Registry (cumulatively referred to as the “Declaration”).
(B)Quiet Possession. Subject to Tenant’s obligation to pay when due to Landlord all sums required hereunder and performing and observing all of Tenant’s obligations hereunder, Tenant shall peacefully and quietly have, hold and enjoy the Demised Premises throughout the Term without interference by Landlord.
(C)Title Exceptions. Tenant acknowledges receipt of the Investors Title Insurance Company commitment number 202112852NCT/RC01, having an effective date of May 13, 2021, together with copies of the documents identified· as exceptions to coverage on Schedule B thereof (collectively, the “Title Report”) for the Demised Premises. Those matters identified on the Title Report, including without limitation, the Declaration, are referred to herein as the “Permitted Encumbrances”.
(D)ADA. Landlord covenants and agrees that, as of the Completion Date, the Demised Premises shall comply in all respects with the Americans With Disabilities Act or any similar federal, state or local statute, law, or ordinance (the “ADA”). In the event the Demised Premises must be modified or any other action relating to the Demised Premises must be undertaken in the future to comply with ADA the responsibility for such modification or action (including the payment of all costs incurred in connection therewith) shall belong to Landlord unless such modification or action is required because
of (i) any special or unique use or activity in the Demised Premises other than Tenant’s Intended Use or (ii) the performance of any alterations within the Demised Premises.
13.ASSIGNMENT AND SUBLETTING BY TENANT. Anything else herein to the contrary notwithstanding, Tenant shall not be allowed to assign its leasehold interest created herein, this Lease, or any of its rights, privileges, or obligations created hereunder without prior written consent of Landlord, which Landlord may withhold in its sole discretion. Any other provision in this Lease to the contrary notwithstanding, the Tenant shall have no right to sublet the Demised Premises, or any part thereof to subtenants without Landlord’s prior written consent which it may withhold in its sole discretion. Landlord’s consent to any assignment, subletting, occupation, or use by another person other than Tenant shall not be deemed to be a consent to any subsequent assignment, subletting, occupation or use by another person other than Tenant, it being understood that Landlord’s rights hereunder are continuing and shall not be exhausted, regardless of the number of times such assignment, subletting, occupation or use shall occur. Provided, however, and notwithstanding the foregoing to the contrary,’ Tenant shall be entitled, without seeking or obtaining the prior written consent of Landlord, to assign this Lease or to sublet the Demised Premises (in whole or in part) to any entity that controls Tenant, that is under common control with Tenant or that is controlled by Tenant (a “Permitted Assignee”), provided that the Guaranty remains in full force and effect and such Permitted Assignee has a tangible net worth, at the time the assignment or sublease transaction is completed, which is not less than the net worth of Tenant named herein as of the Lease Date, and further provided Tenant delivers to Landlord, prior to the effective date of such assignment or sublease transaction involving the Permitted Assignee, documentation reasonably acceptable to Landlord to evidence same. A change in control of Tenant resulting from a merger, consolidation, or a transfer of partnership or membership interests, a stock transfer, or any sale of substantially all of the assets of Tenant shall be deemed a transfer to a Permitted Assignee. In the event that Tenant desires at any time to assign this Lease or sublet all or any part of the Demised Premises to any party other than a Permitted Assignee which satisfies the terms of this Section 13, Tenant shall submit to Landlord in writing at least thirty (30) days prior to the proposed effective date of the assignment or sublease transaction (i) a request for permission to assign or sublet setting forth the proposed effective date (which shall be no less than thirty (30) days after the delivery of such notice to Landlord); (ii) the name of the proposed subtenant or assignee; (iii) the nature of the business to be carried on in the Demised Premises after the assignment or sublease; (iv) the terms and provisions of the proposed assignment or sublease instrument(s); (v) current financial statements of the proposed subtenant or assignee; and (vi) such additional information as Landlord may reasonably request in order to make a reasoned judgment regarding the proposed assignment or sublease.
all or any part of the Demised Premises to any party other than a Permitted Assignee which satisfies the terms of this Section 13, Tenant shall submit to Landlord in writing at least thirty (30) days prior to the proposed effective date of the assignment or sublease transaction (i) a request for permission to assign or sublet setting forth the proposed effective date (which shall be no less than thirty (30) days after the delivery of such notice to Landlord); (ii) the name of the proposed subtenant or assignee; (iii) the nature of the business to be carried on in the Demised Premises after the assignment or sublease; (iv) the terms and provisions of the proposed assignment or sublease instrument(s); (v) current financial statements of the proposed subtenant or assignee; and (vi) such additional information as Landlord may reasonably request in order to make a reasoned judgment regarding the proposed assignment or sublease.
If Tenant assigns, sublets or makes any other transfer of all or any portion of its interest(s) hereunder, Tenant named in this Lease shall remain directly and primarily responsible for the faithful performance and observance of all of the covenants and obligations on Tenant’s part to be performed in this Lease unless otherwise agreed by the parties in writing. Any assignee or subtenant hereunder shall be bound by and shall comply with all of the terms and provisions in this Lease, including, without limitation, the Prohibited Uses. Tenant shall give Landlord prompt written notice of any assignment or sublease that is completed by Tenant pursuant to this Section 13, and each such written notice shall include the complete name and notice address of the assignee or subtenant.
Notwithstanding the generality of the foregoing, Tenant shall be permitted to finance its leasehold interest, Required Tenant Improvements and Tenant’s Prope1ty, and Landlord shall enter into such customary landlord consent and estoppel agreements as any lender to Tenant shall reasonably request. However, Landlord shall under no circumstances be required to subordinate nor hypothecate its fee ownership interest in the Demised Premises to any lender to Tenant, nor to provide a waiver of any default by Tenant under the Lease. In addition, Tenant shall be entitled to enter into equipment leasing arrangements, provided that such arrangements do not result in any security or lien filing on the Demised Premises and/or the Building or grant any rights (other than access and removal) to the Demised Premises.
14.ASSIGNMENT BY LANDLORD. If the Building is sold, Landlord may assign its interest in this Lease to a subsequent owner of the Building without Tenant’s prior consent. Landlord shall notify Tenant of such assignment within fifteen (15) days of any such assignment, including the name, address, phone number and other information for the new Landlord. Landlord shall transfer the Security Deposit to the assignee and Landlord shall require such assignee unconditionally assume in writing Landlord’s obligations hereunder from and after the date of such sale, subject to the terms and conditions hereof.
15.SIGNS. Tenant may install such signs in sizes, number and locations in and around the Demised Premises, including, without limitation, on the exterior fa9ade of the Building, as determined by Tenant, so long as such additional signs are in accordance with applicable Laws and Permitted Encumbrances. Tenant shall obtain, at its sole cost and expense, any permit required for the installation of Tenant’s signs provided Landlord agrees to assist Tenant, at no additional cost to Landlord, in Tenant’s pursuit of any approvals required by Permitted Encumbrances for the installation of Tenant’s signs.
16.REPAIRS AND MAINTENANCE BY LANDLORD. Commencing on the Rent Commencement Date and during the Term, Landlord at its sole cost and expense, shall maintain and as
needed repair or replace, the following components of the Demised Premises in accordance with all Laws and the terms of this Lease:
(i)the drains, gutters and downspouts such that the Building remains weather- tight and storm water is directed away from the foundation;
(ii)the roof, provided however if Tenant installs, affixes, or otherwise places Telecom Equipment on the roof pursuant to Paragraph 20 hereof, the maintenance, repair and replacement of such Telecom Equipment shall be the responsibility of Tenant, and further if Tenant fails to notify, and follow instruction from Landlord’s roofing contractor with regard to the installation of the Telecom Equipment, then Tenant shall be responsible for repairs to and replacement of the roofing system;
(iii)the foundation, exterior walls structural frame, floor slabs and sprinkler systems of the Building;
(iv)the Exterior Common Areas; and,
(v)all exterior utility lines and pipes to the point of entry to the Building which are not otherwise maintained or operated by a utility company; provided, however, Landlord shall be responsible for pursuing a utility company to make any repairs necessary.
Landlord has the right and responsibility to enter the Demised Premises periodically, at any reasonable time and upon reasonable advance notice to Tenant, to inspect the condition of the Demised Premises and to perform any maintenance, repairs or replacements required to be performed by Landlord hereunder. Except in the case of emergency, Landlord and Landlord’s agents and representatives shall be accompanied by an agent or employee of Tenant during any access or inspection of the Demised Premises. Landlord agrees to use commercially reasonable efforts to avoid contacting or materially interfering with Tenant’s employees or invitees and their confidential business records when accessing the Demised Premises as permitted hereunder. Any maintenance, repairs, or replacements by Landlord shall be performed diligently and in a manner so as to cause the least interference possible with Tenant’s business operation. If Landlord shall complete any maintenance, repair or replacement, of which it receives notice from Tenant, within fifteen (15) days after the date of Tenant’s notice to Landlord (or sooner in the event of an emergency); provided, however, if any required maintenance, repair, or replacement cannot reasonably be completed within such fifteen (15) day period, Landlord shall commence same within such fifteen (15) day period and diligently pursue same to completion, but in no event shall such completion extend beyond sixty (60) days after the date of Tenant’s notice. Should Landlord fail to complete any required maintenance, repair or replacement within the time provided in
this Section, Landlord’s failure shall constitute an event of default hereunder without further notice from Tenant, and Tenant shall have the right to perform such maintenance, repair and replacement, and Landlord expressly authorizes Tenant to offset the cost thereof against future rent payments, in addition to any other remedies available to Tenant under this Lease or at law or in equity.
17.REPAIRS AND MAINTENANCE BY TENANT. Commencing on the Rent Commencement Date and during the Term, Tenant shall, except for Landlord’s obligations under Section 16, be responsible, at its sole cost and expense, for all maintenance, repairs, and replacements of the Demised Premises, including but not limited to:
(a)repairing and replacing glass, windows and doors, and except when damage is caused by latent defects (which shall be the Responsibility, of Landlord);
(b)the roof, if Tenant installs, affixes, or otherwise places on the roof Telecom Equipment pursuant to Paragraph 20 hereof;
(c)maintaining the interior of the Building;
(d)maintaining all elevators serving the Building, which shall include Tenant entering into an elevator service agreement for periodic servicing of the elevator such that it at all times is operating and in compliance with all applicable laws and regulations;
(e)maintaining, repairing and replacing the electrical and plumbing systems and equipment, and all utility lines serving the Building located within the Building;
(f)maintaining all HVAC systems serving the Building, which shall include Tenant entering into a HVAC maintenance agreement for the periodic servicing of the HVAC systems with a licensed HVAC contractor, all at the expense of Tenant;
(g)maintaining, repairing and replacing Tenant’s building sign(s) and the pole, lamps, box and sign face(s) of Tenant’s pylon and/or monument sign, if any; and,
(h)repairs or replacements necessitated by the negligence or intentional misconduct of Tenant, and/or its agents, contractors or employees, including items which would be Landlord’s responsibility under Section 16 but for such negligence or intentional misconduct, or Tenant’s failure to observe or perform any of its covenants or obligations under this Lease.
Tenant shall provide to Landlord copies of all service and/or maintenance agreements set forth herein as Tenant’s responsibility.
18.SURRENDER OF DEMISED PREMISES. Upon the expiration of the Term, Tenant shall peaceably surrender the Demised Premises to Landlord in the same condition in which they were received from Landlord on the Completion Date, except: (i) as altered as permitted or required by this Lease or Laws; (ii) for repairs, replacements and maintenance required to be performed by Landlord; and (iii) for ordinary wear and tear; provided, however, Tenant shall have no obligation or liability to Landlord for conditions resulting from a Damage Event (except as otherwise expressly provided herein) or Condemnation Proceedings. Tenant shall remove Tenant Property from the Demised Premises upon expiration of the Term.
19.UTILITIES, EASEMENTS, JANITORIAL, Etc.
(A)Utilities. Tenant may select its utility service providers. Tenant shall pay directly for all utilities used by Tenant in the Demised Premises as of the Rent Commencement Date and during the Tenn. As used herein the term “utilities” shall include, but shall not necessarily be limited to, all electric, gas, internet, cable, telephone, water, sewer (including any grinder pumps), janitorial, and trash removal services.
(B)Easements and Licenses. Within fifteen (15) days after Tenant’s request, Landlord shall grant and provide, in writing, any easements or licenses reasonably required by any utility providers for which Tenant seeks services for the operation of its business, including internet or telecommunication services.
(C)Common Area Assessments. The costs incurred by Landlord in performing the maintenance, repair and replacement obligations set forth in Section 16 above in good condition and repair and in compliance with all Laws for a particular Lease Year shall be included in Operating Expenses (except as may otherwise be set forth above) and paid by Tenant as Additional Rent pursuant to Section 8 above. The cost of any Operating Expenses that are capital in nature shall be amortized over the useful life of the improvement (as reasonably determined by Landlord and Tenant), and only the amortized portion shall be included in Operating Expenses. As used herein, “Exterior Common Area” shall mean those areas and facilities provided for the use or benefit of tenants generally and/or the public, including the exterior of the Building and all Site Improvements on the Land, including, without limitation; (A) all parking areas, enclosed or otherwise, and all streets, sidewalks, signs and landscaped areas located on or within the Land; (B) all improvements and landscaped areas located in public rights-of-way directly contiguous to the Land if and to the extent Landlord maintains areas from time to time;
(C) all driveways and paved areas servicing the Building; (D) landscaped areas on the Land; and (E) any signage that is not the property of Tenant and landscaping improvements (including any lighting improvements, irrigation improvements, fountain or other water amenity improvements or other similar improvements associated with or constituting an integral part of the signage and landscaping improvements) located on the Land.
20.ALTERATIONS.
(A)General. Tenant shall not make any structural alterations or additions to the Demised Premises without first obtaining Landlord’s written consent, however, non-structural alterations are hereby permitted. Tenant shall comply in all material respects with all Laws in its construction of any alterations.
(B)Telecom Equipment. Additionally, Tenant shall have the exclusive right, at no additional charge, to use a reasonable percentage of all of the risers located in the Building to install, operate, maintain, replace and remove electrical and telecommunications wiring and related equipment therein (e.g., antennae, satellite dish(es), microwave dishes, signal repeaters, computer systems and like equipment, and/or other communications and/or television equipment, together with related equipment, mountings and supports) (collectively, “Telecom Equipment”). Tenant shall also have the right, in its sole and absolute discretion without charge by Landlord, to install, operate and maintain (or allow to be installed, operated and maintained by a third party service provider) Telecom Equipment, including wireless office and communications services (specifically including, without limitation, “wi-fi” systems, cellular signal repeaters and/or distributed antenna systems) for wireless communication (“Wireless Network”), within the Demised Premises, including the roof or any setbacks reasonably designated by Landlord, provided that such Telecom Equipment is installed, maintained, and operated exclusively to serve the operations of Tenant without the need for any further consent or approval from Landlord. Without limiting the generality of the foregoing, but subject to applicable Law, nothing contained herein shall be deemed to restrict Tenant’s right to install Telecom Equipment within the Demised Premises. Tenant shall be permitted to access all communications hardlines entering into the Building directly from the street. Landlord shall have no interest in any Telecom Equipment installed by or for the benefit of Tenant, it being understood and agreed that any and all such installations shall be and remain the sole property of Tenant and such installations may be removed from the Demised Premises at any time during the Term hereof by Tenant. Upon Tenant’s removal of any of its Telecom Equipment, or any other of Tenant’s property whatsoever, Tenant shall repair, and as necessary replace, the Demised Premises to the condition that the same was in when Tenant took occupancy, reasonable wear and tear excepted.
the Demised Premises, including the roof or any setbacks reasonably designated by Landlord, provided that such Telecom Equipment is installed, maintained, and operated exclusively to serve the operations of Tenant without the need for any further consent or approval from Landlord. Without limiting the generality of the foregoing, but subject to applicable Law, nothing contained herein shall be deemed to restrict Tenant’s right to install Telecom Equipment within the Demised Premises. Tenant shall be permitted to access all communications hardlines entering into the Building directly from the street. Landlord shall have no interest in any Telecom Equipment installed by or for the benefit of Tenant, it being understood and agreed that any and all such installations shall be and remain the sole property of Tenant and such installations may be removed from the Demised Premises at any time during the Term hereof by Tenant. Upon Tenant’s removal of any of its Telecom Equipment, or any other of Tenant’s property whatsoever, Tenant shall repair, and as necessary replace, the Demised Premises to the condition that the same was in when Tenant took occupancy, reasonable wear and tear excepted.
