
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of The Securities Exchange Act of 1934
Date of Report (Date of earliest event reported)
(Exact name of Registrant as Specified in its Charter)
| (State or other jurisdiction of incorporation) |
(Commission File Number) |
(I.R.S. Employer Identification No.) |
| (Address of principal executive offices) | (Zip Code) |
(Registrant’s telephone number, including area code)
Not Applicable
(Former name or former address, if changed since last report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
| Title of Each Class |
Trading |
Name of Each Exchange on Which Registered | ||
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act.
| Item 7.01 | Regulation FD Disclosure. |
On May 1, 2023, Cabaletta Bio, Inc. (the “Company” or “Cabaletta”) posted to the “Investors & Media” section of the Company’s website at www.cabalettabio.com an updated corporate presentation providing a corporate overview and updated development plan (the “Corporate Presentation”). A copy of the Corporate Presentation is attached hereto as Exhibit 99.1 and is incorporated by reference into this Item 7.01 of this Current Report on Form 8-K.
On May 1, 2023, the Company issued a Press Release announcing that the U.S. Food and Drug Administration (“FDA”) has granted Fast Track Designation for CABA-201, a 4-1BB-containing fully human CD19-CAR T cell investigational therapy, to deplete CD19-positive B cells and improve disease activity in patients with systemic lupus erythematosus (SLE) and lupus nephritis (LN) (the “Press Release”). A copy of the Press Release is attached hereto as Exhibit 99.2 and is incorporated by reference into this Item 7.01 of this Current Report on Form 8-K.
The information contained in Item 7.01 of this Current Report on Form 8-K, including Exhibits 99.1 and 99.2 attached hereto, is being furnished and shall not be deemed to be “filed” for the purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that section and shall not be incorporated by reference in any filing under the Securities Act of 1933, as amended, or the Exchange Act, except as shall be expressly set forth by specific reference in such filing.
| Item 8.01 | Other Events. |
On May 1, 2023, the Company issued the Press Release announcing that the FDA has granted Fast Track Designation for CABA-201, a 4-1BB-containing fully human CD19-CAR T cell investigational therapy, designed to deplete CD19-positive B cells and improve disease activity in patients with SLE and LN. Cabaletta has been cleared to initiate a Phase 1/2 clinical trial of CABA-201 for the treatment of 6 SLE patients with active LN, and in a separate parallel cohort, 6 patients with active SLE without renal involvement.
SLE is a chronic, potentially severe, autoimmune disease in which the immune system attacks healthy tissue throughout the body, most commonly impacting young women between the ages of 15 and 40 with higher frequency and greater severity in people of color. It is characterized by abnormal B cell function and autoantibody production resulting in a range of clinical manifestations including end organ damage and an increased risk of death. SLE affects an estimated 160,000-320,000 patients in the U.S, with LN as the most common end-organ manifestation, affecting approximately 40% of SLE patients. Among these patients, the risk of end-stage renal disease is approximately 17% and the risk of death is approximately 12%, each within 10 years of diagnosis.
CABA-201 is designed to be given as a one-time infusion, to evaluate its potential to transiently, but fully, eliminate B cells, enabling an “immune system reset” with durable remission in patients with SLE. The Phase 1/2 clinical trial is an open-label study designed to evaluate CABA-201 in SLE subjects with active LN or active SLE without renal involvement. CABA-201 will be administered at a dose of 1.0 x 106 cells/kg. This study will enroll 6 subjects in an active LN cohort and 6 subjects in an active SLE without renal involvement cohort, in parallel. Subjects will be treated with a standard preconditioning regimen consisting of fludarabine and cyclophosphamide prior to CABA-201 infusion. This represents the first trial assessing Cabaletta’s CARTA (Chimeric Antigen Receptor T cells for Autoimmunity) strategy.
Forward-Looking Statements
This 8-K contains “forward-looking statements” of Cabaletta within the meaning of the Private Securities Litigation Reform Act of 1995, as amended, including without limitation, express or implied statements regarding: expectations regarding: Cabaletta’s ability to retain and recognize the intended incentives conferred Fast Track Designation for CABA-201 to deplete CD19-positive B cells and improve disease activity in patients with SLE and LN; Cabaletta’s ability to grow its autoimmune-focused pipeline; the Company’s business plans and objectives; Cabaletta Bio’s expectations around the potential success and therapeutic benefits of CABA-201, including its belief that CABA-201 may enable an “immune system reset” and provide deep and durable responses for patients with SLE and potentially for patients diagnosed with other autoimmune disease; the Company’s plans to initiate a Phase 1/2 clinical trial of CABA-201 in patients with SLE, including its anticipated progress, clinical trial design, ability to leverage its experience in autoimmune cell therapy and lupus product development; Cabaletta’s ability to enroll the requisite number of patients, dose each dosing cohort in the intended manner and advance the trial as planned in its Phase 1/2 clinical trial of CABA-201; and the ability to accelerate Cabaletta’s pipeline and develop meaningful therapies for patients, including in collaboration with academic and industry partners and the ability to optimize such collaborations on its development programs.
Any forward-looking statements in this 8-K are based on management’s current expectations and beliefs of future events, and are subject to a number of risks and uncertainties that could cause actual results to differ materially and adversely from those set forth in or implied by such forward-looking statements. These risks and uncertainties include, but are not limited to: the risk that signs of biologic activity or persistence may not inform long-term results; Cabaletta’s ability to demonstrate sufficient evidence of safety, efficacy and tolerability in its preclinical studies and clinical trials of DSG3-CAART, MuSK-CAART and CABA-201; the risk that the results observed with the similarly-designed construct employed in the recent Nature Medicine publication, including due to the dosing regimen, are not indicative of the results we seek to achieve with CABA-201; risks related to clinical trial site activation or enrollment rates that are lower than expected; risks related to unexpected safety or efficacy data observed during clinical studies; risks related to volatile market and economic conditions; risks related to the impact of public health epidemics affecting countries or regions in which Cabaletta has operations or does business, such as COVID-19; Cabaletta’s ability to retain and recognize the intended incentives conferred by Orphan Drug Designation and Fast Track Designation for its product candidates, as applicable; risks related to Cabaletta’s ability to protect and maintain its intellectual property position; risks related to fostering and maintaining successful relationships with Cabaletta’s collaboration and manufacturing partners; uncertainties related to the initiation and conduct of studies and other development requirements for its product candidates; the risk that any one or more of Cabaletta’s product candidates will not be successfully developed and/or commercialized; and the risk that the initial or interim results of preclinical studies or clinical studies will not be predictive of future results in connection with future studies. For a discussion of these and other risks and uncertainties, and other important factors, any of which could cause Cabaletta’s actual results to differ from those contained in the forward-looking statements, see the section entitled “Risk Factors” in Cabaletta’s most recent annual report on Form 10-K as well as discussions of potential risks, uncertainties, and other important factors in Cabaletta’s other filings with the Securities and Exchange Commission. All information in this 8-K is as of the date hereof, and Cabaletta undertakes no duty to update this information unless required by law.
| Item 9.01 | Financial Statements and Exhibits. |
(d) Exhibits
| 99.1 | Cabaletta Bio, Inc. Corporate Presentation, dated May 1, 2023, furnished herewith. | |
| 99.2 | Press Release issued by the registrant on May 1, 2023, furnished herewith. | |
| 104 | Cover Page Interactive Data File (embedded within the Inline XBRL Document). | |
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, as amended, the registrant has duly caused this report to be signed on its behalf by the undersigned, hereunto duly authorized.