21.TAXES AND ASSESSMENTS. Landlord shall list the Demised Premises for taxes and Tenant shall pay, as Additional Rent, all ad valorem taxes and all other assessments of whatever kind or nature assessed against the Demised Premises and against or on the furnishings, fixtures, inventory, equipment, leasehold improvements and other prope11y situated or placed upon, in or about the Demised Premises. Landlord agrees to send to Tenant promptly copies of any notices for any such taxes, assessments or other charges if such notices are received by Landlord. Upon reasonable notice to Landlord, Tenant shall have the right to protest any tax assessments in the name of the Landlord and as Landlord’s agent, but without expense to Landlord.
22.INSURANCE.
(A)Tenant at its sole cost and expense, shall procure and keep in force at all times during the Term or any extended Term, fire and extended coverage insurance on the improvements constructed on the Demised Premises, including without limitation any plate glass, in an amount equal to the “full replacement cost” thereof, insuring the interest of Landlord and Tenant. The “full replacement cost” shall be determined from time to time, but not more frequently than once in any twenty-four calendar months at the request of the Landlord, by one of the insurers, or at the option of the Landlord, at its sole cost and expense, by an appraiser, engineer, architect or contractor who shall be mutually acceptable to the parties.
(B)Tenant shall continuously maintain and pay the premium upon a policy of liability insurance during the term of this Lease, naming Landlord as an additional insured and insuring against any loss or liability connected with the above in the amount of FIVE MILLION AND NO/100 DOLLARS ($5,000,000.00) for injuries· to one or more persons in any one accident, and in the amount of FIVE HUNDRED THOUSAND AND NO/100 DOLLARS ($500,000.00) for damage to property.
(C)All such policies of insurance shall be in standard form and shall name Landlord and Tenant as additional insureds as their interests may appear. Premiums for all policies of insurance herein referred to and all renewals thereof shall be paid by Tenant as provided above, on or before the beginning date of the next annual policy or renewal period. If Tenant shall at any time fail to insure or keep insured as aforesaid, Landlord may obtain and maintain such insurance on Tenant’s behalf, and all sums expended by the Landlord for that purpose shall be repayable by Tenant on demand.
(D)In addition to the insurance coverage as set forth above, Tenant shall maintain throughout the Term and any extended Term, at its expense, such other insurance on the Demised Premises in such amounts and against such other insurance hazards which at the time are commonly obtained by Tenant in the normal course of its business.
(E)The limit of any of the insurance required by this section shall not limit the liability of Tenant. The limits of all insurance required herein shall be increased from time to time to reflect the increase in the Consumer Price Index (if defined herein) or as required by good business practice.
(F)During the Term, Landlord shall maintain the following types of insurance: (a) Commercial General Liability Insurance, ISO Form CG 00 01, or its equivalent, covering the Demised Premises and Exterior Common Area against claims for bodily injury or death and property damage, which insurance shall be primary and non-contributory and shall provide coverage on an occurrence basis with a per occurrence limit of not less than $5,000,000 for each policy year, or such higher amount as would be maintained by a commercially reasonable owner of any of the comparable building, which limit may be satisfied by any combination of primary and excess or umbrella per occurrence policies; (b) Special Form Insurance in the amount of the full replacement cost of the Building and Required Tenant Improvements but excluding the Tenant Property (as defined in Section 35), and (c) Business and/or Rent Interruption Insurance to provide Landlord coverage in the event of loss or rent abatement under Paragraph 26 hereof. The expense of insurance premiums and deductibles required under this Paragraph 22(F) shall be an Operating Expense and considered a component of Additional Rent to be paid by Tenant.
23.WAIVER OF SUBROGATION. Notwithstanding anything contained in this Lease to the contrary, Landlord (and its affiliates, property managers and mortgagees) and Tenant (and its affiliates) hereby waive any rights each may have against the other on account of any loss of or damage to their respective property, the Demised Premises, its contents, or other portions of the Building, Landlord’s Work or Land arising from any risk which is required to be insured against by Section 22 above. The special form property insurance policies maintained by Landlord and Tenant as provided in this Lease shall include an endorsement containing an express waiver of any rights of subrogation by the insurance company against Landlord and Tenant, as applicable.
24.INDEMNIFICATION. Landlord and Tenant shall each defend, indemnify and save harmless the other and its guarantor (if any), its affiliates and successors each of their respective directors, officers, partners, employees, representatives, agents and assignees (“Indemnitees”) from all claims, costs, damages, judgments, expenses, fines, liabilities and losses (including reasonable attorneys’ fees, paralegal fees, expert witness fees, consultant fees, and other costs of defense) arising from or as a result of (i) any injury, including death, loss or damage of any kind whatsoever to any person or entity or to the property of any person or entity as shall occur on the Demised Premises during the effectiveness of this Lease and that is caused by the negligence or misconduct of the indemnifying party, its agents, employees
or contractors, or (ii) the indemnifying party’s failure to perform its obligations under this Lease. The indemnities provided herein are ones of first defense and payment, not of reimbursement or surety and shall in no way be limited by or to the amount of insurance carried, or required to be carried hereunder. The obligations of this Section shall survive expiration of the Term or te1mination of this Lease.
25.ENVIRONMENTAL CONDITIONS.
(A)Landlord warrants to Tenant that Landlord neither knows of nor has been advised of any legal or administrative proceedings, claims or alleged claims, violations or alleged violations, infractions or alleged infractions, pertaining to the Demised Premises of any existing or issued statutes, laws, rules, ordinances, or orders permits, and regulations of all state, federal, local, and other governmental and regulatory authorities, agencies, and bodies applicable to the Demised Premises, pertaining to environmental matters, or regulating, prohibiting or otherwise having to do with asbestos and all other toxic, radioactive, or hazardous wastes or material including, but not limited to, the Federal Clean Air Act, the Federal Water Pollution Control Act, or the Comprehensive Environmental Response, Compensation, and Liability Act of 1980, as amended (all hereafter collectively called “Environmental Laws”).
(B)Landlord herein agrees to indemnify and hold harmless the Tenant Indemnitees against and in respect of any and all damages, claims, losses, liabilities and expenses, including without limitation, reasonable legal, accounting, consulting, engineering and other expenses which may be imposed upon or incurred by Tenant, its successors or assigns, by any other party or parties (including without limitation a governmental entity), arising out of or in connection with any condition at, on or about the Demised Premises existing prior to the commencement of this Lease, or thereafter to the extent caused by the negligence or willful misconduct of Landlord, its employees, contractors and agents, including the exposure of any person to any such environmental condition, regardless of whether such environmental condition or exposure resulted from activities of Landlord or prior occupants of the Demised Premises. Landlord hereby agrees to promptly remediate or remove, or cause the remediation or removal of, to the extent required by applicable Environmental Laws, any such hazardous materials from the Demised Premises except to the extent required to be removed by Tenant pursuant to this Lease. Any such remediation by Landlord will be accomplished in such a manner so as to minimize interference with Tenant’s use and occupancy of the Demised Premises. If such remediation prohibits or interferes with Tenant’s ability to conduct business from the Demised Premises from and after the Rent Commencement Date, Rent shall be equitably abated during the period of such interference with respect to that p01tion of the Demised Premises interfered with.
(C)Tenant covenants and agrees that it will do nothing that would cause the Demised Premises to be in violation of any of Environmental Laws. Tenant shall execute reasonable affidavits, representations and the like from time to time at Landlord’s request concerning Tenant’s best knowledge and belief regarding the presence of hazardous substances or materials at, on, or about the Demised Premises.
(D)Tenant herein agrees to indemnify and hold harmless Landlord, its successors and assigns, against and in respect of any and all damages, claims, losses, liabilities and expenses, including without limitation, reasonable legal, accounting, consulting, engineering and other expenses which may be imposed upon or incurred by Landlord, its successors or assigns, by any other party or parties (including without limitation a governmental entity), arising out of or in connection with Tenant’s breach of Section 25C.
(E)For the purpose of applying the covenants of Section 25C, the Demised Premises shall also mean, refer to and include all the Tenant Property used in connection with the Demised Premises (including that owned by Tenant) and the soil, ground water and surface water of the Demised Premises.
(F)Landlord and its engineers, technicians and consultants may, from time to time as Landlord deems appropriate, conduct periodic tests and examinations (“Audits”) of the Demised Premises to confirm and monitor compliance with provisions of this Section 25. Such Audits shall be conducted in such a manner as to minimize the interference with Tenant’s permitted activities on the Demised Premises; however, in all cases, the Audits shall be of such nature and scope as shall be reasonably required by then existing technology to confirm compliance with the provisions herein. Tenant shall fully cooperate with Landlord and other persons in the conduct of such Audits. The cost of such Audits shall be paid by Landlord unless an Audit shall disclose a material failure of Tenant to comply with the provisions herein, in which case the cost of such Audit, and the cost of all subsequent Audits made during the Term and within one hundred (100) days thereafter, shall be paid by Tenant on demand. The covenants contained herein shall survive the expiration or termination of this Lease, and shall continue for so long as Landlord and its successors and assigns may be subject to any expense, liability, charge, penalty, or obligation against which Tenant has agreed to indemnify Landlord under the terms hereof.
26.DAMAGE. If the Demised Premises shall be destroyed or damaged, in whole or in part, by fire or other casualty (a “Damage Event”), Tenant shall promptly notify Landlord when Tenant has knowledge of same, then Landlord shall, at its sole cost and expense, diligently repair and restore the
Building to substantially the condition immediately prior to the damage in such manner as to not, to the extent practicable, interfere with Tenant’s conduct of its business in the Demised Premises; provided that, in no event shall Landlord be required to repair or restore the Tenant Property, which shall be repaired and restored by Tenant. At the request of Tenant, Landlord shall, from time to time, promptly inform Tenant of the progress of Landlord’s restoration work and of the estimated date of completion of the same and otherwise consult with Tenant. Notwithstanding the generality of the foregoing, at Tenant’s election, provided that Landlord assigns to Tenant all insurance proceeds payable to Landlord under the property insurance policy, Tenant may elect to restore the portion of the Demised Premises so damaged or destroyed to the same condition, as nearly as possible, as existed prior to such Damage Event in good faith, and with promptness and diligence. In such event, Landlord shall cooperate with Tenant in all ways necessary to expedite the restoration. From the time of any damage or destruction to the Building until the earlier to occur of (i) the date when Landlord redelivers possession of the Demised Premises (or the applicable portion thereof) to Tenant in the condition required above or (ii) or the date when Tenant resumes operations of its business in such portion of the Demised Premises, Rent shall be reduced in the proportion to the Rentable Area of the portion of the Demised Premises that is not usable or reasonably accessible by Tenant for the conduct of its business.
Notwithstanding the generality of the foregoing, if (a) the Demised Premises are totally damaged or are rendered wholly untenantable, (b) if a reputable licensed engineer estimates that the period of time to restore the Demised Premises exceeds three hundred sixty (360) days from the date of such fire or other casualty or (c) there are less than twelve (12) months remaining in the Term and Tenant elects not to exercise its option to extend the Tenn, or no such option is remaining, then in either of such events, Tenant may, not later than ninety (90) days following the date of the damage, give Landlord a written notice terminating this Lease. If this Lease is so terminated, (a) the Tenn shall expire upon the date set forth in such notice, which shall not be less than thirty (30) days after such notice is given, and Tenant shall vacate the Demised Premises and surrender the same to Landlord no later than the date set forth in the notice, (b) Tenant’s liability for rent hereunder shall cease as of the date of the damage, (c) any prepaid Rent for any period after the date of the damage shall be refunded by Landlord to Tenant, and (d) Landlord shall be entitled to collect all insurance proceeds of policies held by Landlord or Tenant providing coverage for alterations and other improvements to the Demised Premises. Landlord shall retain such proceeds from Tenant’s insurance only to the extent that Landlord performed or paid for such alterations and improvements, whether by contribution, offset or otherwise, and the balance of such proceeds, if any, shall be paid to Tenant.
27.CONDEMNATION.
(A)Complete Taking. In the event the entire Demised Premises are taken in Condemnation Proceedings, Tenant may terminate this Lease upon written notice without further liability under this Lease, except for such obligations as may be due as of the date of termination, and Landlord shall refund any unearned rent to Tenant.
(B)Partial Taking. In the event any part of the Demised Premises, or all or part of rights-of-way adjoining or approaches to the Demised Premises, are taken in Condemnation Proceedings (other than widening of adjacent highways that do not affect access to the Demised Premises or loss of any merchandise display area or a taking of a portion of the parking lot that leaves a sufficient number of parking spaces remaining for the operation of Tenant’s business and to meet governmental requirements, which shall be considered a “de minimus” taking) so that in the reasonable business judgment of Tenant the Demised Premises remaining would be unsatisfactory for Tenant’s business operation, Tenant may terminate this Lease upon written notice to Landlord without further liability under this Lease, except for such obligations as may be due as of the date of termination, and Landlord shall refund any unearned rent to Tenant. Should Tenant elect to retain that portion of the Demised Premises not taken, Landlord shall promptly and with due diligence restore the remaining Demised Premises to as close to their condition as existed prior to the taking as is feasible. Commencing on the date Tenant relinquishes possession of that portion of the Demised Premises taken and during the remainder of the Term, rent shall be reduced (i) in proportion to the amount of the Demised Premises lost, except that there shall be no reduction for a de minimus taking, or (ii) if Tenant shall elect, in proportion to the effect of the loss of such area on Tenant’s business. Notwithstanding the foregoing, should Tenant be required to relinquish possession of the entire Demised Premises during the period of restoration, Rent shall abate completely from the time Tenant relinquishes possession of the entire Demised Premises until the first to occur of (i) sixty (60) days after the restoration is Complete and exclusive possession of the Demised Premises is delivered to Tenant, or (ii) the date on which Tenant reopens its business to the public.
(C)Condemnation Proceedings. For purposes of this Lease, the term “Condemnation Proceeding” shall mean a governmental taking of all or any part of the Demised Premises, and shall include conveyances and grants made in anticipation of or in lieu of governmental taking. Tenant may participate in any proceeding pertaining to condemnation of the Demised Premises whether or not Tenant elects to terminate this Lease. Landlord and Tenant shall each be entitled to their separate claims based on their respective interests even if a single award for all damages is given by the condemning authority.
28.LIENS. Subject to Section 29, neither Landlord nor Tenant shall allow any liens to be filed against the Demised Premises arising from work performed, service rendered, or materials supplied
to the Demised Premises by either party or their respective agents, employees, contractors, subcontractors or materialmen. In the event any such lien is filed, Landlord shall, within the statutory time frame (and Tenant shall within thirty (30) days of receipt of Landlord’s notice of the existence of a lien caused by Tenant or Tenant’s agents, employees, contractors, subcontractors or materialmen) discharge or, where allowed by law, bond over such lien.
29.MEMORANDUM OF LEASE. Landlord shall record a memorandum of lease (the “Memorandum of Lease”) executed contemporaneously with this Lease upon the earlier of the following event or occurrence: (i) if Landlord did not own fee simple title to the Land on the Effective Date, the date of Landlord’s acquisition of title to the Land and prior to any mortgage or deed of trust executed in connection with such acquisition, or (ii) no later than five (5) days prior to the furnishing of any labor or materials on the Land or such date as is necessary to preserve the priority of Tenant’s leasehold interest in the Demised Premises over any mechanics’ or materialman’s lien.
30.CONFIDENTIALITY. Landlord and Tenant shall each hold in confidence the existence of and ALL terms and conditions of this Lease (except as set forth in the Memorandum of Lease) unless the party desiring to disclose the confidential information receives the consent of the non-disclosing party. Notwithstanding the foregoing, either party may disclose the terms of this Lease to its appraisers, lenders, accountants, attorneys, and potential buyers, potential subtenants or assignees, or as may be required by court order or Laws; provided, however, the disclosing party shall be responsible for obtaining adequate assuranc.es from such parties that the disclosed terms hereof shall be held in confidence and the disclosing party shall continue to be liable hereunder for any disclosure. For any action brought for the enforcement of rights or remedies under this Section, Landlord and Tenant hereby consent to the exercise of personal jurisdiction over them by any court (federal or state) of competent jurisdiction in the State of North Carolina, waive all defenses for lack of personal jurisdiction and forum non convenience, and agree that this confidentiality provision is to be governed by the laws of the State of North Carolina without regard to its conflicts of laws.