| CABALETTA BIO, INC. | ||||||
| Date: May 1, 2023 | By: | /s/ Steven Nichtberger | ||||
| Steven Nichtberger, M.D. | ||||||
| President and Chief Executive Officer | ||||||

Corporate Presentation MAY 2023 Exhibit 99.1

Disclaimer The following presentation, including any printed or electronic copy of these slides, the talks given by the presenters, the information communicated during any delivery of the presentation and any question and answer session and any document or material distributed at or in connection with the presentation (collectively, the “Presentation”) has been prepared by Cabaletta Bio, Inc. (“we,” “us,” “our,” “Cabaletta” or the “Company”) and is made for informational purposes only. This Presentation does not purport to be a prospectus, to be complete or to contain all of the information you may desire. Statements contained herein are made as of the date of this Presentation unless stated otherwise, and this Presentation shall not under any circumstances create an implication that the information contained herein is correct as of any time after such date or that information will be updated or revised to reflect information that subsequently becomes available or changes occurring after the date hereof. This Presentation may contain “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995 relating to our business, operations, and financial conditions, and include, but are not limited to, express or implied statements regarding our current beliefs, expectations and assumptions regarding: our ability to retain and recognize the intended incentives conferred Fast Track Designation for CABA-201 to deplete CD19-positive B cells and improve disease activity in patients with systemic lupus erythematosus (SLE) and lupus nephritis (LN); our business, future plans and strategies for our CAAR T and CARTA technologies and CABA™ platform; our ability to grow our autoimmune-focused pipeline; the ability to capitalize on and potential benefits resulting from the translational research partnership with Professor Georg Schett and the exclusive license agreement with IASO Bio; our expectations around the potential success and therapeutic benefits of CABA-201, including our belief that CABA-201 may enable an “immune system reset” and provide deep and durable responses for patients with Systemic Lupus Erythematosus ("SLE") and potentially for patients diagnosed with other autoimmune disease; our plans to initiate a Phase 1/2 clinical trial of CABA-201 in patients with SLE, including our anticipated progress, clinical trial design, ability to leverage our experience in autoimmune cell therapy and lupus product development; our planned initial clinical data read-out in the first half of 2024; our ability to enroll the requisite number of patients, dose each dosing cohort in the intended manner, and advance the trial as planned in our Phase 1/2 clinical trial of CABA-201; the timing any planned regulatory filings for our development programs; the progress and results of our DesCAARTes™ Phase 1 trial, including the significance and impact around reported safety and clinical and translational data of cohorts from our DesCAARTes™ trial, and our ability to advance dose escalation and initiate combination cohorts and to optimize our targeted cell therapy; our ability to implement a pre-treatment regimen and the potential ability to enhance in vivo DSG3-CAART exposure; the therapeutic potential and clinical benefits of our product candidates; the expectation that Cabaletta may improve outcomes for patients suffering from mucosal pemphigus vulgaris, myasthenia gravis, or other autoimmune diseases; our ability to escalate dosing as high as 10 to 15 billion cells in cohort A6m, initiate dosing in a combination cohort or otherwise; our ability to implement a pre-treatment regimen and the potential ability to enhance in vivo DSG3-CAART exposure; our ability to evaluate, and the potential significance of, the relationship between DSG3-CAART persistence and potential clinical responses in patients with mPV; our ability to safely retreat additional patients and whether we will continue to observe a lack of immune-mediated clearance of DSG3-CAART cells after retreatment and repeat dosing of patients; our ability to successfully complete our preclinical and clinical studies for our product candidates, including CABA-201, our ongoing Phase 1 DesCAARTes™ trial, and our ongoing Phase 1 MusCAARTes™ trial of MuSK-CAART, including our ability to enroll the requisite number of patients, dose each dosing cohort in the intended manner, and progress the trial; the ability of MuSK-CAART to target B cells that differentiate into antibody secreting cells, which produce autoantibodies against muscle-specific kinase; our ability to obtain and maintain regulatory approval of our product candidates, including our expectations regarding the intended incentives conferred by and ability to retain Orphan Drug Designation and Fast Track Designation for our product candidates, as applicable; the further expansion and development of our modular CABA™ platform across a range of autoimmune diseases; our ability to contract with third-party suppliers and manufacturers, implement an enhanced manufacturing process and further develop our internal manufacturing strategy, capabilities and facilities; our potential commercial opportunities, including value and addressable market, for our product candidates; our expectations regarding our use of capital and other financial results; and our ability to fund operations into the first quarter of 2025. Words such as, but not limited to, “look forward to,” “believe,” “expect,” “anticipate,” “estimate,” “intend,” “plan,” “would,” “should” and “could,” and similar expressions or words, identify forward-looking statements. Various risks, uncertainties and assumptions could cause actual results to differ materially from those anticipated or implied in our forward-looking statements. Such risks and uncertainties include, but are not limited to, risks related to the success, cost, and timing of our product candidate development activities and preclinical studies and clinical trials, risks related to our ability to demonstrate sufficient evidence of safety, efficacy and tolerability in our preclinical studies and clinical trials of CABA-201, DSG3-CAART and MuSK-CAART, the risk that the results observed with the similarly-designed construct employed in the recent Nature Medicine publication, including due to the dosing regimen, are not indicative of the results we seek to achieve with CABA-201, our plans to evaluate additional cohorts in the DesCAARTes™ trial, including a cohort implementing a pre-treatment regimen, the risk that signs of biologic activity or persistence may not inform long-term results, the risk that persistence observed with effective CART-19 oncology studies in combination with lymphodepletion is not indicative of, or applicable to, clinical responses in patients with mPV, risks related to clinical trial site activation or enrollment rates that are lower than expected, our ability to protect and maintain our intellectual property position, risks related to our relationship with third parties, uncertainties related to regulatory agencies’ evaluation of regulatory filings and other information related to our product candidates, our ability to retain and recognize the intended incentives conferred by any Orphan Drug Designation and Fast Track Designations, the risk that any one or more of our product candidates will not be successfully developed and commercialized, the risk that the results of preclinical studies or clinical studies will not be predictive of future results in connection with future studies, the impact of COVID-19 on the timing, progress, interpretability of data, and results of ongoing or planned clinical trials and risks relating to as a result of extraordinary events or circumstances such as the COVID-19 pandemic, and any business interruptions to our operations or to those of our clinical sites, manufacturers, suppliers, or other vendors resulting from the COVID-19 pandemic or similar public health crisis. New risks and uncertainties may emerge from time to time, and it is not possible to predict all risks and uncertainties. Except as required by applicable law, we do not plan to publicly update or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise. Although we believe the expectations reflected in such forward-looking statements are reasonable, we can give no assurance that such expectations will prove to be correct. Accordingly, you are cautioned not to place undue reliance on these forward-looking statements. No representations or warranties (expressed or implied) are made about the accuracy of any such forward-looking statements. For a discussion of these and other risks and uncertainties, and other important factors, any of which could cause our actual results to differ materially from those contained in the forward-looking statements, see the section entitled "Risk Factors" in our most recent annual report on Form 10-K, as well as discussions of potential risks, uncertainties, and other important factors in our other and subsequent filings with the Securities and Exchange Commission. Certain information contained in this Presentation relates to or is based on studies, publications, surveys and other data obtained from third-party sources and the Company’s own internal estimates and research. While the Company believes these third-party sources to be reliable as of the date of this Presentation, it has not independently verified, and makes no representation as to the adequacy, fairness, accuracy or completeness of, any information obtained from third-party sources. The Company is the owner of various trademarks, trade names and service marks. Certain other trademarks, trade names and service marks appearing in this Presentation are the property of third parties. Solely for convenience, the trademarks and trade names in this Presentation are referred to without the ® and TM symbols, but such references should not be construed as any indicator that their respective owners will not assert, to the fullest extent under applicable law, their rights thereto.