31.SUBORDINATION TO MORTGAGES. Tenant agrees that, conditioned upon Landlord obtaining and delivering to Tenant (which delivery shall be made concurrently with the execution of this Lease), with respect to each Mortgage (as defined below), a subordination, non-disturbance and attornment agreement with such terms and on such form reasonably required by any Mortgage Lender of Landlord executed by Landlord and the lender under such Mortgage, and in recordable form, this Lease Agreement and the rights of Tenant will be subordinate to and shall be subordinate to the lien of any mortgage or deed of trust (hereinafter called a “Mortgage”) whether such Mortgage is currently a lien on the Demised Premises or hereafter becomes a lien on the Demised
Premises and no further agreements or documents shall be required to render this Lease and the Tenant’s rights subordinate to such Mortgage (unless the lender under a Mortgage requests that this Lease be superior to its Mortgage). Tenant shall execute at Landlord’s request, no more than twice per Lease Year and within twenty (20) days after request therefor, an estoppel ce1iificate addressed to (a) any mortgagee or prospective mortgagee of Landlord; or (b) any purchaser or prospective purchaser of all or any portion of, or interest in, the Demised Premises setting forth, to Tenant’s knowledge: the date Tenant accepted possession; that Tenant occupies the Demised Premises; the termination date of its Lease; the date which rent has been paid; the amount of monthly rent in effect as of such certification; whether or not Tenant has knowledge of any default or breach by Landlord; and that this Lease is in full force and effect except as to modifications or amendments thereto, copies of each of which shall be attached to the certificate. Landlord represents and wan-ants to Tenant that, as of the date hereof, (i) the only mortgage, deed of trust, deed to secure debt or other security instrument encumbering all or any portion of the Building or Land is that Deed of Trust in favor of First Bank, and recorded in Book 4631 Page 150 of the Brunswick County, North Carolina, Register of Deeds office; and (ii) there are no ground or underlying lease(s) of or affecting all or any portion of the Building or Land.
32.DEFAULT AND REMEDIES.
(A)Default. Tenant is in default if Tenant:
(i)fails to pay all or any portion of the Rent or any other sum required by this Lease within five (5) business days after the same is due; provided, however, relative to the first (1st) failure to timely pay such sums in any twelve (12) month period, Tenant shall not be in default if Tenant makes full payment within five (5) business days after receipt of written notice of such delinquency;
(ii)fails to cease conduct prohibited by this Lease immediately upon receipt of written notice from Landlord;
(iii)fails to maintain continuous uninterrupted utilities services to the Demised Premises for more than ten (10) consecutive days; provided, however it shall not be a default of Tenant hereunder if Tenant remedies such failure within ten (10) days after written notice from Landlord thereof;
(iv)fails to have any bankruptcy proceedings dismissed within thirty (30) days after filing;
(v)fails to cease committing waste to the Demised Premises for a period of thirty (30) days after written notice thereof from Landlord; provided, however, that if the nature of Tenant’s default is such that more than thirty (30) days are reasonably required to cure, then such default shall be deemed to have been cured if Tenant commences such performance within said thirty (30) day period and thereafter diligently completes the required action within a reasonable time.
(vi)voluntarily makes an assignment for the benefit of creditors or if a receiver or trustee is appointed for Tenant’s property; or
(vii)fails to perform to perform or observe any other term, condition, covenant or obligation required under this Lease for a period of thirty (30) days after written notice thereof from Landlord; provided, however, that if the nature of Tenant’s default is such that more than thirty (30) days are reasonably required to cure, then such default shall be deemed to have been cured if Tenant commences such performance within said thi1ty (30) day period and thereafter diligently completes the required action within a reasonable time;
(B)Rights and Remedies. Following the occurrence and during the continuance of any default beyond any applicable notice and cure periods, Landlord, without declaring a termination of this Lease (which right is, however, unconditionally and absolutely reserved), may at its election pursue any one or more of the following remedies in addition to any other remedies available to Landlord at law, in equity, or pursuant to the terms of this Lease:
(i)Landlord shall have the right to continue this Lease in full force and effect, and the right to enter the Demised Premises and te1minate Tenant’s possession and right to possess the Demised Premises and relet the same, including without limitation to the right to change any or all locks on the Demised Premises, all without being liable for forcible entry, trespass or other tort. Tenant shall be liable immediately to Landlord for the actual and reasonable costs Landlord shall incur in reletting the Demised Premises and Tenant shall pay to Landlord the Rent that would have been payable under this Lease for the remainder of the Term discounted at the ten-year treasury rate less the rent Landlord actually receives from any reletting the Demised Premises. Rent received from such reletting shall be applied: first to payment of costs incident to reletting and regaining possession; then to any indebtedness to Landlord from Tenant other than for Rent; and any remaining excess shall then be applied to the payment of Rent due and unpaid hereunder. Tenant shall have no right to any proceeds of reletting that remain following application of the proceeds as above.
(ii)Landlord shall have the right to terminate without notice to vacate (any right to which is hereby waived by Tenant) this Lease and Tenant’s rights to possession of the Demised
Premises at any time, and re-enter the Demised Premises, and Landlord shall have the right to immediately recover from Tenant the Rent that would have been payable under this Lease for the remainder of the Term discounted at the ten-year treasury rate less the rent Landlord actually receives from any reletting the Demised Premises.
(iii)Landlord with or without terminating this Lease may immediately or at any other time thereafter re-enter the Demised Premises and cure any event of default and/or correct or repair any conditions which shall constitute a failure on Tenant’s part to perform any obligation to be performed by it under this Lease, and Tenant shall pay Landlord on demand any and all reasonable and actual costs or expenses paid or incurred by the Landlord in making any such cure, correction or repair.
(C)In the event of any re-entry of the Demised Premises and/or changing of the locks on the Demised Premises and/or termination of this Lease by Landlord pursuant to any of the provisions of this Lease following a Tenant default and any applicable notice and cure periods, Tenant hereby waives all claims for damages which may be caused by such re-entry or changing of locks or termination by Landlord, and Tenant shall save Landlord harmless from any loss, cost (including legal expense and reasonable attorney’s fees) or damages suffered by Landlord by reason of such re-entry or changing of locks or termination, and no such re-entry or changing of locks shall be considered or construed to be a forcible entry. No act by Landlord shall terminate this Lease unless Landlord notifies Tenant that Landlord elects to terminate this Lease.
(D)Landlord shall have the right to pursue its remedies at law or in equity to recover from Tenant all amounts then due or thereafter accruing, including Rent, and such other damages as are caused by Tenant’s default, excluding punitive damages.
(E)Notwithstanding anything herein to the contrary, if Landlord has terminated this Lease or Tenant’s right to possession, Landlord agrees to use commercially reasonable efforts to mitigate its damages.
(F)Bankruptcy. If Landlord cannot terminate this Lease because of law, then Tenant, as a debtor in possession or on behalf of any trustee for Tenant, shall (i) within the statutory time, assume or reject this Lease, and (ii) not seek or request any extension or adjournment of any application to assume or reject this Lease by Landlord. In such event, Tenant or any trustee for Tenant may only assume this Lease if (A) it cures or provides adequate assurance that it will promptly cure any default hereunder, (B) it compensates or provides adequate assurance that Tenant will promptly compensate Landlord for any actual pecuniary loss to Landlord resulting from Tenant’s defaults, and (C) it provides adequate assurance of performance during the Term of all of the terms, covenants and provisions of this Lease to
be performed by Tenant. In no event after the assumption of this Lease shall any then-existing default remain uncured for a period in excess of the earlier of ten (10) days or the time period set forth herein. Adequate assurance of performance shall include, without limitation, adequate assurance (1) of the source of rent reserved hereunder, and (2) the assumption of this Lease will not breach any provision hereunder.
(G)Attorney’s Fees and Acceleration. If Landlord requires an attorney to enforce any of the provisions of this Lease, Landlord shall be entitled to all reasonable fees, expenses and costs incurred by it.
33.NOTICES. Except for notices given to terminate the Lease, notices required under Section 3 and Subsections 6C, and 6D may be given via electronic mail to Landlord at ______________________ and to Tenant at _________________ cc: ____________. Notices sent via electronic mail shall be deemed to have been properly served when sent with a delivery receipt and receipt shall be deemed to be the date identified on the delive1y receipt.
Except as othe1wise expressly provided in this Section, all notices required under this Lease shall be given in writing and be deemed to have been properly served when given by any of the following methods:
| | | | | |
| METHODS OF GIVING NOTICE: | RECEIPT DEEMED TO BE: |
| Certified mail, return receipt requested | Date of delivery or rejection, or date notice is deemed unclaimed by carrier. |
| Facsimile | Date on proof of transmission |
| Hand-delivered | Date of personal delivery |
| Nationally recognized courier | Date of delivery or rejection, or date notice is deemed unclaimed by carrier. |
To Landlord at:
CIGG, LLC
c/o ___________, Manager 3514 S. College Road Wilmington, NC 28412
To Tenant at:
[Prior to the Rent Commencement Date]: CYGNUS TECHNOLOGIES, LLC
4332 Southport Supply Rel, SE Southport, NC 28461
Attn: Chief Operating Officer
[Following the Rent Commencement Date]:
CYGNUS TECHNOLOGIES, LLC
(to the mailing address of the Demised Premises)
Attn: Chief Operating Officer
With a copies to:
Maravai LifeSciences
10770 Wateridge Circle, Suite 200 San Diego, CA.92121
Attn: General Counsel
And to:
Troutman Pepper Hamilton Sanders LLP 11682 El Camino Real, Suite 400
San Diego, CA 92130 Attention: _____________ Telephone: ___________
or such other place or places as either party may designate in writing in accordance with this Section, Any notice of transfer of ownership of the Demised Premises shall be accompanied by evidence of transfer of ownership, e.g. deed, court order, deed in lieu of foreclosure.
34.CONSENTS AND APPROVALS.
(A)Landlord’s Consents and Approvals. Whenever it is stated in this Lease that Landlord’s consent or approval is required, such consent or approval shall not be unreasonably withheld, conditioned or delayed. If, in any instance, Landlord refuses to grant its consent or approval, Landlord shall simultaneously give Tenant written notice of the reason(s) for such refusal. If Landlord’s consent or approval is not given or refused within ten (10) days after Tenant’s written request therefor, such consent or approval shall be deemed automatically granted,
(B)Tenant’s Consents and Approvals. Unless otherwise expressly stated in any written notice to Landlord, whenever it is stated in this Lease that Tenant’s consent or approval is required, such consent or approval shall be binding on Tenant only if in a writing signed by an officer of Tenant (authorized Vice President or higher level).
35.TENANT PROPERTY. Tenant’s personal property, merchandise, inventory, furniture, furnishings, signs, equipment, machinery, and trade fixtures, but excluding any generators, walk in coolers, and other machinery and equipment which is affixed to the Demised Premises, (collectively, “Tenant Property”) located at the Demised Premises shall remain the property of Tenant and may be removed from the Demised Premises by Tenant at any time. Tenant shall repair any damage to the Demised Premises caused by such removal. Landlord shall have no lien or other interest whatsoever in any Tenant Property and within ten (10) days following Tenant’s request, Landlord shall execute
documents in reasonable form to evidence Landlord’s waiver of any right, title, lien, or interest in Tenant Property.
36.FORCE MAJEURE. If either party shall be prevented or delayed from punctually performing any obligation under this Lease by reason of a Force Majeure Event (as defined below) the time to perform the obligation shall be extended for a period of time equal in length to the duration of the Force Majeure Event; provided, however, the party claiming a delay due to a Force Majeure Event must notify the other party hereto in writing within ten (1,0) days after such party first becomes aware of the Force Majeure Event likely to cause a delay in order to claim any delay in connection with such event and provided further that in no event will the Fixed Completion Date established in Subsection 6C be extended due to a Force Majeure Event. “Force Majeure Event” shall include only the following: (a) any strike or lockout; (b) an Act of God; (c) governmental restrictions; regulations or controls; (d) war or terrorism; (e) fire or other casualty not resulting from the non-performing party’s negligence or misconduct; (f) the shortage of materials or labor; (g) the failure of the other party to timely perform an obligation, which obligation is a condition to the performance of the obligation for which force majeure is being claimed.
37.GOVERNING LAW: SUBMISSION TO VENUE AND JURISDICTION. Except as provided in Section 30 - CONFIDENTIALITY, this Lease shall be governed by and construed in accordance with the laws of North Carolina, without giving effect to its conflict of laws principles or rules, and Landlord and Tenant hereby consent to the exercise of personal jurisdiction over them by any court of competent jurisdiction within the locus of the Demised Premises in connection with any action brought for the enforcement of rights or remedies under this Lease and waive all defenses of lack of personal jurisdiction and forum non convenience.
38.ATTORNEYS’ FEES: LEGAL COLLECTION. Except for actions for default by Tenant which shall be governed by Section 33 hereof, if either party commences an action against the other party arising out of or in connection with the parties’ obligations under this Lease, the prevailing party shall be entitled to recover from the losing party reasonable attorneys’ fees and costs of suit, including, but not limited to, fees and costs of appeal. In the event that Landlord fails to comply with any court order or executed settlement agreement benefiting Tenant, Tenant may offset from rent and other monies due Landlord under this Lease (i) all monies due to Tenant under such court order or executed settlement agreement, and/or (ii) all costs incurred by Tenant in connection with actions taken in order to effectuate such court order or executed settlement agreement.
39.GUARANTY OF LEASE. Simultaneous with the execution of this Lease by Tenant, Maravai Intermediate Holdings, LLC shall execute a Guaranty of Lease in the form attached hereto as Exhibit “D”. The original Guaranty of Lease shall be delivered by Tenant to Landlord with a fully executed counterpart of this Lease.
40.NO WAIVER. The failure of Landlord or Tenant to insist upon the strict performance of any provision of this Lease, or the failure of Landlord or Tenant to exercise any right, option or remedy contained in this Lease; shall not be construed as a waiver for the future of any such provision, right, option, or remedy, or as a waiver of any subsequent breach. No provision of this Lease shall be deemed to have been waived unless such waiver shall be in writing signed by the party to be charged, except as specifically provided herein.
41.INVALIDITY OF CERTAIN PROVISIONS. If any provisions of this Lease shall be invalid or unenforceable, the remainder of the provisions of this Lease shall not be affected and every other provision of this Lease shall be enforceable to the fullest extent permitted by Laws.
42.CAPTIONS. All captions and headings are for convenience of reference only and in no way shall be used to construe or modify the provisions set forth in this Lease.
43.ENTIRE AGREEMENT. This Lease, its attachments and the documents incorporated into the Lease by reference, if any, contain the entire agreement between the parties, and no covenants exist, express or implied, except as contained herein. No statement, promise or inducement made by either party or agent of either party that is not contained in this written agreement shall be valid or binding. This Lease may not be modified in any manner except by a written instrument executed by the parties hereto or their respective successors or assigns.
44.BINDING EFFECT. This Lease, as of the Effective Date, shall bind and inure to the benefit of the parties hereto, their heirs, successors, executors, administrators, and assigns.
45.TIME IS OF THE ESSENCE. It is expressly agreed that time shall be of the essence of this Lease.
46.LEASE CONSTRUCTION. The parties agree that the interpretation, application and construction of this Lease shall in no way take into consideration the fact that this Lease was drafted by for, or on behalf of either party; both parties having had full opportunity to seek the advice of counsel.
47.COMPUTATION OF DAYS. When referred to in this Lease, days shall mean calendar days unless otherwise provided. Unless otherwise expressly provided herein, any period of time required
to be calculated under the Lease shall begin on the next day after the act, event or default giving rise for such computation. In the event the last day of the period shall fall on a Saturday, Sunday or federally adopted holiday, then the next business day shall be the last day.
48.SUBDIVISION. No later than the Completion Date, Landlord shall have approved and recorded in the Register of Deeds for Brunswick County the Subdivision Plat attached hereto as Exhibit “E” and provide a copy to Tenant within fifteen (15) days after such recordation (the “Subdivision Plat”). Landlord and Tenant acknowledge that it is anticipated that the Town of Leland will extend Olde Waterford Way to intersect with Old Regent Way, as shown upon the Subdivision Plat. Access to the Land and Demised Premises shall be through curb cuts provided by the Town of Leland onto Olde Waterford Way. It is anticipated that the construction of such roadway will be complete by the Completion Date. However, if such construction of Olde Waterford Way is not completed by the Completion Date, Landlord shall provide a reasonable access easement of twenty-five feet (25’) in width for ingress/egress to the Property across Tract 1 shown on the Subdivision Plat until such time that Olde Waterford Way has been completed (the “Access Drive”) such that Landlord shall guaranty Tenant and its employees, contractors, guests and invitees pedestrian and vehicular access to the Demised Premises to and from the public rights of way in compliance with all Laws.
49.INDEMNIFICATION FOR LEASING COMMISSIONS. The parties hereby represent and warrant that Environments Unlimited Commercial Real Estate (Firm) and Jeremy Phillips (Broker) represents the Landlord in this transactions and will be paid by the Landlord pursuant to the terms of a separate agency agreement. Otherwise the Landlord and Tenant represent that no real estate brokers are or have been involved in the negotiation and execution of this Lease and that no other party is entitled, as a result of the actions of the respective party, to a commission or other fee resulting from the execution of this Lease. Each party shall indemnify the other from any and all liability for the breach of this representation and warranty on its part and shall pay any compensation to any other broker or person who may be entitled thereto.
50.REPRESENTATIONS AND WARRANTIES. Tenant hereby represents and warrants that (i) Tenant is duly organized, validly existing and in good standing (if applicable) in accordance with the laws of the State under which it was organized; (ii) Tenant is authorized to do business in the State where the Building is located; and (iii) the individual(s) executing and delivering this Lease on behalf of Tenant has been properly authorized to do so, and such execution and delivery shall bind Tenant to its terms. Landlord hereby represents and warrants that (i) Landlord is duly organized, validly existing and in good standing (if applicable) in accordance with the laws of the State under which it was organized; (ii) Landlord is authorized to do business in the State where the Building is located; and (iii) the individual(s)
executing and delivering this Lease on behalf of Landlord has been properly authorized to do so, and such execution and delivery shall bind Landlord to its terms.
[The reminder of this page is intentionally blank]
[Signatures begin on following page]
IN WITNESS WHEREOF, the parties have signed, sealed and delivered this Lease in multiple counterparts, each of which shall be an original document, as the day and year first above written.
LANDLORD:
CIGG, LLC,
a North Carolina limited liability company
By: /s/ John Gray
John Gray, Manager
TENANT:
CYGNUS TECHNOLOGIES, LLC,
a Delaware limited liability company
By: /s/ Kevin Herde
Kevin Herde, Chief Financial Officer
DocumentExhibit 10.21
EXECUTION VERSION
FIRST AMENDMENT TO CREDIT AGREEMENT
This FIRST AMENDMENT TO CREDIT AGREEMENT (this “Amendment”) is dated as of August 11, 2021 and is entered into by and among MARAVAI INTERMEDIATE HOLDINGS, LLC, a Delaware limited liability company (“Parent Borrower”), and MORGAN STANLEY SENIOR FUNDING, INC. (“MS”), as administrative agent for the Lenders (in such capacity, the “Administrative Agent”), and amends that certain Credit Agreement, dated as of October 19, 2020, by and among Parent Borrower, CYGNUS TECHNOLOGIES, LLC, a Delaware limited liability company (“Cygnus”), TRILINK BIOTECHNOLOGIES, LLC, a Delaware limited liability company (“TriLink”), Vector Laboratories, Inc., a California corporation (“Vector”; and together with the Parent Borrower, Cygnus and TriLink, the “Borrowers” and each, a “Borrower”), MARAVAI TOPCO HOLDINGS, LLC, a Delaware limited liability company (“Holdings”), each lender from time to time party thereto (collectively, the “Lenders”), each L/C Issuer party hereto, the Administrative Agent and MS, as Collateral Agent (the “Credit Agreement”, and as amended by this Amendment, the “Amended Credit Agreement”). Capitalized terms used herein without definition shall have the same meanings herein as set forth in the Amended Credit Agreement.
RECITALS
WHEREAS, pursuant to Section 10.01 of the Credit Agreement, if the Administrative Agent and the Parent Borrower have jointly identified an obvious error or any error, ambiguity or omission, defect or inconsistency of a technical nature, in each case, in any provision of the Loan Documents, then the Administrative Agent and the Parent Borrower are permitted to amend such provision;
WHEREAS, the Parent Borrower and the Administrative Agent have jointly identified an omission of a technical nature in Section 9.11 of the Credit Agreement; and
WHEREAS, the Parent Borrower and the Administrative Agent have jointly agreed to effect this Amendment to correct such omission;
NOW, THEREFORE, in consideration of the premises and the agreements, provisions and covenants herein contained, the parties hereto agree as follows:
SECTION I. AMENDMENTS TO CREDIT AGREEMENT
A.The definition of “Borrowers” is hereby amended by inserting the following proviso at the end of such definition immediately after the words “Loan Document”: “; provided, further, that upon a Borrower that is a Subsidiary of the Parent Borrower ceasing to be a Subsidiary in a transaction not prohibited by the Loan Documents, such Borrower shall automatically cease to be a “Borrower” at such time and shall be released from its obligations under the Loan Documents at such time (it being understood that the other Borrowers shall remain jointly and severally liable for the Obligations as provided in Section 2.20(b) notwithstanding such release)”;
B.Section 9.11(a)(i)(E) of the Credit Agreement is hereby amended by adding the words “or owned by a Borrower that is a Subsidiary of the Parent Borrower upon such Subsidiary ceasing to constitute a Borrower in accordance with the definition of “Borrower” (unless such Borrower remains a Guarantor)” immediately after the words “clause (iii) below”.
SECTION II. CONDITION TO EFFECTIVENESS
The effectiveness of this Amendment is subject to the execution and delivery of this Amendment by the Administrative Agent and the Parent Borrower (the date of such execution, the “First Amendment Closing Date”).
SECTION III. REPRESENTATIONS AND WARRANTIES
The Parent Borrower hereby represents and warrants that, as of the date hereof, this Amendment has been duly executed and delivered by the Parent Borrower and constitutes a legal, valid and binding obligation of the Parent Borrower, enforceable against the Parent Borrower in accordance with its terms, subject to applicable bankruptcy, insolvency, reorganization, moratorium or other laws affecting creditors’ rights generally and subject to general principles of equity, regardless of whether considered in a proceeding in equity or at law.
SECTION IV. MISCELLANEOUS
A.On and after the First Amendment Closing Date, each reference in the Credit Agreement to “this Agreement”, “hereunder”, “hereof”, “herein” or words of like import referring to the Credit Agreement, and each reference in the other Credit Documents to the “Credit Agreement”, “thereunder”, “thereof” or words of like import referring to the Credit Agreement, shall mean and be a reference to the Amended Credit Agreement. This Amendment shall constitute a “Loan Document.”
B.Section headings herein are included for convenience of reference only and shall not affect the interpretation of this Amendment.
C.This Amendment shall be governed by, and construed in accordance with, the law of the State of New York without giving effect to the conflicts of laws principles thereof, but including section 5-1401 of the New York General Obligations Law.
D.The provisions of Sections 10.15(b) and (c), 10.16 and 10.17 of the Credit Agreement pertaining to submission to jurisdiction, waiver of venue, service of process and waiver of right to trial by jury are hereby also incorporated by reference, mutatis mutandis.
E.Except as set forth herein, this Amendment shall not operate as a waiver of any right, power or remedy of any Lender under any of the Loan Documents. This Amendment does not constitute a novation by the Parent Borrower of any of its obligations under any of the Loan Documents.
F.This Amendment may be executed in one or more counterparts (and by different parties hereto in different counterparts), each of which shall be deemed an original, but all of which together shall constitute one and the same instrument. Delivery by telecopier or other electronic transmission of an executed counterpart of a signature page to this Amendment shall be effective as delivery of an original executed counterpart of this Agreement and such other Loan Document. The words “execution,” “signed,” “signature,” “delivery,” and words of like import in or relating to this Amendment or any other documents executed in connection herewith and the transactions contemplated hereby shall be deemed to include electronic signatures, deliveries or the keeping of records in electronic form, each of which shall be of the same legal effect, validity or enforceability as a manually executed signature, physical delivery thereof or the use of a paper-based recordkeeping system, as the case may be, to the extent and as provided for in any applicable law, including the federal Electronic Signatures in Global and National Commerce Act, the New York State Electronic Signatures and
Records Act, or any other state laws based on the Uniform Electronic Transactions Act, and the parties hereto consent to conduct the transactions contemplated hereunder by electronic means.
[Remainder of page intentionally blank]
IN WITNESS WHEREOF, the parties hereto have caused this Amendment to be duly executed and delivered by their respective officers thereunto duly authorized as of the date first written above.
| | |
MARAVAI INTERMEDIATE HOLDINGS, LLC, as Parent Borrower
By: /s/ Kevin Herde Name: Kevin Herde Title: Chief Financial Officer |
MORGAN STANLEY SENIOR FUNDING, INC., as Administrative Agent
By: /s/ Vanessa Roberts Name: Vanessa Roberts Title: Authorized Signatory |
|
Signature Page to First Amendment to Credit Agreement
AMENDMENT NO. 1 TO AMENDED AND RESTATED EMPLOYMENT AGREEMENT
This AMENDMENT NO. 1 TO AMENDED AND RESTATED EMPLOYMENT AGREEMENT (this “Amendment”), entered into on July 27, 2023 (the “Effective Date”), is by and among Maravai LifeSciences Holdings, Inc., a Delaware corporation (“Parent”), Maravai Intermediate Holdings, LLC, a Delaware limited liability company (“Employer”), and Carl W. Hull (“Executive”).
WHEREAS, Executive, Parent and Employer are party to that certain Amended and Restated Employment Agreement, dated as of May 8, 2023 (the “Employment Agreement”);
WHEREAS, Executive’s successor was appointed as chief executive officer of Parent and its Subsidiaries (as defined in the Employment Agreement) effective as of 12:01 am Pacific Time on the Effective Date (the “CEO Succession”); and
WHEREAS, Executive, Parent and Employer desire to amend the Employment Agreement pursuant to the terms and conditions set forth herein in connection with the CEO Succession.
NOW, THEREFORE, in consideration of the foregoing and of the respective covenants and agreements set forth below, the parties hereto agree as follows:
1.Amendment to Employment Agreement.
(a)The definition of “Effective Date” in the preamble of the Employment Agreement shall be deleted in its entirety and replaced with the definition of “Effective Date” in this Amendment.
(b)Section 1(a)(i) of the Employment Agreement titled “Position and Duties” shall be deleted in its entirety and replaced with the following:
“During the Employment Period, Executive shall serve as the Executive Chairman of the board of directors of Parent (the “Board”) and shall have the normal duties, responsibilities and authority implied by such position, which shall include leadership of the Board and its strategic priorities, and assisting the new chief executive officer with the transition, subject in each case to the power of the Board to expand, limit or otherwise alter such duties, responsibilities, positions and authority and to otherwise override actions of officers.”
(c)Section 1(b) of the Employment Agreement titled “Salary, Bonus and Benefits” shall be deleted in its entirety and replaced with the following:
“Commencing on the date of the start of the first payroll period to begin after the Effective Date and continuing until a Separation, Employer will pay Executive a base salary at a rate of $500,000 per annum (the “Annual Base Salary”). The Annual Base Salary shall be reviewed annually by the Board. For each fiscal year of Employer ending during the Employment Period, Executive shall be eligible for an annual bonus with a target amount equal to $757,667 for 2023 and 100% of the Annual Base Salary in subsequent years (such amount, the “Annual Bonus”), as determined by the Board based upon the performance of Executive and the achievement by Parent, Employer and the other Subsidiaries of Parent of financial, operating and other objectives set by the Board. Each Annual Bonus, if any, shall be paid as soon as administratively feasible after the Board (or a committee thereof) certifies whether the applicable performance targets for the applicable year have been achieved but in no event later than March 15 following the end of such year. Notwithstanding anything in this Section 1(b) to the contrary, no Annual Bonus, if any, or any portion thereof, shall be payable for any year unless Executive remains continuously employed by Employer from the Effective Date through the last day of such year. In addition, during the Employment Period, Executive will be entitled to such other benefits as are approved by the Board and made generally available to all senior management of Parent and Employer.”
2.Entire Agreement. Except as amended pursuant to this Amendment, the Employment Agreement remains in full force and effect, and the Employment Agreement, together with this Amendment, sets forth the entire agreement of the parties hereto in respect of the subject matter contained herein and supersedes any and all prior agreements or understandings between Executive, Parent and Employer with respect to the subject matter hereof.
3.Counterparts and Signatures. This Amendment may be executed in counterparts, each of which shall be deemed an original, and together any counterparts shall constitute one and the same instrument. Additionally, the parties hereto agree that electronic reproductions of signatures (i.e., scanned PDF versions of original signatures, facsimile transmissions, and the like) shall be treated as original signatures for purposes of execution of this Amendment.
* * *
IN WITNESS WHEREOF, the parties hereto have executed this Amendment as of the date first written above.
MARAVAI LIFESCIENCES HOLDINGS, INC.
By: /s/ Kurt Oreshack _
Name: Kurt A. Oreshack
Its: EVP & General Counsel
MARAVAI INTERMEDIATE HOLDINGS, LLC
By: /s/ Kur Oreshack _
Name: Kurt A. Oreshack
Its: EVP & General Counsel
EXECUTIVE
/s/ Carl W. Hull _
Carl W. Hull
Amendment No. 1 to
Amended and Restated Employment Agreement
MARAVAI LIFESCIENCES HOLDINGS, INC.
2020 OMNIBUS INCENTIVE PLAN
STOCK OPTION GRANT NOTICE
Pursuant to the terms and conditions of the Maravai LifeSciences Holdings, Inc. 2020 Omnibus Incentive Plan, as amended from time to time (the “Plan”), Maravai LifeSciences Holdings, Inc., a Delaware corporation (the “Company”), hereby grants to the individual listed below (“Participant”) the stock option (the “Option”) set forth below. This award of the Option (this “Award”) is subject to the terms and conditions set forth herein and in the Stock Option Agreement attached hereto as Exhibit A (the “Agreement”), which is incorporated herein by reference. Capitalized terms used but not defined herein shall have the meanings set forth in the Plan.
Participant:
Grant Date: ______________________________
Exercise Price per Share: $____________ per share
Shares Subject to the Option: _____________________ shares of Class A Common Stock of the Company
Type of Option: Non-Qualified Stock Option
Vesting Start Date: _____________________________
Vesting Schedule: Subject to the Agreement including Section 3.3 thereof, the Plan and other terms and conditions set forth herein, the Option will vest and become exercisable with respect to [●], so long as Participant has not incurred a Termination of Service prior to the applicable vesting date.
Final Expiration Date: ______________________________
[Signature Page Follows]
By Participant’s signature below, Participant agrees to be bound by the terms of this Grant Notice, the Plan and the Agreement. Participant has reviewed the Plan, this Grant Notice and the Agreement in their entirety, has had an opportunity to obtain the advice of counsel prior to executing this Grant Notice and fully understands all provisions of the Plan, this Grant Notice and the Agreement. Participant hereby agrees to accept as binding, conclusive and final all decisions or interpretations of the Administrator upon any questions arising under the Plan, this Grant Notice or the Agreement. This Grant Notice may be executed in one or more counterparts (including portable document format (.pdf) and facsimile counterparts), each of which shall be deemed to be an original, but all of which together shall constitute one and the same agreement.
Notwithstanding any provision of this Grant Notice or the Agreement, if Participant has not executed this Grant Notice within 90 days following the Grant Date set forth above, Participant will be deemed to have accepted this Award, subject to all of the terms and conditions of this Grant Notice, the Agreement and the Plan.
MARAVAI LIFESCIENCES PARTICIPANT
HOLDINGS, INC.
By:
Name:
Title:
Exhibit A
STOCK OPTION AGREEMENT
Capitalized terms not specifically defined in this Agreement have the meanings specified in the Grant Notice or, if not defined in the Grant Notice, in the Plan.
Article I.
GENERAL
1.1Grant of Option. The Company has granted to Participant the Option effective as of the grant date set forth in the Grant Notice (the “Grant Date”).
1.2Incorporation of Terms of Plan. The Option is subject to the terms and conditions set forth in this Agreement and the Plan, which is incorporated herein by reference. In the event of any inconsistency between the Plan and this Agreement, the terms of the Plan will control.
Article II.