Develop and launch the first curative targeted cellular therapies for patients with autoimmune diseases

Leveraging years of experience with clinical sites to initiate Phase 1/2 trial of CABA-201 in SLE & LN Cabaletta®: Pursuing cures for a broad range of autoimmune diseases CAART – Chimeric AutoAntibody Receptor T cells; CARTA – Chimeric Antigen Receptor T cells for Autoimmunity; IND – Investigational New Drug; SLE – Systemic lupus erythematosus Mackensen, Andreas, et al. "Anti-CD19 CAR T cell therapy for refractory systemic lupus erythematosus." Nature Medicine (2022): 1-9. Müller, Fabian, et al. "CD19-targeted CAR T cells in refractory antisynthetase syndrome." The Lancet (2023). Assumes no dose-limiting toxicities are observed in the cohort and uninterrupted enrollment occur in the trials. Dai, Zhenyu, et al. "Development and functional characterization of novel fully human anti‐CD19 CARs for T‐cell therapy." Journal of Cellular Physiology 236.8 (2021): 5832-5847. Efficient clinical trial design – including initial dose and number of patients in each cohort 1.0 x 106 cells/kg initial dose of CABA-201 is identical to dose used in academic SLE1 and myositis2 studies Two parallel cohorts with 6 patients each – 1) lupus nephritis (LN) and 2) SLE without renal involvement; FDA Fast Track Designation CABA-201 has been specifically engineered for patients with autoimmune diseases Fully human CD19 binder with data in ~20 oncology patients with safety profile supporting further evaluation in autoimmunity Same 4-1BB costimulatory domain and similar CD19 binder affinity4 as used in the academic SLE1 & myositis studies2 Potential to cure a broad range of autoimmune diseases where B cells have a role initiating or maintaining disease DesCAARTes™ trial in mucosal pemphigus vulgaris – 1 month safety & persistence data anticipated 1H234 Enrolling in combination sub-study using pre-treatment with IVIg & cyclophosphamide MusCAARTes™ trial in MuSK myasthenia gravis – leveraging insights from experience with DSG3-CAART Initiated in 4Q22; received FDA Fast Track Designation & Orphan Drug Designation CARTA Strategy | CABA-201 (4-1BB CD19-CAR T) to be evaluated in a Phase 1/2 study in SLE CAART Strategy | DSG3-CAART & MuSK-CAART clinical studies evaluating combination regimens Initial CABA-201 clinical data & 6-mo combination data from CAART trials expected by 1H244
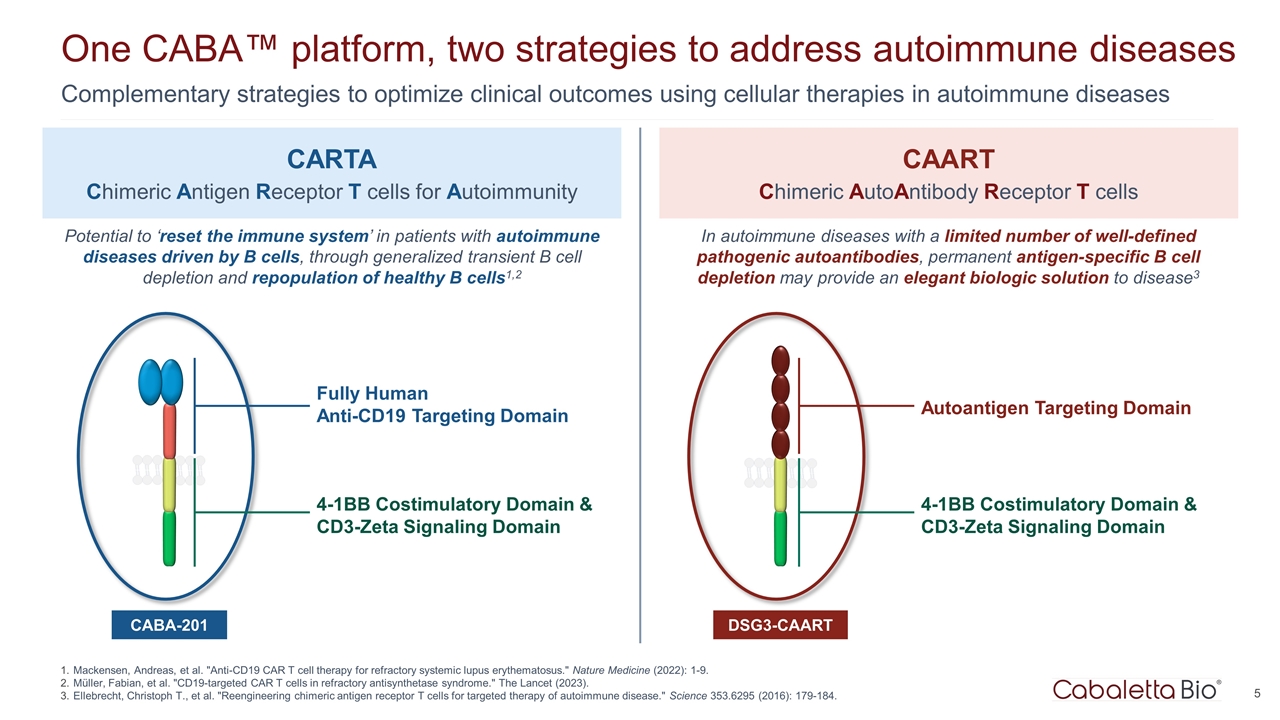
One CABA™ platform, two strategies to address autoimmune diseases Complementary strategies to optimize clinical outcomes using cellular therapies in autoimmune diseases Mackensen, Andreas, et al. "Anti-CD19 CAR T cell therapy for refractory systemic lupus erythematosus." Nature Medicine (2022): 1-9. Müller, Fabian, et al. "CD19-targeted CAR T cells in refractory antisynthetase syndrome." The Lancet (2023). Ellebrecht, Christoph T., et al. "Reengineering chimeric antigen receptor T cells for targeted therapy of autoimmune disease." Science 353.6295 (2016): 179-184. Fully Human Anti-CD19 Targeting Domain CARTA Chimeric Antigen Receptor T cells for Autoimmunity CAART Chimeric AutoAntibody Receptor T cells CABA-201 4-1BB Costimulatory Domain & CD3-Zeta Signaling Domain DSG3-CAART Potential to ‘reset the immune system’ in patients with autoimmune diseases driven by B cells, through generalized transient B cell depletion and repopulation of healthy B cells1,2 In autoimmune diseases with a limited number of well-defined pathogenic autoantibodies, permanent antigen-specific B cell depletion may provide an elegant biologic solution to disease3 Autoantigen Targeting Domain 4-1BB Costimulatory Domain & CD3-Zeta Signaling Domain
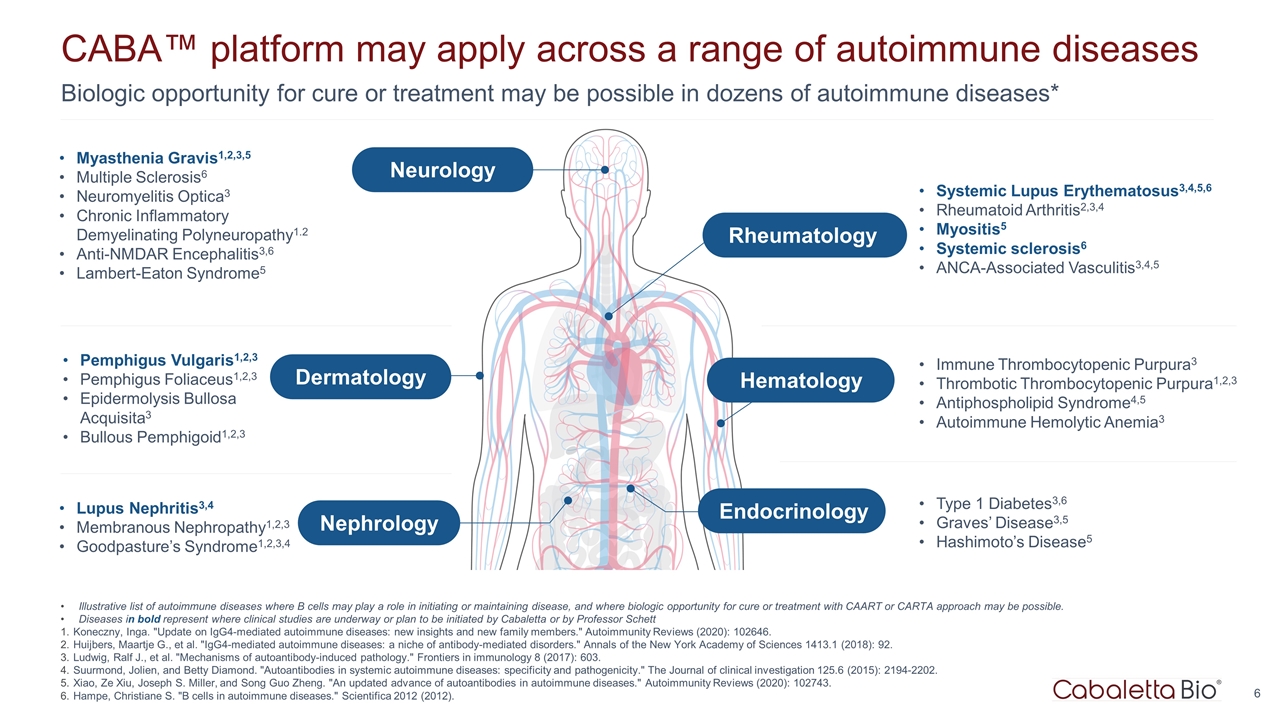
Biologic opportunity for cure or treatment may be possible in dozens of autoimmune diseases* CABA™ platform may apply across a range of autoimmune diseases Pemphigus Vulgaris1,2,3 Pemphigus Foliaceus1,2,3 Epidermolysis Bullosa Acquisita3 Bullous Pemphigoid1,2,3 Lupus Nephritis3,4 Membranous Nephropathy1,2,3 Goodpasture’s Syndrome1,2,3,4 Myasthenia Gravis1,2,3,5 Multiple Sclerosis6 Neuromyelitis Optica3 Chronic Inflammatory Demyelinating Polyneuropathy1.2 Anti-NMDAR Encephalitis3,6 Lambert-Eaton Syndrome5 Systemic Lupus Erythematosus3,4,5,6 Rheumatoid Arthritis2,3,4 Myositis5 Systemic sclerosis6 ANCA-Associated Vasculitis3,4,5 Immune Thrombocytopenic Purpura3 Thrombotic Thrombocytopenic Purpura1,2,3 Antiphospholipid Syndrome4,5 Autoimmune Hemolytic Anemia3 Type 1 Diabetes3,6 Graves’ Disease3,5 Hashimoto’s Disease5 Illustrative list of autoimmune diseases where B cells may play a role in initiating or maintaining disease, and where biologic opportunity for cure or treatment with CAART or CARTA approach may be possible. Diseases in bold represent where clinical studies are underway or plan to be initiated by Cabaletta or by Professor Schett Koneczny, Inga. "Update on IgG4-mediated autoimmune diseases: new insights and new family members." Autoimmunity Reviews (2020): 102646. Huijbers, Maartje G., et al. "IgG4-mediated autoimmune diseases: a niche of antibody-mediated disorders." Annals of the New York Academy of Sciences 1413.1 (2018): 92. Ludwig, Ralf J., et al. "Mechanisms of autoantibody-induced pathology." Frontiers in immunology 8 (2017): 603. Suurmond, Jolien, and Betty Diamond. "Autoantibodies in systemic autoimmune diseases: specificity and pathogenicity." The Journal of clinical investigation 125.6 (2015): 2194-2202. Xiao, Ze Xiu, Joseph S. Miller, and Song Guo Zheng. "An updated advance of autoantibodies in autoimmune diseases." Autoimmunity Reviews (2020): 102743. Hampe, Christiane S. "B cells in autoimmune diseases." Scientifica 2012 (2012). Dermatology Nephrology Neurology Rheumatology Hematology Endocrinology
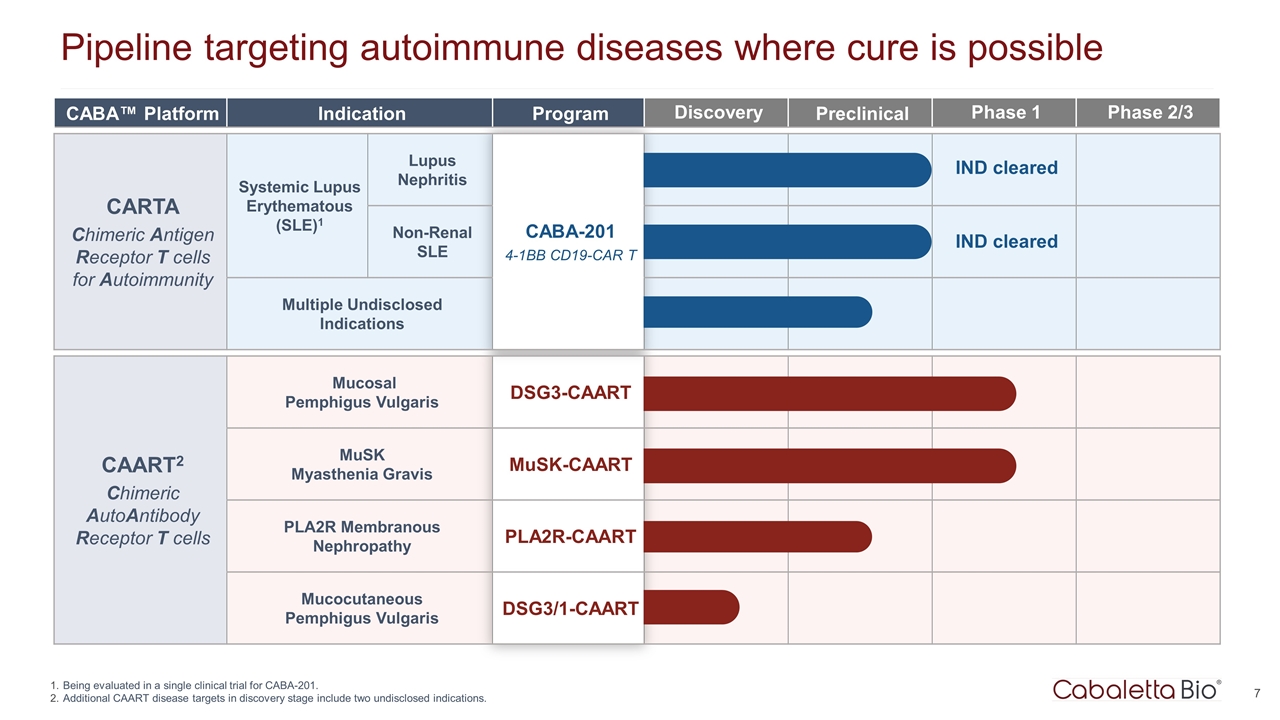
Pipeline targeting autoimmune diseases where cure is possible Being evaluated in a single clinical trial for CABA-201. Additional CAART disease targets in discovery stage include two undisclosed indications. CABA™ Platform Indication Program Discovery Preclinical Phase 1 Phase 2/3 CARTA Chimeric Antigen Receptor T cells for Autoimmunity Systemic Lupus Erythematous (SLE)1 Lupus Nephritis CABA-201 4-1BB CD19-CAR T Non-Renal SLE CABA-201 4-1BB CD19-CAR T Multiple Undisclosed Indications CAART2 Chimeric AutoAntibody Receptor T cells Mucosal Pemphigus Vulgaris DSG3-CAART MuSK Myasthenia Gravis MuSK-CAART PLA2R Membranous Nephropathy PLA2R-CAART Mucocutaneous Pemphigus Vulgaris DSG3/1-CAART IND cleared IND cleared

Chimeric Antigen Receptor T Cells for Autoimmunity CABA-201
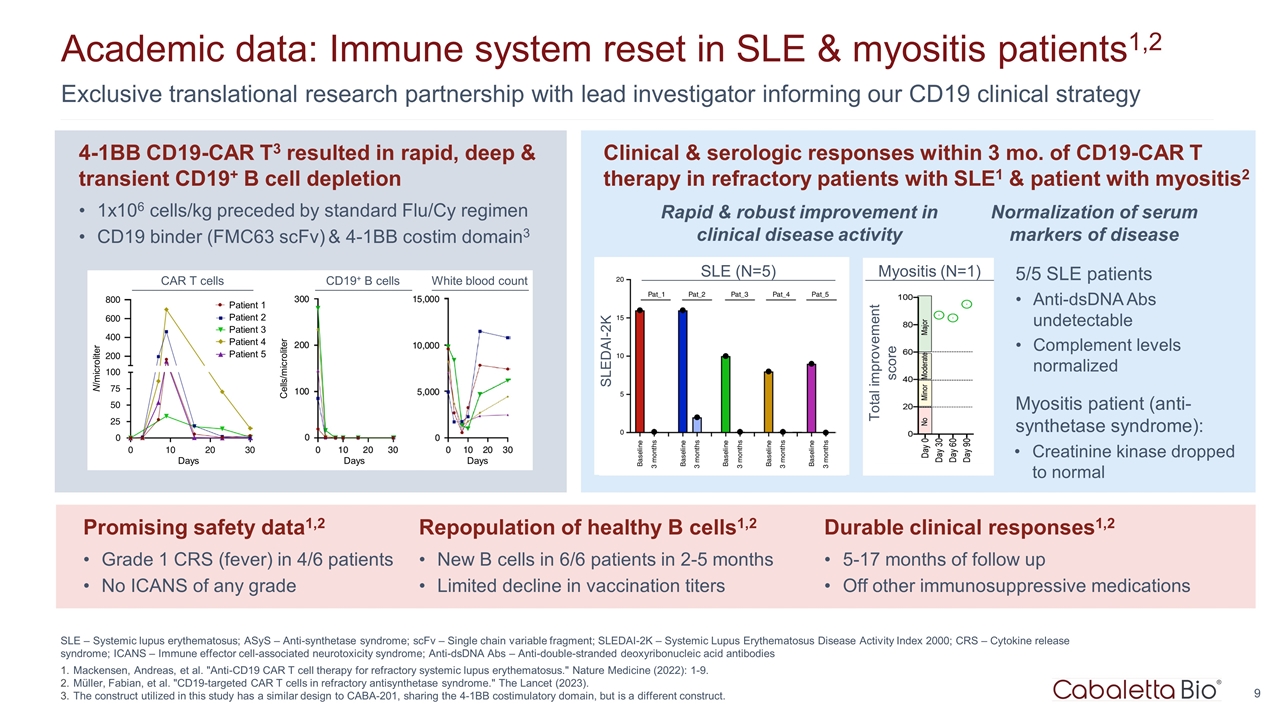