PERIOD OF EXERCISABILITY
1.1Commencement of Exercisability. The Option will vest and become exercisable according to the vesting schedule in the Grant Notice (the “Vesting Schedule”). Notwithstanding anything in the Grant Notice, the Plan or this Agreement to the contrary, unless the Administrator otherwise determines, the Option will immediately expire and be forfeited as to any portion that is not vested and exercisable as of Participant’s Termination of Service for any reason except as provided in Section 3.3.
1.2Duration of Exercisability. The Vesting Schedule is cumulative. Any portion of the Option which vests and becomes exercisable will remain vested and exercisable until the Option expires. The Option will be forfeited immediately upon its expiration.
1.3Expiration of Option. The Option may not be exercised to any extent by anyone after, and will expire on, the first of the following to occur:
(a)The final expiration date in the Grant Notice;
(b)Except as the Administrator may otherwise approve, the expiration of ninety (90) days from the date of Participant’s Termination of Service, unless Participant’s Termination of Service is for Cause or by reason of Participant’s death or Disability;
(c)Except as the Administrator may otherwise approve, the expiration of one (1) year from the date of Participant’s Termination of Service by reason of Participant’s death or Disability; and
(d)Except as the Administrator may otherwise approve, Participant’s Termination of Service for Cause.
Article III.
EXERCISE OF OPTION
1.1Person Eligible to Exercise. During Participant’s lifetime, only Participant may exercise the Option. After Participant’s death, any exercisable portion of the Option may, prior to the time the Option expires, be exercised by Participant’s Designated Beneficiary as provided in the Plan.
1.2Partial Exercise. Any exercisable portion of the Option or the entire Option, if then wholly exercisable, may be exercised, in whole or in part, according to the procedures in the Plan at any time prior to the time the Option or portion thereof expires, except that the Option may only be exercised for whole Shares.
1.3Change in Control.
(a)Notwithstanding anything in the Grant Notice, this Agreement or the Plan to the contrary, all of the Shares subject to the Option will vest and become exercisable as provided in this Agreement and the Plan if Participant’s Termination of Service is by involuntary termination without Cause or by voluntary resignation for Good Reason, in either case within one year following a Change in Control.
(b)As used herein, “Good Reason” means (a) in the case where there is no employment agreement, offer letter, consulting agreement, change in control agreement, or similar agreement in effect between the Company or an Affiliate and Participant at the time of the grant of the Award (or where there is such agreement in effect but it does not define “good reason” (or words of like import)), (i) any action by the Company or the surviving entity following a Business Combination which results in a material reduction in Participant’s title, status, authority or responsibility, or (ii) a reduction in Participant’s annual base salary or target annual bonus, in each case without the prior written consent of Participant or (b) in the case where there is an employment agreement, offer letter, consulting agreement, change in control agreement, or similar agreement in effect between the Company or an Affiliate and Participant at the time of the grant of the Award that defines “good reason” (or words of like import), “good reason” as defined under such agreement. Notwithstanding anything herein to the contrary, any assertion by Participant of a termination for Good Reason will not be effective unless all of the following conditions are satisfied: (A) Participant must provide written notice to the Board or the board of the surviving entity following a Business Combination of the existence of such condition(s) giving rise to Good Reason within thirty (30) days after the initial occurrence of such condition(s); (B) the condition(s) specified in such notice must remain uncorrected for thirty (30) days following the Board’s or the surviving entity board’s receipt of such written notice; and (C) the date of Participant’s Termination of Service must occur within sixty (60) days after the initial occurrence of the condition(s) specified in such notice. Further and notwithstanding anything herein to the contrary, any Change in Control does not and will not in and of itself constitute Good Reason.
1.4Tax Withholding. To the extent that the receipt, vesting or exercise of this Award results in compensation income or wages to Participant for federal, state, local and/or foreign tax purposes, Participant shall make arrangements satisfactory to the Company for the satisfaction of obligations for the payment of withholding taxes and other tax obligations relating to this Award, which arrangements include the delivery of cash or cash equivalents, Common Stock (including previously owned Common Stock, net exercise, a broker-assisted sale, or other cashless withholding or reduction of the amount of shares otherwise issuable or delivered pursuant to this Award), other property, or any other legal consideration the Committee deems appropriate. If such tax obligations are satisfied through net exercise or the surrender of previously owned Common Stock, the maximum number of shares of Common Stock that may be so withheld (or surrendered) shall be the number of shares of Common Stock that have an aggregate Fair Market Value on the date of withholding or surrender equal to the aggregate amount of such tax liabilities determined based on the greatest withholding rates for federal, state, local and/or foreign tax purposes, including payroll taxes, that may be utilized without creating adverse accounting treatment for the Company with respect to this Award, as determined by the Committee. Any fraction of a share of Common Stock required to satisfy such tax obligations shall be disregarded and the amount due shall be paid instead in cash to Participant. Participant acknowledges that there may be adverse tax consequences upon the receipt, vesting or exercise
of this Award or disposition of the underlying shares and that Participant has been advised, and hereby is advised, to consult a tax advisor. Participant represents that Participant is in no manner relying on the Board, the Committee, the Company or an Affiliate or any of their respective managers, directors, officers, employees or authorized representatives (including attorneys, accountants, consultants, bankers, lenders, prospective lenders and financial representatives) for tax advice or an assessment of such tax consequences.
1.5Automatic Exercise.
(a)Notwithstanding any provision of this Agreement, the Grant Notice or the Plan to the contrary, if at any time after the Grant Date, (i) the Option has become vested and exercisable in whole or in part; (ii) such vested and exercisable portion of the Option is scheduled to expire or terminate for any reason other than due to a termination of Participant’s employment with the Company or any Affiliate for Cause; and (iii) the then-current Fair Market Value of a Share of Common Stock of the Company exceeds the sum of (A) the Exercise Price per Share of the Option and (B) the minimum amount of taxes required to be withheld (if any) if Participant were to exercise the Option at such time, then, unless Participant (or, if applicable, the Participant’s estate or the person who acquires the Option by will or the laws of descent and distribution or otherwise by reason of the death of Participant) provides written notice to the Committee at least ten (10) business days prior to such scheduled expiration or termination date indicating that Participant or such holder desires that the Option expire or terminate without any exercise of the portion of the Option (if any) that has become vested and exercisable, such vested and exercisable portion of the Option shall be automatically exercised as of the date of such scheduled expiration or termination through a Net Issuance Exercise and the minimum amount of taxes required to be withheld (if any) upon such automatic exercise shall be satisfied by withholding shares of Common Stock otherwise issuable in connection with such exercise having a Fair Market Value equal to such amount.
(b)As used herein, “Net Issuance Exercise” means delivering or constructively tendering to the Company Shares of Common Stock having a Fair Market Value equal to the Exercise Price (provided such shares used for this purpose must have been held by Participant for such minimum period of time as may be established from time to time by the Committee to avoid adverse accounting consequences).
Article IV.
OTHER PROVISIONS
1.1Adjustments. Participant acknowledges that the Option is subject to adjustment, modification and termination in certain events as provided in this Agreement and the Plan.
1.2Notices. All notices and other communications under this Agreement shall be in writing and shall be delivered to the parties at the following addresses (or at such other address for a party as shall be specified by like notice):
If to the Company, unless otherwise designated by the Company in a written notice to the Participant (or other holder):
Maravai LifeSciences Holdings, Inc.
Attn: General Counsel
10770 Wateridge Circle Suite 200
San Diego, CA 92121
If to Participant, at Participant’s last known address on file with the Company. Any notice that is delivered personally or by overnight courier or telecopier in the manner provided
herein shall be deemed to have been duly given to Participant when it is mailed by the Company or, if such notice is not mailed to Participant, upon receipt by Participant. Any notice that is addressed and mailed in the manner herein provided shall be conclusively presumed to have been given to the party to whom it is addressed at the close of business, local time of the recipient, on the fourth day after the day it is so placed in the mail.
1.3Titles. Titles are provided herein for convenience only and are not to serve as a basis for interpretation or construction of this Agreement.
1.4Conformity to Securities Laws. Participant acknowledges that the Plan, the Grant Notice and this Agreement are intended to conform to the extent necessary with all Applicable Laws and, to the extent Applicable Laws permit, will be deemed amended as necessary to conform to Applicable Laws.
1.5Successors and Assigns. The Company may assign any of its rights under this Agreement to single or multiple assignees, and this Agreement will inure to the benefit of the successors and assigns of the Company. Subject to the restrictions on transfer set forth in the Plan, this Agreement will be binding upon and inure to the benefit of the heirs, legatees, legal representatives, successors and assigns of the parties hereto.
1.6Limitations Applicable to Section 16 Persons. Notwithstanding any other provision of the Plan or this Agreement, if Participant is subject to Section 16 of the Exchange Act, the Plan, the Grant Notice, this Agreement and the Option will be subject to any additional limitations set forth in any applicable exemptive rule under Section 16 of the Exchange Act (including any amendment to Rule 16b-3) that are requirements for the application of such exemptive rule. To the extent Applicable Laws permit, this Agreement will be deemed amended as necessary to conform to such applicable exemptive rule.
1.7Entire Agreement. The Plan, the Grant Notice and this Agreement (including any exhibit hereto) constitute the entire agreement of the parties and supersede in their entirety all prior undertakings and agreements of the Company and Participant with respect to the subject matter hereof.
1.8Agreement Severable. In the event that any provision of the Grant Notice or this Agreement is held illegal or invalid, the provision will be severable from, and the illegality or invalidity of the provision will not be construed to have any effect on, the remaining provisions of the Grant Notice or this Agreement.
1.9Limitation on Participant’s Rights. Participation in the Plan confers no rights or interests other than as herein provided. This Agreement creates only a contractual obligation on the part of the Company as to amounts payable and may not be construed as creating a trust. Neither the Plan nor any underlying program, in and of itself, has any assets. Participant will have only the rights of a general unsecured creditor of the Company with respect to amounts credited and benefits payable, if any, with respect to the Option, and rights no greater than the right to receive the Shares as a general unsecured creditor with respect to the Option, as and when exercised pursuant to the terms hereof.
1.10No Right to Continued Service or Awards. Nothing in the Plan, the Grant Notice or this Agreement confers upon Participant any right to continue in the service of the Company or any Affiliate or interferes with or restricts in any way the rights of the Company and its Subsidiaries, which rights are hereby expressly reserved, to discharge or terminate the services of Participant at any time for any reason whatsoever, with or without Cause, except to the extent expressly provided otherwise in a written agreement between the Company or an Affiliate and Participant. The grant of the Option is a one-time benefit and does not create any contractual or
other right to receive a grant of Awards or benefits in lieu of Awards in the future. Any future Awards will be granted at the sole discretion of the Company.
1.11Satisfaction of Claims. Any issuance or transfer of Shares or other property to Participant or Participant’s legal representative, heir, legatee or distribute, in accordance with the Plan, the Grant Notice and this Agreement shall be in full satisfaction of all claims of such person hereunder.
1.12Counterparts. The Grant Notice may be executed in one or more counterparts, including by way of any electronic signature, subject to Applicable Law, each of which will be deemed an original and all of which together will constitute one instrument.
1.13Company Recoupment of Awards. Participant’s rights with respect to this Award shall in all events be subject to (a) any right that the Company may have under any Company recoupment policy or other agreement or arrangement with Participant, or (b) any right or obligation that the Company may have regarding the clawback of “incentive-based compensation” under Section 10D of the Exchange Act and any applicable rules and regulations promulgated thereunder from time to time by the U.S. Securities and Exchange Commission.
* * * * *
DocumentMARAVAI LIFESCIENCES HOLDINGS, INC.
2020 OMNIBUS INCENTIVE PLAN
RESTRICTED STOCK UNIT GRANT NOTICE
Pursuant to the terms and conditions of the Maravai LifeSciences Holdings, Inc. 2020 Omnibus Incentive Plan, as amended from time to time (the “Plan”), Maravai LifeSciences Holdings, Inc., a Delaware corporation (the “Company”), hereby grants to the individual listed below (“Participant”) the number of restricted stock units (the “RSUs”) set forth below. This award of RSUs (this “Award”) is subject to the terms and conditions set forth herein and in the Restricted Stock Unit Agreement attached hereto as Exhibit A (the “Agreement”), which is incorporated herein by reference. Capitalized terms used but not defined herein shall have the meanings set forth in the Plan.
Participant:
Grant Date:
Number of RSUs:
Vesting Commencement Date:
Vesting Schedule: Subject to the Agreement, the Plan and other terms and conditions set forth herein, [●] of the RSUs will vest on the [●] and [●] anniversaries of the Vesting Commencement Date set forth above, so long as Participant has not incurred a Termination of Service prior to the applicable vesting date.
[Signature Page Follows]
By Participant’s signature below, Participant agrees to be bound by the terms of this Grant Notice, the Plan and the Agreement. Participant has reviewed the Plan, this Grant Notice and the Agreement in their entirety, has had an opportunity to obtain the advice of counsel prior to executing this Grant Notice and fully understands all provisions of the Plan, this Grant Notice and the Agreement. Participant hereby agrees to accept as binding, conclusive and final all decisions or interpretations of the Administrator upon any questions arising under the Plan, this Grant Notice or the Agreement. This Grant Notice may be executed in one or more counterparts (including portable document format (.pdf) and facsimile counterparts), each of which shall be deemed to be an original, but all of which together shall constitute one and the same agreement.
Notwithstanding any provision of this Grant Notice or the Agreement, if Participant has not executed this Grant Notice within 90 days following the Grant Date set forth above, Participant will be deemed to have accepted this Award, subject to all of the terms and conditions of this Grant Notice, the Agreement and the Plan.
MARAVAI LIFESCIENCES PARTICIPANT
HOLDINGS, INC.
By:
Name:
Title:
Exhibit A
RESTRICTED STOCK UNIT AGREEMENT
Capitalized terms not specifically defined in this Agreement have the meanings specified in the Grant Notice or, if not defined in the Grant Notice, in the Plan.
Article I.
GENERAL
1.1Award of RSUs. The Company has granted the RSUs to Participant effective as of the grant date set forth in the Grant Notice (the “Grant Date”). Each RSU represents the right to receive one Share as set forth in this Agreement. Participant will have no right to the distribution of any Shares or payment of any cash until the time (if ever) the RSUs have vested.
1.2Incorporation of Terms of Plan. The RSUs are subject to the terms and conditions set forth in this Agreement and the Plan, which is incorporated herein by reference. In the event of any inconsistency between the Plan and this Agreement, the terms of the Plan will control.
1.3Unsecured Promise. The RSUs will at all times prior to settlement represent an unsecured Company obligation payable only from the Company’s general assets.
Article II.
VESTING; FORFEITURE AND SETTLEMENT
1.1Vesting; Forfeiture.
(a)The RSUs will vest according to the vesting schedule in the Grant Notice. In the event of Participant’s Termination of Service for any reason, all unvested RSUs will immediately and automatically be cancelled and forfeited, except as otherwise determined by the Administrator or provided in a binding written agreement between Participant and the Company.
(b)Notwithstanding anything in the Grant Notice, this Agreement or the Plan to the contrary, the RSUs will vest in their entirety if Participant’s Termination of Service is by involuntary termination without Cause or by voluntary resignation for Good Reason, in either case within two years following a Change in Control.
(c)As used herein, “Good Reason” means (a) in the case where there is no employment agreement, offer letter, consulting agreement, change in control agreement, or similar agreement in effect between the Company or an Affiliate and Participant at the time of the grant of the Award (or where there is such agreement in effect but it does not define “good reason” (or words of like import)), (i) any action by the Company or the surviving entity following a Business Combination which results in a material reduction in Participant’s title, status, authority, duties or responsibilities, (ii) a reduction in Participant’s annual base salary, target annual bonus or the target grant date value of Participant’s annual equity award (in relation to the target for Participant’s most recent annual equity award prior to the Change in Control or, if no such award, the target for such award for the Company executive most similarly situated to Participant), or (iii) the relocation of Participant’s principal office or place of work to a location that would cause an increase by more than thirty-five (35) miles in Participant’s one-way commuting distance from Participant’s principal personal residence at the time of the Change in Control, in each case without the prior written consent of Participant or (b) in the case where there is an employment agreement, offer letter, consulting agreement, change in control agreement, or similar agreement in effect between the Company or an Affiliate and Participant at the time of the Change in Control that defines “good reason” (or words of like import), “good
reason” as defined under such agreement. Notwithstanding anything herein to the contrary, any assertion by Participant of a termination for Good Reason will not be effective unless all of the following conditions are satisfied: (A) Participant must provide written notice to the Board or the board of the surviving entity following a Business Combination of the existence of such condition(s) giving rise to Good Reason within thirty (30) days after the initial occurrence of such condition(s); (B) the condition(s) specified in such notice must remain uncorrected for thirty (30) days following the Board’s or the surviving entity board’s receipt of such written notice; and (C) the date of Participant’s Termination of Service must occur within sixty (60) days after the initial occurrence of the condition(s) specified in such notice. Further and notwithstanding anything herein to the contrary, any Change in Control does not and will not in and of itself constitute Good Reason.