SLE – Systemic lupus erythematosus; ASyS – Anti-synthetase syndrome; scFv – Single chain variable fragment; SLEDAI-2K – Systemic Lupus Erythematosus Disease Activity Index 2000; CRS – Cytokine release syndrome; ICANS – Immune effector cell-associated neurotoxicity syndrome; Anti-dsDNA Abs – Anti-double-stranded deoxyribonucleic acid antibodies Mackensen, Andreas, et al. "Anti-CD19 CAR T cell therapy for refractory systemic lupus erythematosus." Nature Medicine (2022): 1-9. Müller, Fabian, et al. "CD19-targeted CAR T cells in refractory antisynthetase syndrome." The Lancet (2023). The construct utilized in this study has a similar design to CABA-201, sharing the 4-1BB costimulatory domain, but is a different construct. Exclusive translational research partnership with lead investigator informing our CD19 clinical strategy Academic data: Immune system reset in SLE & myositis patients1,2 5/5 SLE patients Anti-dsDNA Abs undetectable Complement levels normalized Myositis patient (anti-synthetase syndrome): Creatinine kinase dropped to normal Durable clinical responses1,2 5-17 months of follow up Off other immunosuppressive medications CAR T cells CD19+ B cells White blood count Patient 1 Patient 2 Patient 3 Patient 4 Patient 5 Days Days Days Cells/microliter N/microliter Promising safety data1,2 Grade 1 CRS (fever) in 4/6 patients No ICANS of any grade 4-1BB CD19-CAR T3 resulted in rapid, deep & transient CD19+ B cell depletion 1x106 cells/kg preceded by standard Flu/Cy regimen CD19 binder (FMC63 scFv) & 4-1BB costim domain3 Clinical & serologic responses within 3 mo. of CD19-CAR T therapy in refractory patients with SLE1 & patient with myositis2 Repopulation of healthy B cells1,2 New B cells in 6/6 patients in 2-5 months Limited decline in vaccination titers Rapid & robust improvement in clinical disease activity Normalization of serum markers of disease SLE (N=5) SLEDAI-2K Myositis (N=1) Total improvement score
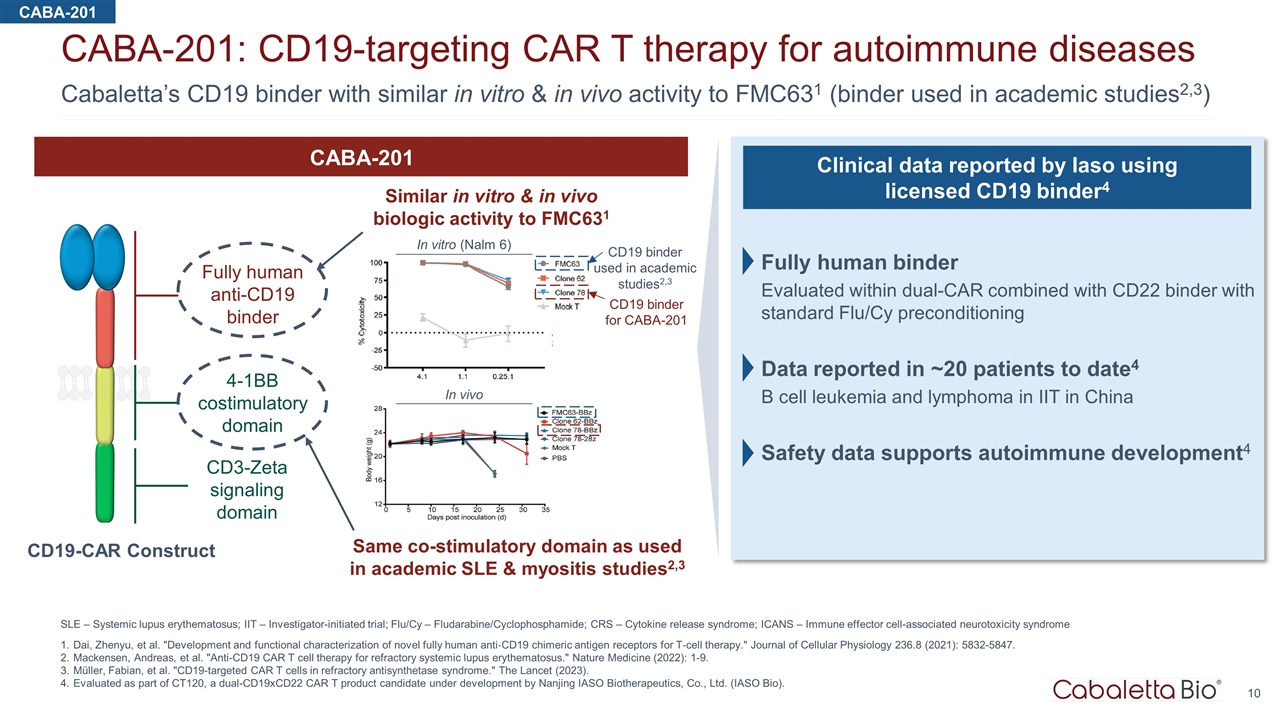
Cabaletta’s CD19 binder with similar in vitro & in vivo activity to FMC631 (binder used in academic studies2,3) CABA-201: CD19-targeting CAR T therapy for autoimmune diseases SLE – Systemic lupus erythematosus; IIT – Investigator-initiated trial; Flu/Cy – Fludarabine/Cyclophosphamide; CRS – Cytokine release syndrome; ICANS – Immune effector cell-associated neurotoxicity syndrome Dai, Zhenyu, et al. "Development and functional characterization of novel fully human anti‐CD19 chimeric antigen receptors for T‐cell therapy." Journal of Cellular Physiology 236.8 (2021): 5832-5847. Mackensen, Andreas, et al. "Anti-CD19 CAR T cell therapy for refractory systemic lupus erythematosus." Nature Medicine (2022): 1-9. Müller, Fabian, et al. "CD19-targeted CAR T cells in refractory antisynthetase syndrome." The Lancet (2023). Evaluated as part of CT120, a dual-CD19xCD22 CAR T product candidate under development by Nanjing IASO Biotherapeutics, Co., Ltd. (IASO Bio). CD19 binder for CABA-201 CD19 binder used in academic studies2,3 In vitro (Nalm 6) In vivo Fully human anti-CD19 binder 4-1BB costimulatory domain CD3-Zeta signaling domain CABA-201 Similar in vitro & in vivo biologic activity to FMC631 Same co-stimulatory domain as used in academic SLE & myositis studies2,3 CD19-CAR Construct Clinical data reported by Iaso using licensed CD19 binder4 Fully human binder Evaluated within dual-CAR combined with CD22 binder with standard Flu/Cy preconditioning Data reported in ~20 patients to date4 B cell leukemia and lymphoma in IIT in China Safety data supports autoimmune development4 CABA-201
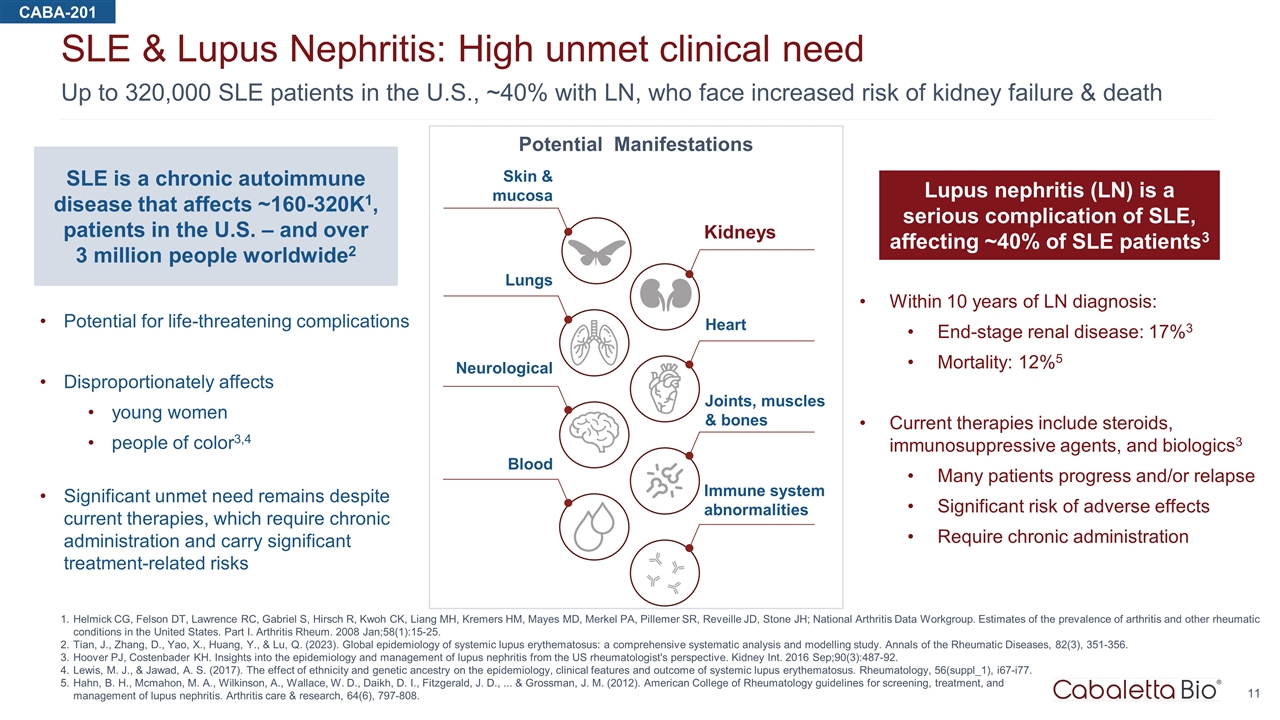
Up to 320,000 SLE patients in the U.S., ~40% with LN, who face increased risk of kidney failure & death SLE & Lupus Nephritis: High unmet clinical need Helmick CG, Felson DT, Lawrence RC, Gabriel S, Hirsch R, Kwoh CK, Liang MH, Kremers HM, Mayes MD, Merkel PA, Pillemer SR, Reveille JD, Stone JH; National Arthritis Data Workgroup. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part I. Arthritis Rheum. 2008 Jan;58(1):15-25. Tian, J., Zhang, D., Yao, X., Huang, Y., & Lu, Q. (2023). Global epidemiology of systemic lupus erythematosus: a comprehensive systematic analysis and modelling study. Annals of the Rheumatic Diseases, 82(3), 351-356. Hoover PJ, Costenbader KH. Insights into the epidemiology and management of lupus nephritis from the US rheumatologist's perspective. Kidney Int. 2016 Sep;90(3):487-92. Lewis, M. J., & Jawad, A. S. (2017). The effect of ethnicity and genetic ancestry on the epidemiology, clinical features and outcome of systemic lupus erythematosus. Rheumatology, 56(suppl_1), i67-i77. Hahn, B. H., Mcmahon, M. A., Wilkinson, A., Wallace, W. D., Daikh, D. I., Fitzgerald, J. D., ... & Grossman, J. M. (2012). American College of Rheumatology guidelines for screening, treatment, and management of lupus nephritis. Arthritis care & research, 64(6), 797-808. CABA-201 Within 10 years of LN diagnosis: End-stage renal disease: 17%3 Mortality: 12%5 Current therapies include steroids, immunosuppressive agents, and biologics3 Many patients progress and/or relapse Significant risk of adverse effects Require chronic administration SLE is a chronic autoimmune disease that affects ~160-320K1, patients in the U.S. – and over 3 million people worldwide2 Potential for life-threatening complications Disproportionately affects young women people of color3,4 Significant unmet need remains despite current therapies, which require chronic administration and carry significant treatment-related risks Lupus nephritis (LN) is a serious complication of SLE, affecting ~40% of SLE patients3 Skin & mucosa Kidneys Heart Joints, muscles & bones Immune system abnormalities Blood Neurological Lungs Potential Manifestations
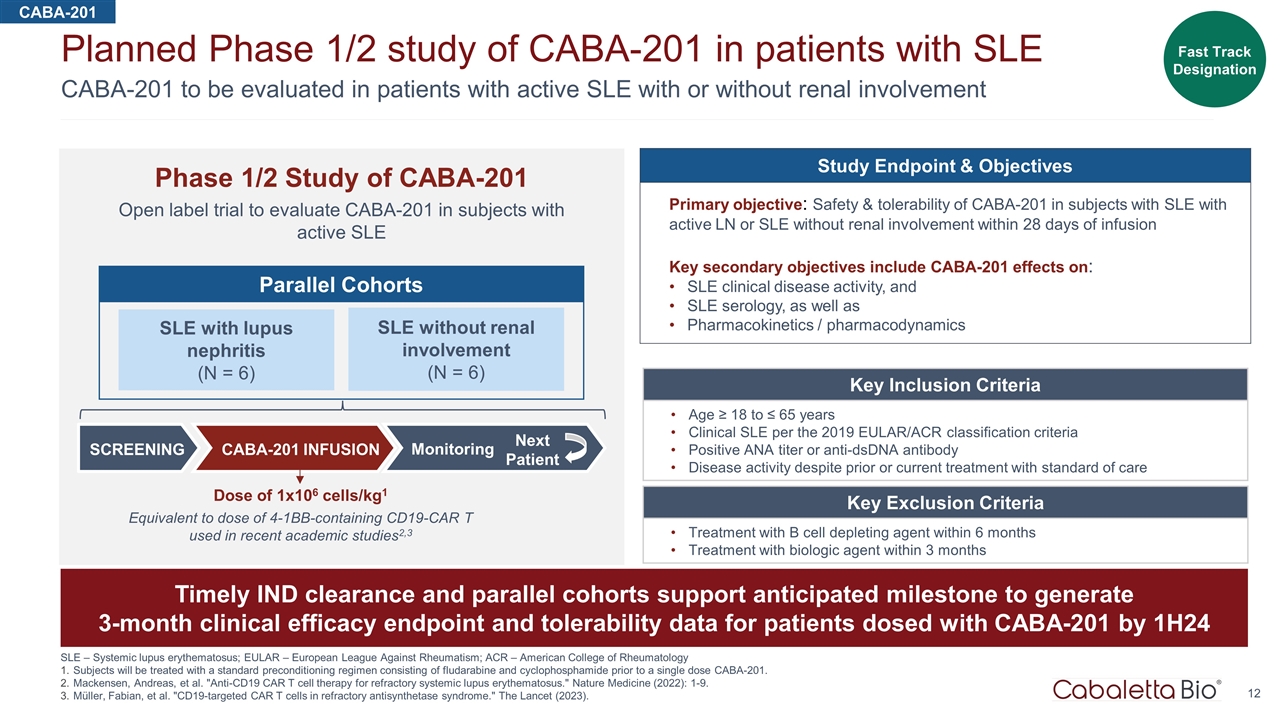
CABA-201 to be evaluated in patients with active SLE with or without renal involvement Planned Phase 1/2 study of CABA-201 in patients with SLE SLE – Systemic lupus erythematosus; EULAR – European League Against Rheumatism; ACR – American College of Rheumatology Subjects will be treated with a standard preconditioning regimen consisting of fludarabine and cyclophosphamide prior to a single dose CABA-201. Mackensen, Andreas, et al. "Anti-CD19 CAR T cell therapy for refractory systemic lupus erythematosus." Nature Medicine (2022): 1-9. Müller, Fabian, et al. "CD19-targeted CAR T cells in refractory antisynthetase syndrome." The Lancet (2023). Phase 1/2 Study of CABA-201 Open label trial to evaluate CABA-201 in subjects with active SLE CABA-201 Key Inclusion Criteria Age ≥ 18 to ≤ 65 years Clinical SLE per the 2019 EULAR/ACR classification criteria Positive ANA titer or anti-dsDNA antibody Disease activity despite prior or current treatment with standard of care Key Exclusion Criteria Treatment with B cell depleting agent within 6 