1.2Settlement. As soon as administratively practicable following the vesting of RSUs pursuant to Section 2.1, but in no event later than 30 days after such vesting date, the Company shall deliver to Participant a number of Shares equal to the number of RSUs subject to this Award. All Shares issued hereunder shall be delivered either by delivering one or more certificates for such shares to Participant or by entering such shares in book-entry form, as determined by the Committee in its sole discretion. The value of Shares shall not bear any interest owing to the passage of time.
Article III.
TAXATION AND TAX WITHHOLDING
1.1Representation. Participant represents to the Company that Participant has reviewed with Participant’s own tax advisors the tax consequences of this Award and the transactions contemplated by the Grant Notice and this Agreement. Participant is relying solely on such advisors and not on any statements or representations of the Company or any of its agents.
1.2Tax Withholding. To the extent that the receipt, vesting or settlement of this Award results in compensation income or wages to the Participant for federal, state, local and/or foreign tax purposes, Participant shall make arrangements satisfactory to the Company regarding the payment of, any income tax, social insurance contribution or other applicable taxes that are required to be withheld in respect of this Award, which arrangements include the delivery of cash or cash equivalents, Shares (including previously owned Shares (which is not subject to any pledge or other security interest), net settlement, a broker-assisted sale, or other cashless withholding or reduction of the amount of shares otherwise issuable or delivered pursuant to this Award), other property, or any other legal consideration the Committee deems appropriate. If such tax obligations are satisfied through net settlement or the surrender of previously owned Shares, the maximum number of Shares that may be so withheld (or surrendered) shall be the number of Shares that have an aggregate Fair Market Value on the date of withholding or surrender equal to the aggregate amount of such tax liabilities determined based on the greatest withholding rates for federal, state, local and/or foreign tax purposes, including payroll taxes, that may be utilized without creating adverse accounting treatment for the Company with respect to this Award, as determined by the Committee. Any fraction of a Share required to satisfy such tax obligations shall be disregarded and the amount due shall be paid instead in cash to Participant. Participant acknowledges that there may be adverse tax consequences upon the receipt, vesting or settlement of this Award or disposition of the underlying shares and that Participant has been advised, and hereby is advised, to consult a tax advisor. Participant represents that Participant is in no manner relying on the Board, the Committee, the Company or an Affiliate or any of their respective managers, directors, officers, employees or authorized representatives (including attorneys, accountants, consultants, bankers, lenders, prospective lenders and financial representatives) for tax advice or an assessment of such tax consequences.
Article IV.
OTHER PROVISIONS
1.1Adjustments. Participant acknowledges that the RSUs and the Shares subject to the RSUs are subject to adjustment, modification and termination in certain events as provided in this Agreement and the Plan.
1.2Notices. All notices and other communications under this Agreement shall be in writing and shall be delivered to the parties at the following addresses (or at such other address for a party as shall be specified by like notice):
If to the Company, unless otherwise designated by the Company in a written notice to Participant (or other holder):
Maravai LifeSciences Holdings, Inc.
Attn: General Counsel
10770 Wateridge Circle Suite 200
San Diego, CA 92121
If to Participant, at Participant’s last known address on file with the Company. Any notice that is delivered personally or by overnight courier or telecopier in the manner provided herein shall be deemed to have been duly given to Participant when it is mailed by the Company or, if such notice is not mailed to Participant, upon receipt by Participant. Any notice that is addressed and mailed in the manner herein provided shall be conclusively presumed to have been given to the party to whom it is addressed at the close of business, local time of the recipient, on the fourth day after the day it is so placed in the mail.
1.3Titles. Titles are provided herein for convenience only and are not to serve as a basis for interpretation or construction of this Agreement.
1.4Conformity to Securities Laws. Participant acknowledges that the Plan, the Grant Notice and this Agreement are intended to conform to the extent necessary with all Applicable Laws and, to the extent Applicable Laws permit, will be deemed amended as necessary to conform to Applicable Laws.
1.5Successors and Assigns. The Company may assign any of its rights under this Agreement to single or multiple assignees, and this Agreement will inure to the benefit of the successors and assigns of the Company. Subject to the restrictions on transfer set forth in the Plan, this Agreement will be binding upon and inure to the benefit of the heirs, legatees, legal representatives, successors and assigns of the parties hereto.
1.6Limitations Applicable to Section 16 Persons. Notwithstanding any other provision of the Plan or this Agreement, if Participant is subject to Section 16 of the Exchange Act, the Plan, the Grant Notice, this Agreement, the RSUs will be subject to any additional limitations set forth in any applicable exemptive rule under Section 16 of the Exchange Act (including any amendment to Rule 16b-3) that are requirements for the application of such exemptive rule. To the extent Applicable Laws permit, this Agreement will be deemed amended as necessary to conform to such applicable exemptive rule.
1.7Entire Agreement. The Plan, the Grant Notice and this Agreement (including any exhibit hereto) constitute the entire agreement of the parties and supersede in their entirety all prior undertakings and agreements of the Company and Participant with respect to the subject matter hereof.
1.8Agreement Severable. In the event that any provision of the Grant Notice or this Agreement is held illegal or invalid, the provision will be severable from, and the illegality or invalidity of the provision will not be construed to have any effect on, the remaining provisions of the Grant Notice or this Agreement.
1.9Limitation on Participant’s Rights. Participation in the Plan confers no rights or interests other than as herein provided. This Agreement creates only a contractual obligation on the part of the Company as to amounts payable and may not be construed as creating a trust. Neither the Plan nor any underlying program, in and of itself, has any assets. Participant will have only the rights of a general unsecured creditor of the Company with respect to amounts credited and benefits payable, if any, with respect to the RSUs, and rights no greater than the right to receive cash or the Shares as a general unsecured creditor with respect to the RSUs, as and when settled pursuant to the terms of this Agreement.
1.10Non-Transferability. During the lifetime of Participant, the RSUs may not be sold, pledged, assigned or transferred in any manner other than by will or the laws of descent and distribution, unless and until the Shares underlying the RSUs have been issued, and all restrictions applicable to such Shares have lapsed. Neither the RSUs not any interest or right therein shall be liable for the debts, contracts or engagements of Participant or his or her successors in interest or shall be subject to disposition by transfer, alienation, anticipation, pledge, encumbrance, assignment or any other means whether such disposition be voluntary or involuntary or by operation of law by judgment, levy, attachment, garnishment or any other legal or equitable proceedings (including bankruptcy), and any attempted disposition thereof shall be null and void and of no effect, except to the extent that such disposition is permitted by the preceding sentence.
1.11Legends. If a stock certificate is issued with respect to the Shares delivered hereunder, such certificate shall bear such legend or legends as the Committee deems appropriate in order to reflect the restrictions set forth in this Agreement and to ensure compliance with the terms and provisions of this Agreement, the rules, regulations and other requirements of the Securities and Exchange Commission and any other Applicable Laws. If the Shares issued hereunder are held in book-entry form, then such entry will reflect that the Shares are subject to the restrictions set forth in this Agreement.
1.12No Right to Continued Service or Awards. Nothing in the Plan, the Grant Notice or this Agreement confers upon Participant any right to continue in the service of the Company or any Affiliate or interferes with or restricts in any way the rights of the Company and its Subsidiaries, which rights are hereby expressly reserved, to discharge or terminate the services of Participant at any time for any reason whatsoever, with or without Cause, except to the extent expressly provided otherwise in a written agreement between the Company or an Affiliate and Participant. The grant of the RSUs is a one-time benefit and does not create any contractual or other right to receive a grant of Awards or benefits in lieu of Awards in the future. Any future Awards will be granted at the sole discretion of the Company.
1.13Satisfaction of Claims. Any issuance or transfer of Shares or other property to Participant or Participant’s legal representative, heir, legatee or distribute, in accordance with the Plan, the Grant Notice and this Agreement shall be in full satisfaction of all claims of such person hereunder.
1.14Counterparts. The Grant Notice may be executed in one or more counterparts, including by way of any electronic signature, subject to Applicable Law, each of which will be deemed an original and all of which together will constitute one instrument.
1.15Company Recoupment of Awards. Participant’s rights with respect to this Award shall in all events be subject to (a) any right that the Company may have under any Company recoupment policy or other agreement or arrangement with Participant, or (b) any right or obligation that the Company may have regarding the clawback of “incentive-based compensation” under Section 10D of the Exchange Act and any applicable rules and regulations promulgated thereunder from time to time by the U.S. Securities and Exchange Commission.
* * * * *
DocumentMARAVAI LIFESCIENCES HOLDINGS, INC.
2020 OMNIBUS INCENTIVE PLAN
PERFORMANCE STOCK UNIT GRANT NOTICE
Pursuant to the terms and conditions of the Maravai LifeSciences Holdings, Inc. 2020 Omnibus Incentive Plan, as amended from time to time (the “Plan”), Maravai LifeSciences Holdings, Inc., a Delaware corporation (the “Company”), hereby grants to the individual listed below (“Participant”) the number of performance-based Restricted Stock Units (the “PSUs”) set forth below. This award of PSUs (this “Award”) is subject to the terms and conditions set forth herein and in the Performance Stock Unit Agreement attached hereto as Exhibit A (the “Agreement”), which is incorporated herein by reference. Capitalized terms used but not defined herein shall have the meanings set forth in the Plan.
Participant:
Grant Date:
Number of PSUs:
| | | | | |
| Vesting of PSUs: | Subject to the Agreement, the Plan and other terms and conditions set forth herein, the PSUs will vest, if at all, on the Certification Date (as defined in Exhibit B) based on achievement of the performance criteria set forth in Exhibit B, so long as Participant has not incurred a Termination of Service prior to the settlement date. |
By Participant’s signature below, Participant agrees to be bound by the terms of this Grant Notice, the Plan and the Agreement. Participant has reviewed the Plan, this Grant Notice and the Agreement in their entirety, has had an opportunity to obtain the advice of counsel prior to executing this Grant Notice and fully understands all provisions of the Plan, this Grant Notice and the Agreement. Participant hereby agrees to accept as binding, conclusive and final all decisions or interpretations of the Committee upon any questions arising under the Plan, this Grant Notice or the Agreement. This Grant Notice may be executed in one or more counterparts (including portable document format (.pdf) and facsimile counterparts), each of which shall be deemed to be an original, but all of which together shall constitute one and the same agreement.
Notwithstanding any provision of this Grant Notice or the Agreement, if Participant has not executed this Grant Notice within 90 days following the Grant Date set forth above, Participant will be deemed to have accepted this Award, subject to all of the terms and conditions of this Grant Notice, the Agreement and the Plan.
[Signature Page Follows]
MARAVAI LIFESCIENCES PARTICIPANT
HOLDINGS, INC.
By:
Name:
Title:
Exhibit A
PERFORMANCE STOCK UNIT AGREEMENT
Capitalized terms not specifically defined in this Agreement have the meanings specified in the Grant Notice or, if not defined in the Grant Notice, in the Plan.
Article I.
GENERAL
1.1Award of PSUs.
(a)The Company has granted the PSUs to Participant effective as of the grant date set forth in the Grant Notice (the “Grant Date”). Each PSU represents the right to receive one Share as set forth in this Agreement. Participant will have no right to the distribution of any Shares or payment of any cash until the time (if ever) the PSUs have vested.
1.2Incorporation of Terms of Plan. The PSUs are subject to the terms and conditions set forth in this Agreement, the Grant Notice and the Plan, which are incorporated herein by reference. In the event of any inconsistency between the Plan and this Agreement, the terms of the Plan will control.
1.3Unsecured Promise. The PSUs will at all times prior to settlement represent an unsecured Company obligation payable only from the Company’s general assets.
Article II.
VESTING; FORFEITURE AND SETTLEMENT
1.1Vesting; Forfeiture.
(a)The PSUs will vest according to the terms set forth in the Grant Notice. In the event of Participant’s Termination of Service for any reason, all unvested PSUs will immediately and automatically be cancelled and forfeited, except as otherwise determined by the Committee or provided in a binding written agreement between Participant and the Company.
(b)Notwithstanding anything in the Grant Notice, this Agreement or the Plan to the contrary, in the event a Change in Control occurs prior to the third anniversary of the Grant Date, the PSUs will convert automatically to time-based Restricted Stock Units that will vest in their entirety upon the third anniversary of the Grant Date (the “Converted RSUs”), so long as Participant continuously provides services to the Company or any Affiliate through the third anniversary of the Grant Date; provided, that if, within the 24-month period following the consummation of such Change in Control, Participant’s Termination of Service is effected (x) by the Company without Cause (as defined in the Participant’s employment agreement with the Company (as may be amended and/or restated from time to time, the “Employment Agreement”)) or (y) by Participant for Good Reason (as defined in the Employment Agreement), any then-outstanding Converted RSUs shall immediately become vested as of the date of such Termination of Service.
1.2Settlement. As soon as administratively practicable following the vesting of PSUs pursuant to Section 2.1, but in no event later than 30 days after the Certification Date, the Company shall deliver to Participant a number of Shares equal to the number of PSUs that vested. All Shares issued hereunder shall be delivered either by delivering one or more certificates for such shares to Participant or by entering such shares in book-entry form, as
determined by the Committee in its sole discretion. The value of Shares shall not bear any interest owing to the passage of time.
Article III.
TAXATION AND TAX WITHHOLDING
1.1Representation. Participant represents to the Company that Participant has reviewed with Participant’s own tax advisors the tax consequences of this Award and the transactions contemplated by the Grant Notice and this Agreement. Participant is relying solely on such advisors and not on any statements or representations of the Company or any of its agents.
1.2Tax Withholding. To the extent that the receipt, vesting or settlement of this Award results in compensation income or wages to the Participant for federal, state, local and/or foreign tax purposes, Participant shall make arrangements satisfactory to the Company regarding the payment of, any income tax, social insurance contribution or other applicable taxes that are required to be withheld in respect of this Award, which arrangements include the delivery of cash or cash equivalents, Shares (including previously owned Shares (which is not subject to any pledge or other security interest), net settlement, a broker-assisted sale, or other cashless withholding or reduction of the amount of Shares otherwise issuable or delivered pursuant to this Award), other property, or any other legal consideration the Committee deems appropriate. If such tax obligations are satisfied through net settlement or the surrender of previously owned Shares, the maximum number of Shares that may be so withheld (or surrendered) shall be the number of Shares that have an aggregate Fair Market Value on the date of withholding or surrender equal to the aggregate amount of such tax liabilities determined based on the greatest withholding rates for federal, state, local and/or foreign tax purposes, including payroll taxes, that may be utilized without creating adverse accounting treatment for the Company with respect to this Award, as determined by the Committee. Any fraction of a Share required to satisfy such tax obligations shall be disregarded and the amount due shall be paid instead in cash to Participant. Participant acknowledges that there may be adverse tax consequences upon the receipt, vesting or settlement of this Award or disposition of the underlying shares and that Participant has been advised, and hereby is advised, to consult a tax advisor. Participant represents that Participant is in no manner relying on the Board, the Committee, the Company or an Affiliate or any of their respective managers, directors, officers, employees or authorized representatives (including attorneys, accountants, consultants, bankers, lenders, prospective lenders and financial representatives) for tax advice or an assessment of such tax consequences.
Article IV.
OTHER PROVISIONS
1.1Adjustments. Participant acknowledges that the PSUs and the Shares subject to the PSUs are subject to adjustment, modification and termination in certain events as provided in this Agreement and the Plan.
1.2Notices. All notices and other communications under this Agreement shall be in writing and shall be delivered to the parties at the following addresses (or at such other address for a party as shall be specified by like notice):
If to the Company, unless otherwise designated by the Company in a written notice to Participant (or other holder):
Maravai LifeSciences Holdings, Inc.