months Treatment with biologic agent within 3 months Timely IND clearance and parallel cohorts support anticipated milestone to generate 3-month clinical efficacy endpoint and tolerability data for patients dosed with CABA-201 by 1H24 Fast Track Designation SLE with lupus nephritis (N = 6) SLE without renal involvement (N = 6) Parallel Cohorts SCREENING CABA-201 INFUSION Monitoring Next Patient Study Endpoint & Objectives Primary objective: Safety & tolerability of CABA-201 in subjects with SLE with active LN or SLE without renal involvement within 28 days of infusion Key secondary objectives include CABA-201 effects on: SLE clinical disease activity, and SLE serology, as well as Pharmacokinetics / pharmacodynamics Dose of 1x106 cells/kg1 Equivalent to dose of 4-1BB-containing CD19-CAR T used in recent academic studies2,3

Our product candidate, people and partnership enable accelerated progress while integrating unique insights Accelerating development of CABA-201 for autoimmune diseases SLE – Systemic lupus erythematosus Dai, Zhenyu, et al. "Development and functional characterization of novel fully human anti‐CD19 chimeric antigen receptors for T‐cell therapy." Journal of Cellular Physiology 236.8 (2021): 5832-5847. Mackensen, Andreas, et al. "Anti-CD19 CAR T cell therapy for refractory systemic lupus erythematosus." Nature Medicine (2022): 1-9. Müller, Fabian, et al. "CD19-targeted CAR T cells in refractory antisynthetase syndrome." The Lancet (2023). 1 2 Partnership – Exclusive translational research partnership has provided early, actionable insights Scientific and clinical data sharing has already impacted our clinical strategy and design CABA-201 3 Efficient clinical trial design for CABA-201 facilitates rapid and broad development program Product – Candidate with 4-1BB co-stim domain; similar binding activity to academic CD19-CAR T1,2,3 CABA-201 fully human binder clinical tolerability profile based on use in ~20 oncology patients 4-1BB co-stimulatory domain identical to that used in academic CD19-CAR T study1,2 People – Singular focus on potentially curative cell therapies for autoimmune disease since 2018 Deep understanding and experience with complicated cell therapy programs in autoimmune patients Novel insights on clinical designs from prior FDA discussions and 3 timely submitted and cleared IND filings Track record at a dozen US sites with implementation of complicated cell therapy logistics in autoimmune patients Leadership with experience developing both SLE products that were FDA approved in the past 65 years

Chimeric AutoAntibody Receptor T Cells DSG3-CAART & MuSK-CAART
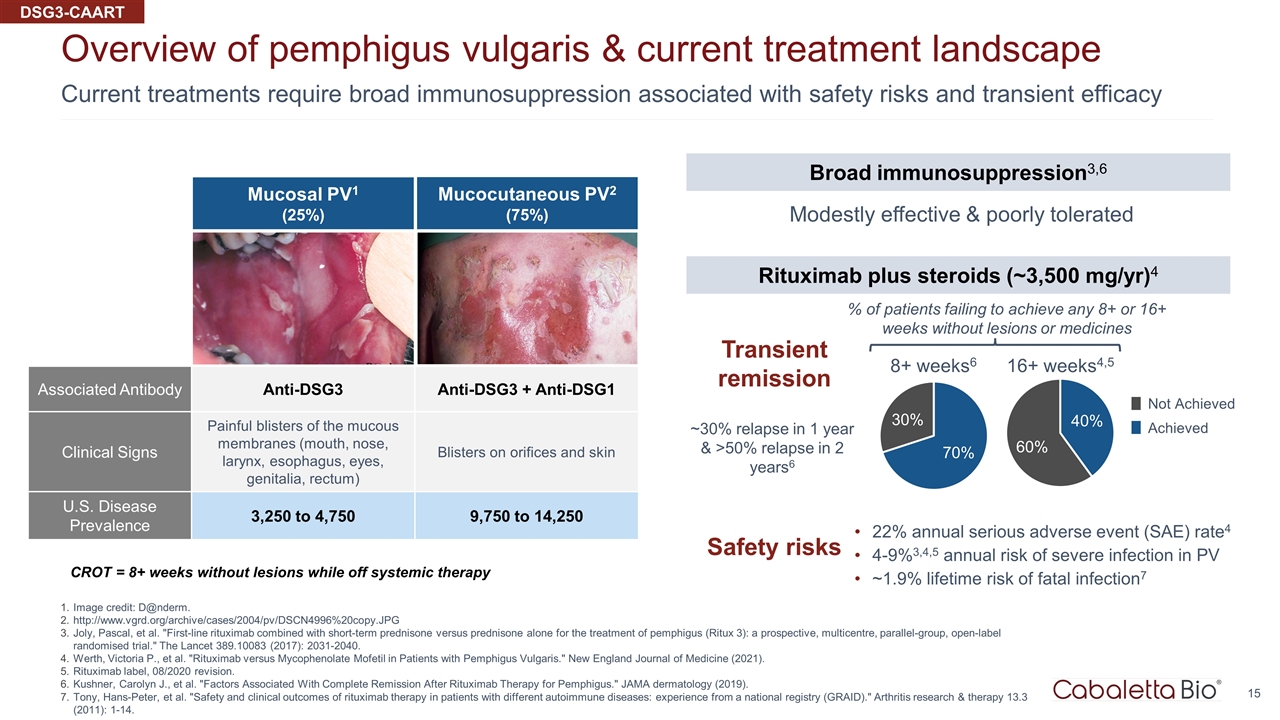
Current treatments require broad immunosuppression associated with safety risks and transient efficacy Overview of pemphigus vulgaris & current treatment landscape Image credit: D@nderm. http://www.vgrd.org/archive/cases/2004/pv/DSCN4996%20copy.JPG Joly, Pascal, et al. "First-line rituximab combined with short-term prednisone versus prednisone alone for the treatment of pemphigus (Ritux 3): a prospective, multicentre, parallel-group, open-label randomised trial." The Lancet 389.10083 (2017): 2031-2040. Werth, Victoria P., et al. "Rituximab versus Mycophenolate Mofetil in Patients with Pemphigus Vulgaris." New England Journal of Medicine (2021). Rituximab label, 08/2020 revision. Kushner, Carolyn