Attn: General Counsel
10770 Wateridge Circle Suite 200
San Diego, CA 92121
If to Participant, at Participant’s last known address on file with the Company. Any notice that is delivered personally or by overnight courier or telecopier in the manner provided herein shall be deemed to have been duly given to Participant when it is mailed by the Company or, if such notice is not mailed to Participant, upon receipt by Participant. Any notice that is addressed and mailed in the manner herein provided shall be conclusively presumed to have been given to the party to whom it is addressed at the close of business, local time of the recipient, on the fourth day after the day it is so placed in the mail.
1.3Titles. Titles are provided herein for convenience only and are not to serve as a basis for interpretation or construction of this Agreement.
1.4Conformity to Securities Laws. Participant acknowledges that the Plan, the Grant Notice and this Agreement are intended to conform to the extent necessary with all Applicable Laws and, to the extent Applicable Laws permit, will be deemed amended as necessary to conform to Applicable Laws.
1.5Successors and Assigns. The Company may assign any of its rights under this Agreement to single or multiple assignees, and this Agreement will inure to the benefit of the successors and assigns of the Company. Subject to the restrictions on transfer set forth in the Plan, this Agreement will be binding upon and inure to the benefit of the heirs, legatees, legal representatives, successors and assigns of the parties hereto.
1.6Limitations Applicable to Section 16 Persons. Notwithstanding any other provision of the Plan or this Agreement, if Participant is subject to Section 16 of the Exchange Act, the Plan, the Grant Notice, this Agreement, and the PSUs will be subject to any additional limitations set forth in any applicable exemptive rule under Section 16 of the Exchange Act (including any amendment to Rule 16b-3) that are requirements for the application of such exemptive rule. To the extent Applicable Laws permit, this Agreement will be deemed amended as necessary to conform to such applicable exemptive rule.
1.7Entire Agreement. The Plan, the Grant Notice and this Agreement (including any exhibit hereto) constitute the entire agreement of the parties and supersede in their entirety all prior undertakings and agreements of the Company and Participant with respect to the subject matter hereof.
1.8Agreement Severable. In the event that any provision of the Grant Notice or this Agreement is held illegal or invalid, the provision will be severable from, and the illegality or invalidity of the provision will not be construed to have any effect on, the remaining provisions of the Grant Notice or this Agreement.
1.9Limitation on Participant’s Rights. Participation in the Plan confers no rights or interests other than as herein provided. This Agreement creates only a contractual obligation on the part of the Company as to amounts payable and may not be construed as creating a trust. Neither the Plan nor any underlying program, in and of itself, has any assets. Participant will have only the rights of a general unsecured creditor of the Company with respect to amounts credited and benefits payable, if any, with respect to the PSUs, and rights no greater than the right to receive cash or the Shares as a general unsecured creditor with respect to the PSUs, as and when settled pursuant to the terms of this Agreement.
1.10Non-Transferability. During the lifetime of Participant, the PSUs may not be sold, pledged, assigned or transferred in any manner other than by will or the laws of descent and distribution, unless and until the Shares underlying the PSUs have been issued, and all
restrictions applicable to such Shares have lapsed. Neither the PSUs not any interest or right therein shall be liable for the debts, contracts or engagements of Participant or Participant’s successors in interest or shall be subject to disposition by transfer, alienation, anticipation, pledge, encumbrance, assignment or any other means whether such disposition be voluntary or involuntary or by operation of law by judgment, levy, attachment, garnishment or any other legal or equitable proceedings (including bankruptcy), and any attempted disposition thereof shall be null and void and of no effect, except to the extent that such disposition is permitted by the preceding sentence.
1.11Legends. If a stock certificate is issued with respect to the Shares delivered hereunder, such certificate shall bear such legend or legends as the Committee deems appropriate in order to reflect the restrictions set forth in this Agreement and to ensure compliance with the terms and provisions of this Agreement, the rules, regulations and other requirements of the Securities and Exchange Commission and any other Applicable Laws. If the Shares issued hereunder are held in book-entry form, then such entry will reflect that the Shares are subject to the restrictions set forth in this Agreement.
1.12No Right to Continued Service or Awards. Nothing in the Plan, the Grant Notice or this Agreement confers upon Participant any right to continue in the service of the Company or any Affiliate or interferes with or restricts in any way the rights of the Company and its Subsidiaries, which rights are hereby expressly reserved, to discharge or terminate the services of Participant at any time for any reason whatsoever, with or without Cause, except to the extent expressly provided otherwise in a written agreement between the Company or an Affiliate and Participant. The grant of the PSUs is a one-time benefit and does not create any contractual or other right to receive a grant of Awards or benefits in lieu of Awards in the future. Any future Awards will be granted at the sole discretion of the Company.
1.13Satisfaction of Claims. Any issuance or transfer of Shares or other property to Participant or Participant’s legal representative, heir, legatee or distribute, in accordance with the Plan, the Grant Notice and this Agreement shall be in full satisfaction of all claims of such person hereunder.
1.14Counterparts. The Grant Notice may be executed in one or more counterparts, including by way of any electronic signature, subject to Applicable Law, each of which will be deemed an original and all of which together will constitute one instrument.
1.15Company Recoupment of Awards. Participant’s rights with respect to this Award shall in all events be subject to (a) any right that the Company may have under any Company recoupment policy or other agreement or arrangement with Participant, or (b) any right or obligation that the Company may have regarding the clawback of “incentive-based compensation” under Section 10D of the Exchange Act and any applicable rules and regulations promulgated thereunder from time to time by the U.S. Securities and Exchange Commission.
* * * * *
Exhibit B
Performance Criteria
This Exhibit B sets forth the performance criteria applicable to the PSUs. Subject to the terms and conditions set forth in the Grant Notice, the Agreement and the Plan, the [portion]/[number] of PSUs subject to this Award that become vested during the Performance Period, if any, will be determined upon the Committee’s certification of the achievement of the performance criteria in accordance with this Exhibit B, which shall occur within 60 days following the end of the Performance Period (the “Certification Date”). Capitalized terms used but not defined herein shall have the same meanings as are ascribed thereto in the Grant Notice, the Agreement or the Plan, as applicable.
The PSUs shall vest, if at all, based on the 60-Day VWAP in accordance with the schedule below:
| | | | | |
| 60-Day VWAP as of – [Vesting Date] | [Percentage]/[Number] of PSUs Granted Hereunder that Become Vested |
Less than $[●] | [●] |
Greater than or equal to $[●], but less than $[●] | [●] |
[Greater than or equal to $[●], but less than $[●]] | [●] |
$[●] or greater | [●] |
For the avoidance of doubt, linear interpolation shall not apply in the event the 60-Day VWAP is between $[●] and $[●].
As used herein, the “60-Day VWAP” means the volume-weighted average price of a Share for the 60 consecutive trading days immediately preceding [Vesting Date] (such period, the “Performance Period”), as adjusted for stock splits, stock dividends and other similar events.
All PSUs subject to this Award that are outstanding as of the date immediately following the Certification Date shall be forfeited and cancelled for no consideration if they do not become vested as set forth above.
Consistent with the terms of the Plan, all designations, determinations, interpretations, and other decisions under or with respect to the terms of the Plan or the Agreement, including this Exhibit B, shall be within the sole discretion of the Committee, and shall be final, conclusive, and binding upon all persons.
DocumentExhibit 21.1
Subsidiaries of the Registrant
Subsidiaries of Maravai LifeSciences Holdings, Inc.
Maravai Topco Holdings, LLC (Delaware)
Subsidiaries of Maravai Topco Holdings, LLC
Maravai Intermediate Holdings, LLC (Delaware)
Subsidiaries of Maravai Intermediate Holdings, LLC
Alphazyme, LLC (Delaware)
Cygnus Technologies, LLC (Delaware)
Maravai LifeSciences International Holdings, Inc. (Delaware)
MLSA Holdings, LLC (Delaware)
MockV Solutions, LLC (Maryland)
TriLink Biotechnologies, LLC (Delaware)
Subsidiaries of TriLink BioTechnologies, LLC (Delaware)
Glen Research, LLC (Delaware)
MyChem, LLC (California)
Subsidiaries of Maravai LifeSciences International Holdings, Inc.
Maravai LifeSciences Deutschland GmbH (Germany)
Document
Exhibit 23.1
Consent of Independent Registered Public Accounting Firm
We consent to the incorporation by reference in the Registration Statement (Form S-8 No. 333-250895) pertaining to the Maravai LifeSciences Holdings, Inc. 2020 Omnibus Incentive Plan and Maravai LifeSciences Holdings, Inc. 2020 Employee Stock Purchase Plan of Maravai LifeSciences Holdings, Inc. of our reports dated February 29, 2024, with respect to the consolidated financial statements of Maravai LifeSciences Holdings, Inc. and the effectiveness of internal control over financial reporting of Maravai LifeSciences Holdings, Inc. included in this Annual Report (Form 10-K) of Maravai LifeSciences Holdings, Inc. for the year ended December 31, 2023.
/s/ Ernst & Young LLP
San Mateo, California
February 29, 2024
DocumentEXHIBIT 31.1
Certification Pursuant to Section 302 of Sarbanes-Oxley Act of 2002
I, William E. Martin, III, certify that:
1. I have reviewed this Annual Report on Form 10-K of Maravai LifeSciences Holdings, Inc.;
2. Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report;
3. Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this report;
4. The registrant’s other certifying officer and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the registrant and have:
a. Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this report is being prepared;
b. Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles;
c. Evaluated the effectiveness of the registrant’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and
d. Disclosed in this report any change in the registrant’s internal control over financial reporting that occurred during the registrant’s most recent fiscal quarter (the registrant’s fourth fiscal quarter in the case of an annual report) that has materially affected, or is reasonably likely to materially affect, the registrant’s internal control over financial reporting; and
5. The registrant’s other certifying officer and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the registrant’s auditors and the audit committee of the registrant’s board of directors (or persons performing the equivalent functions):
a. All significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the registrant’s ability to record, process, summarize and report financial information; and
b. Any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant’s internal control over financial reporting.
Date: February 29, 2024
| | | | | | | | |
| | /s/ William E. Martin, III |
| | William E. Martin, III |
| | Chief Executive Officer |
EXHIBIT 31.2
Certification Pursuant to Section 302 of Sarbanes-Oxley Act of 2002
I, Kevin Herde, certify that:
1. I have reviewed this Annual Report on Form 10-K of Maravai LifeSciences Holdings, Inc.;
2. Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report;
3. Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this report;
4. The registrant’s other certifying officer and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the registrant and have:
a. Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this report is being prepared;
b. Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles;
c. Evaluated the effectiveness of the registrant’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and
d. Disclosed in this report any change in the registrant’s internal control over financial reporting that occurred during the registrant’s most recent fiscal quarter (the registrant’s fourth fiscal quarter in the case of an annual report) that has materially affected, or is reasonably likely to materially affect, the registrant’s internal control over financial reporting; and
5. The registrant’s other certifying officer and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the registrant’s auditors and the audit committee of the registrant’s board of directors (or persons performing the equivalent functions):
a. All significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the registrant’s ability to record, process, summarize and report financial information; and
b. Any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant’s internal control over financial reporting.
Date: February 29, 2024
| | | | | | | | |
| | /s/ Kevin Herde |
| | Kevin Herde |
| | Chief Financial Officer |
EXHIBIT 32.1
Certification of the Chief Executive Officer
Pursuant to Rule 18 U.S.C. Section 1350
In connection with the Annual Report on Form 10-K of Maravai LifeSciences Holdings, Inc. (the “Company”) for the year ended December 31, 2023, as filed with the U.S. Securities and Exchange Commission (the “Report”), I, William E. Martin, III, Chief Executive Officer of the Company, hereby certify, pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002, that, to my knowledge:
1. The Report fully complies with the requirements of Section 13(a) or 15(d) of the Securities Exchange Act of 1934, as amended; and
2. The information contained in the Report fairly presents, in all material respects, the financial condition and results of operations of the Company.
Date: February 29, 2024
| | | | | | | | |
| | /s/ William E. Martin, III |
| | William E. Martin, III |
| | Chief Executive Officer |
DocumentEXHIBIT 32.2
Certification of the Chief Financial Officer
Pursuant to Rule 18 U.S.C. Section 1350
In connection with the Annual Report on Form 10-K of Maravai LifeSciences Holdings, Inc. (the “Company”) for the year ended December 31, 2023, as filed with the U.S. Securities and Exchange Commission (the “Report”), I, Kevin Herde, Chief Financial Officer of the Company, hereby certify, pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002, that, to my knowledge:
1. The Report fully complies with the requirements of Section 13(a) or 15(d) of the Securities Exchange Act of 1934, as amended; and
2. The information contained in the Report fairly presents, in all material respects, the financial condition and results of operations of the Company.
Date: February 29, 2024
| | | | | | | | |
| | /s/ Kevin Herde |
| | Kevin Herde |
| | Chief Financial Officer |
DocumentExhibit 97.1
Effective October 2, 2023
CLAWBACK POLICY
MARAVAI LIFESCIENCES HOLDINGS, INC.
PURPOSE
Maravai LifeSciences Holdings, Inc. (the “Company”) believes that it is in the best interests of the Company and its shareholders to create and maintain a culture that emphasizes integrity and accountability and that reinforces the Company’s pay-for-performance compensation philosophy. The Company’s Board of Directors (the “Board”) has therefore adopted this policy, which provides for the recoupment of certain executive compensation in the event that the Company is required to prepare an accounting restatement of its financial statements due to material noncompliance with any financial reporting requirement under the federal securities laws (this “Policy”). This Policy is designed to comply with Section 10D of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), the rules promulgated thereunder, and the listing standards of the national securities exchange on which the Company’s securities are listed.
ADMINISTRATION
This Policy shall be administered by the Compensation and Leadership Development Committee of the Board (the “Compensation Committee”). Any determinations made by the Compensation Committee shall be final and binding on all affected individuals.
COVERED EXECUTIVES
This Policy applies to the Company’s current and former executive officers (as determined by the Compensation Committee in accordance with Section 10D of the Exchange Act, the rules promulgated thereunder, and the listing standards of the national securities exchange on which the Company’s securities are listed) and such other senior executives or employees who may from time to time be deemed subject to this Policy by the Compensation Committee (collectively, the “Covered Executives”). This Policy shall be binding and enforceable against all Covered Executives.
RECOUPMENT; ACCOUNTING RESTATEMENT
In the event that the Company is required to prepare an accounting restatement of its financial statements due to the Company’s material noncompliance with any financial reporting requirement under the securities laws, including any required accounting restatement (i) to correct an error in previously issued financial statements that is material to the previously issued financial statements, or (ii) that would result in a material misstatement if the error were corrected in the current period or left uncorrected in the current period (each an “Accounting Restatement”), the Compensation Committee will reasonably promptly require reimbursement or forfeiture of the Overpayment (as defined below) received by any Covered Executive (x) after beginning service as a Covered Executive, (y) who served as a Covered Executive at any time during the performance period for the applicable Incentive-Based Compensation (as defined below), and (z) during the three (3) completed fiscal years immediately preceding the date on which the Company is required to prepare an Accounting Restatement and any transition period (that results from a change in the Company’s fiscal year) within or immediately following those three (3) completed fiscal years.
INCENTIVE-BASED COMPENSATION
For purposes of this Policy, “Incentive-Based Compensation” means any compensation that is granted, earned, or vested based wholly or in part upon the attainment of a financial reporting measure, including, but not limited to: (i) non-equity incentive plan awards that are earned solely or in part by satisfying a financial reporting measure performance goal; (ii) bonuses paid from a bonus pool, where the size of the pool is determined solely or in part by satisfying a financial reporting measure performance goal; (iii) other cash awards based on satisfaction of a financial reporting measure performance goal; (iv) restricted stock, restricted stock units, stock options, stock appreciation rights, and performance share units that are granted or vest solely or in part based on satisfaction of a financial reporting measure performance goal; and (v) proceeds from the sale of shares acquired through an incentive plan that were granted or vested solely or in part based on satisfaction of a financial reporting measure performance goal.
Compensation that would not be considered Incentive-Based Compensation includes, but is not limited to: (i) salaries; (ii) bonuses paid solely based on satisfaction of subjective standards, such as demonstrating leadership, and/or completion of a specified employment period; (iii) non-equity incentive plan awards earned solely based on satisfaction of strategic or operational measures; (iv) wholly time-based equity awards; and (v) discretionary bonuses or other compensation that is not paid from a bonus pool that is determined by satisfying a financial reporting measure performance goal.
A financial reporting measure is: (i) any measure that is determined and presented in accordance with the accounting principles used in preparing financial statements, or any measure derived wholly or in part from such measure, such as revenues, EBITDA, or net income or (ii) stock price and total shareholder return. Financial reporting measures include, but are not limited to: revenues; net income; operating income; profitability of one or more reportable segments; financial ratios (e.g., accounts receivable turnover and inventory turnover rates); net assets or net asset value per share; earnings before interest, taxes, depreciation and amortization; funds from operations and adjusted funds from operations; liquidity measures (e.g., working capital, operating cash flow); return measures (e.g., return on invested capital, return on assets); earnings measures (e.g., earnings per share); sales per square foot or same store sales, where sales is subject to an accounting restatement; revenue per user, or average revenue per user, where revenue is subject to an accounting restatement; cost per employee, where cost is subject to an accounting restatement; any of such financial reporting measures relative to a peer group, where the Company’s financial reporting measure is subject to an accounting restatement; and tax basis income.
OVERPAYMENT: AMOUNT SUBJECT TO RECOVERY
The amount to be recovered will be the amount of Incentive-Based Compensation received that exceeds the amount of Incentive-Based Compensation that otherwise would have been received had it been determined based on the restated amounts, and must be computed without regard to any taxes paid (the “Overpayment”). Incentive-Based Compensation is deemed “received” in the Company’s fiscal period during which the financial reporting measure specified in the incentive-based compensation award is attained, even if the vesting, payment or grant of the incentive-based compensation occurs after the end of that period.
For Incentive-Based Compensation based on stock price or total shareholder return, where the amount of erroneously awarded compensation is not subject to mathematical recalculation directly from the information in the Accounting Restatement, the amount must be based on a reasonable estimate of the effect of the Accounting Restatement on the stock price or total shareholder return upon which the Incentive-Based Compensation was received, and the Company must maintain documentation of the determination of that reasonable estimate and provide such documentation to the exchange on which the Company’s securities are listed.
METHOD OF RECOUPMENT
The Compensation Committee will determine, in its sole discretion, the method or methods for recouping any Overpayment hereunder which may include, without limitation:
•requiring reimbursement of cash Incentive-Based Compensation previously paid;
•seeking recovery of any gain realized on the vesting, exercise, settlement, sale, transfer, or other disposition of any equity-based awards granted as Incentive-Based Compensation;
•offsetting any or all of the Overpayment from any compensation otherwise owed by the Company to the Covered Executive;
•cancelling outstanding vested or unvested equity awards; and/or
•taking any other remedial or recovery action permitted by law, as determined by the Compensation Committee.
LIMITATION ON RECOVERY; NO ADDITIONAL PAYMENTS
The right to recovery will be limited to Overpayments received during the three (3) completed fiscal years prior to the date on which the Company is required to prepare an Accounting Restatement and any transition period (that results from a change in the Company’s fiscal year) within or immediately following those three (3) completed fiscal years. In no event shall the Company be required to award Covered Executives an additional payment if the restated or accurate financial results would have resulted in a higher Incentive-Based Compensation payment.
NO INDEMNIFICATION
The Company shall not indemnify any Covered Executives against the loss of any incorrectly awarded Incentive-Based Compensation.
INTERPRETATION
The Compensation Committee is authorized to interpret and construe this Policy and to make all determinations necessary, appropriate, or advisable for the administration of this Policy. It is intended that this Policy be interpreted in a manner that is consistent with the requirements of Section 10D of the Exchange Act and the applicable rules or standards adopted by the Securities and Exchange Commission or any national securities exchange on which the Company’s securities are listed.
EFFECTIVE DATE
This Policy shall be effective as of the date it is adopted by the Board (the “Effective Date”) and shall apply to Incentive-Based Compensation (including Incentive-Based Compensation granted pursuant to arrangements existing prior to the Effective Date). Notwithstanding the foregoing, this Policy shall only apply to Incentive-Based Compensation received (as determined pursuant to this Policy) on or after the effective date of NASDAQ Listing Rule 5608.
AMENDMENT; TERMINATION
The Board may amend this Policy from time to time in its discretion. The Board may terminate this Policy at any time.
OTHER RECOUPMENT RIGHTS
The Board intends that this Policy will be applied to the fullest extent of the law. The Compensation Committee may require that any employment or service agreement, cash-based bonus plan or program, equity award agreement, or similar agreement entered into on or after the adoption of this Policy shall, as a condition to the grant of any benefit thereunder, require a Covered Executive to agree to abide by the terms of this Policy. Any right of recoupment under this Policy is in addition to, and not in lieu of, any other remedies or rights of recoupment that may be available to the Company pursuant to the terms of any similar policy in any employment agreement, equity award agreement, cash-based bonus plan or program, or similar agreement and any other legal remedies available to the Company.
IMPRACTICABILITY
The Compensation Committee shall recover any Overpayment in accordance with this Policy except to the extent that the Compensation Committee determines such recovery would be impracticable because:
(A) The direct expense paid to a third party to assist in enforcing this Policy would exceed the amount to be recovered;
(B) Recovery would violate home country law of the Company where that law was adopted prior to November 28, 2022; or
(C) Recovery would likely cause an otherwise tax-qualified retirement plan, under which benefits are broadly available to employees of the Company, to fail to meet the requirements of 26 U.S.C. 401(a)(13) or 26 U.S.C. 411(a) and regulations thereunder.
SUCCESSORS
This Policy shall be binding and enforceable against all Covered Executives and their beneficiaries, heirs, executors, administrators or other legal representatives.


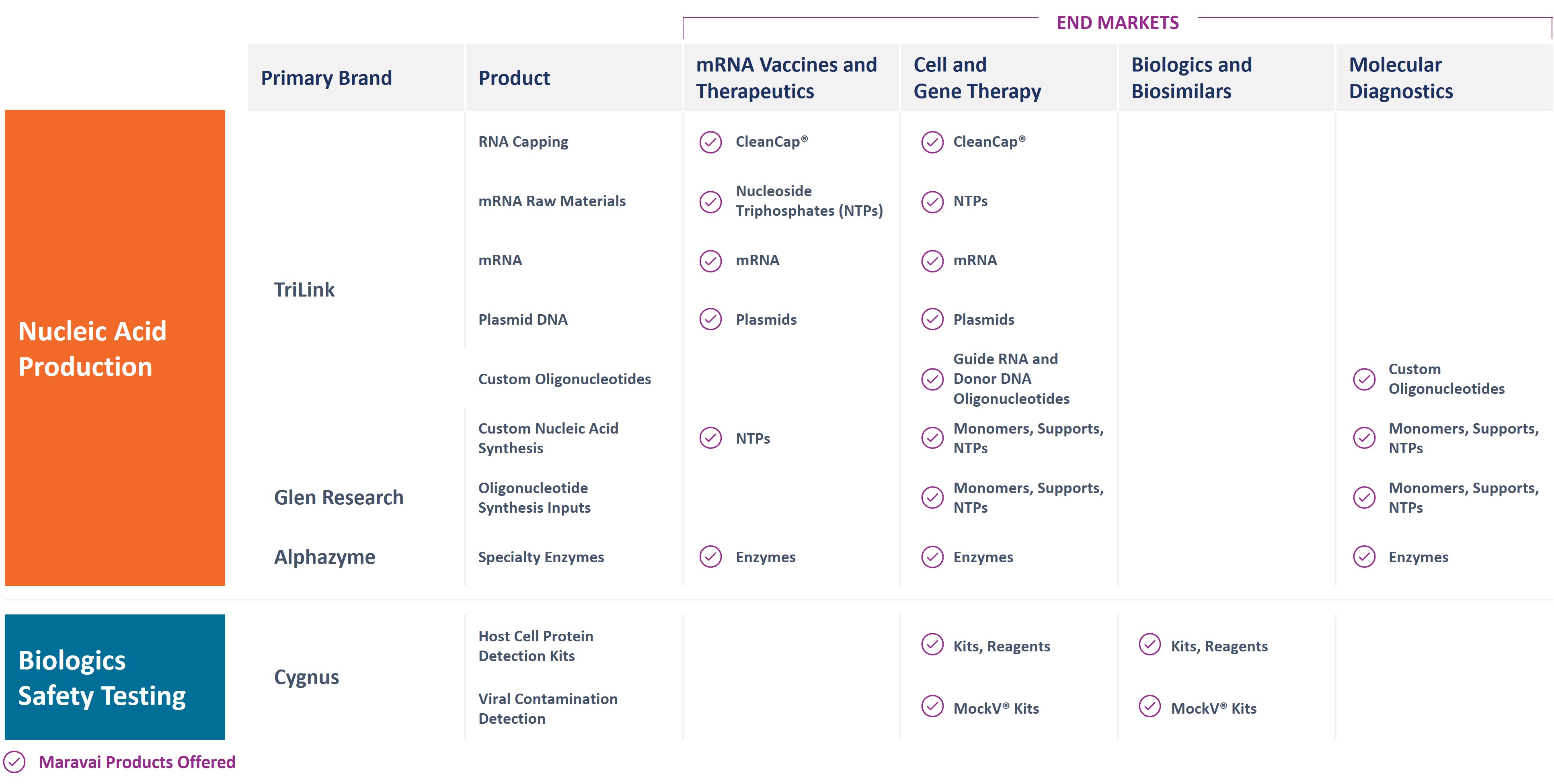

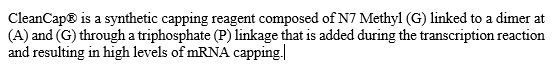

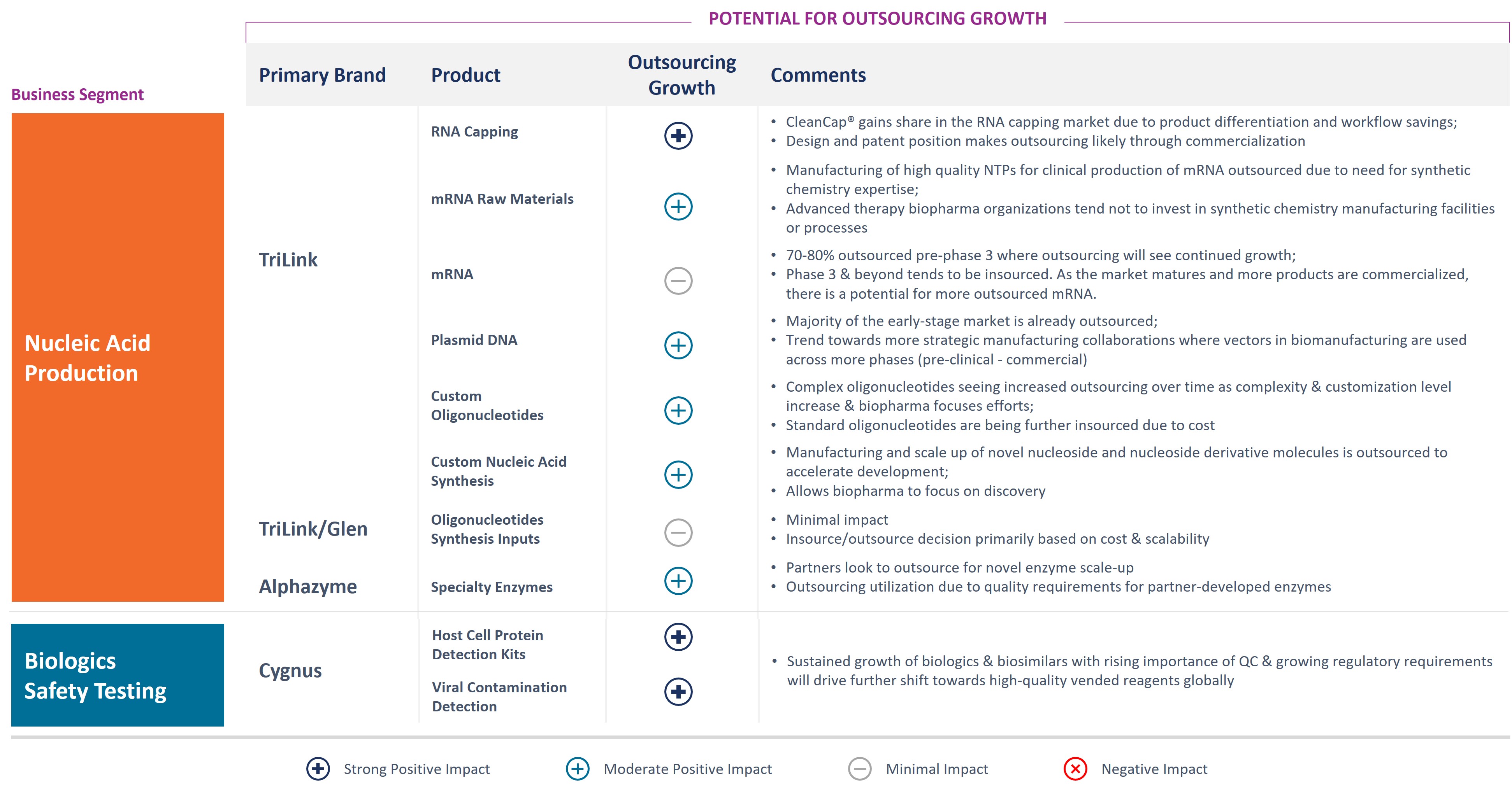

 all or any part of the Demised Premises to any party other than a Permitted Assignee which satisfies the terms of this Section 13, Tenant shall submit to Landlord in writing at least thirty (30) days prior to the proposed effective date of the assignment or sublease transaction (i) a request for permission to assign or sublet setting forth the proposed effective date (which shall be no less than thirty (30) days after the delivery of such notice to Landlord); (ii) the name of the proposed subtenant or assignee; (iii) the nature of the business to be carried on in the Demised Premises after the assignment or sublease; (iv) the terms and provisions of the proposed assignment or sublease instrument(s); (v) current financial statements of the proposed subtenant or assignee; and (vi) such additional information as Landlord may reasonably request in order to make a reasoned judgment regarding the proposed assignment or sublease.
all or any part of the Demised Premises to any party other than a Permitted Assignee which satisfies the terms of this Section 13, Tenant shall submit to Landlord in writing at least thirty (30) days prior to the proposed effective date of the assignment or sublease transaction (i) a request for permission to assign or sublet setting forth the proposed effective date (which shall be no less than thirty (30) days after the delivery of such notice to Landlord); (ii) the name of the proposed subtenant or assignee; (iii) the nature of the business to be carried on in the Demised Premises after the assignment or sublease; (iv) the terms and provisions of the proposed assignment or sublease instrument(s); (v) current financial statements of the proposed subtenant or assignee; and (vi) such additional information as Landlord may reasonably request in order to make a reasoned judgment regarding the proposed assignment or sublease. the Demised Premises, including the roof or any setbacks reasonably designated by Landlord, provided that such Telecom Equipment is installed, maintained, and operated exclusively to serve the operations of Tenant without the need for any further consent or approval from Landlord. Without limiting the generality of the foregoing, but subject to applicable Law, nothing contained herein shall be deemed to restrict Tenant’s right to install Telecom Equipment within the Demised Premises. Tenant shall be permitted to access all communications hardlines entering into the Building directly from the street. Landlord shall have no interest in any Telecom Equipment installed by or for the benefit of Tenant, it being understood and agreed that any and all such installations shall be and remain the sole property of Tenant and such installations may be removed from the Demised Premises at any time during the Term hereof by Tenant. Upon Tenant’s removal of any of its Telecom Equipment, or any other of Tenant’s property whatsoever, Tenant shall repair, and as necessary replace, the Demised Premises to the condition that the same was in when Tenant took occupancy, reasonable wear and tear excepted.
the Demised Premises, including the roof or any setbacks reasonably designated by Landlord, provided that such Telecom Equipment is installed, maintained, and operated exclusively to serve the operations of Tenant without the need for any further consent or approval from Landlord. Without limiting the generality of the foregoing, but subject to applicable Law, nothing contained herein shall be deemed to restrict Tenant’s right to install Telecom Equipment within the Demised Premises. Tenant shall be permitted to access all communications hardlines entering into the Building directly from the street. Landlord shall have no interest in any Telecom Equipment installed by or for the benefit of Tenant, it being understood and agreed that any and all such installations shall be and remain the sole property of Tenant and such installations may be removed from the Demised Premises at any time during the Term hereof by Tenant. Upon Tenant’s removal of any of its Telecom Equipment, or any other of Tenant’s property whatsoever, Tenant shall repair, and as necessary replace, the Demised Premises to the condition that the same was in when Tenant took occupancy, reasonable wear and tear excepted.