J., et al. "Factors Associated With Complete Remission After Rituximab Therapy for Pemphigus." JAMA dermatology (2019). Tony, Hans-Peter, et al. "Safety and clinical outcomes of rituximab therapy in patients with different autoimmune diseases: experience from a national registry (GRAID)." Arthritis research & therapy 13.3 (2011): 1-14. CROT = 8+ weeks without lesions while off systemic therapy Mucosal PV1 (25%) Associated Antibody Anti-DSG3 Anti-DSG3 + Anti-DSG1 Clinical Signs Painful blisters of the mucous membranes (mouth, nose, larynx, esophagus, eyes, genitalia, rectum) Blisters on orifices and skin U.S. Disease Prevalence 3,250 to 4,750 9,750 to 14,250 Mucocutaneous PV2 (75%) DSG3-CAART Transient remission Broad immunosuppression3,6 Rituximab plus steroids (~3,500 mg/yr)4 Modestly effective & poorly tolerated 8+ weeks6 16+ weeks4,5 % of patients failing to achieve any 8+ or 16+ weeks without lesions or medicines Not Achieved Achieved ~30% relapse in 1 year & >50% relapse in 2 years6 Safety risks 22% annual serious adverse event (SAE) rate4 4-9%3,4,5 annual risk of severe infection in PV ~1.9% lifetime risk of fatal infection7
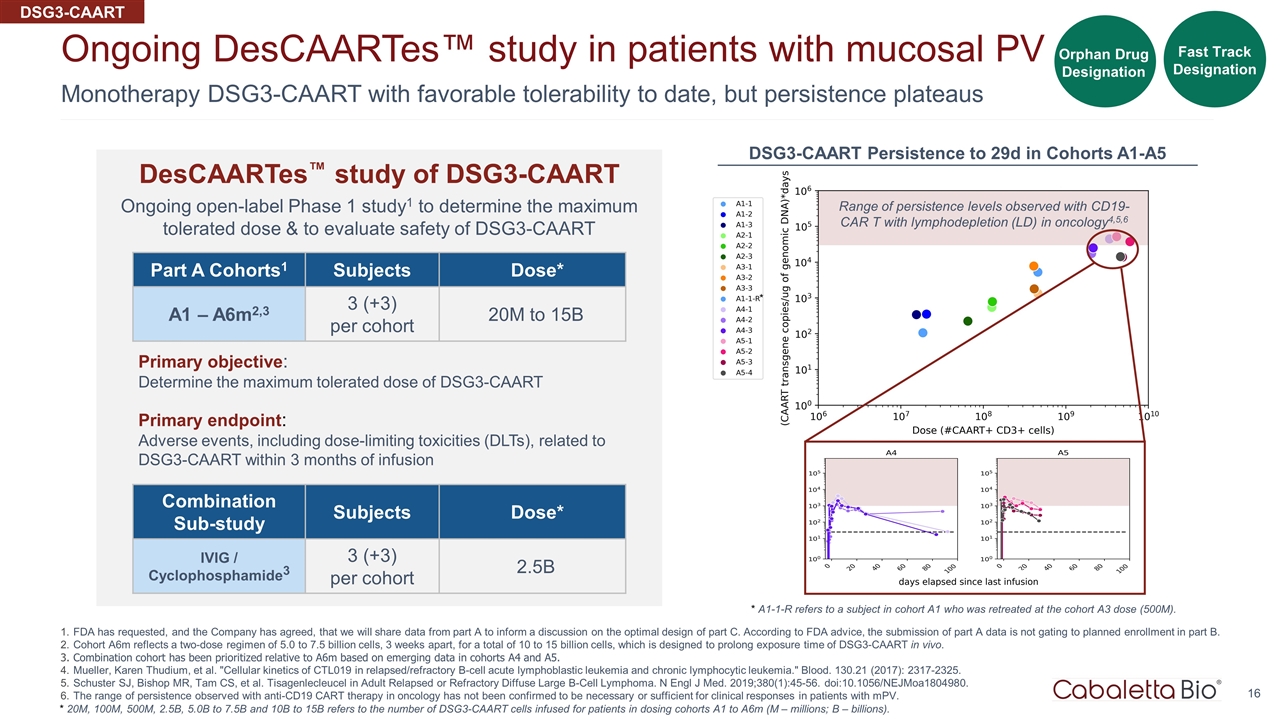
Monotherapy DSG3-CAART with favorable tolerability to date, but persistence plateaus Ongoing DesCAARTes™ study in patients with mucosal PV FDA has requested, and the Company has agreed, that we will share data from part A to inform a discussion on the optimal design of part C. According to FDA advice, the submission of part A data is not gating to planned enrollment in part B. Cohort A6m reflects a two-dose regimen of 5.0 to 7.5 billion cells, 3 weeks apart, for a total of 10 to 15 billion cells, which is designed to prolong exposure time of DSG3-CAART in vivo. Combination cohort has been prioritized relative to A6m based on emerging data in cohorts A4 and A5. Mueller, Karen Thudium, et al. "Cellular kinetics of CTL019 in relapsed/refractory B-cell acute lymphoblastic leukemia and chronic lymphocytic leukemia." Blood. 130.21 (2017): 2317-2325. Schuster SJ, Bishop MR, Tam CS, et al. Tisagenlecleucel in Adult Relapsed or Refractory Diffuse Large B-Cell Lymphoma. N Engl J Med. 2019;380(1):45-56. doi:10.1056/NEJMoa1804980. The range of persistence observed with anti-CD19 CART therapy in oncology has not been confirmed to be necessary or sufficient for clinical responses in patients with mPV. Part A Cohorts1 Subjects Dose* A1 – A6m2,3 3 (+3) per cohort 20M to 15B Primary objective: Determine the maximum tolerated dose of DSG3-CAART Primary endpoint: Adverse events, including dose-limiting toxicities (DLTs), related to DSG3-CAART within 3 months of infusion * 20M, 100M, 500M, 2.5B, 5.0B to 7.5B and 10B to 15B refers to the number of DSG3-CAART cells infused for patients in dosing cohorts A1 to A6m (M – millions; B – billions). Combination Sub-study Subjects Dose* IVIG / Cyclophosphamide3 3 (+3) per cohort 2.5B DSG3-CAART DesCAARTes™ study of DSG3-CAART Ongoing open-label Phase 1 study1 to determine the maximum tolerated dose & to evaluate safety of DSG3-CAART Range of persistence levels observed with CD19-CAR T with lymphodepletion (LD) in oncology4,5,6 * DSG3-CAART Persistence to 29d in Cohorts A1-A5 * A1-1-R refers to a subject in cohort A1 who was retreated at the cohort A3 dose (500M). Fast Track Designation Orphan Drug Designation
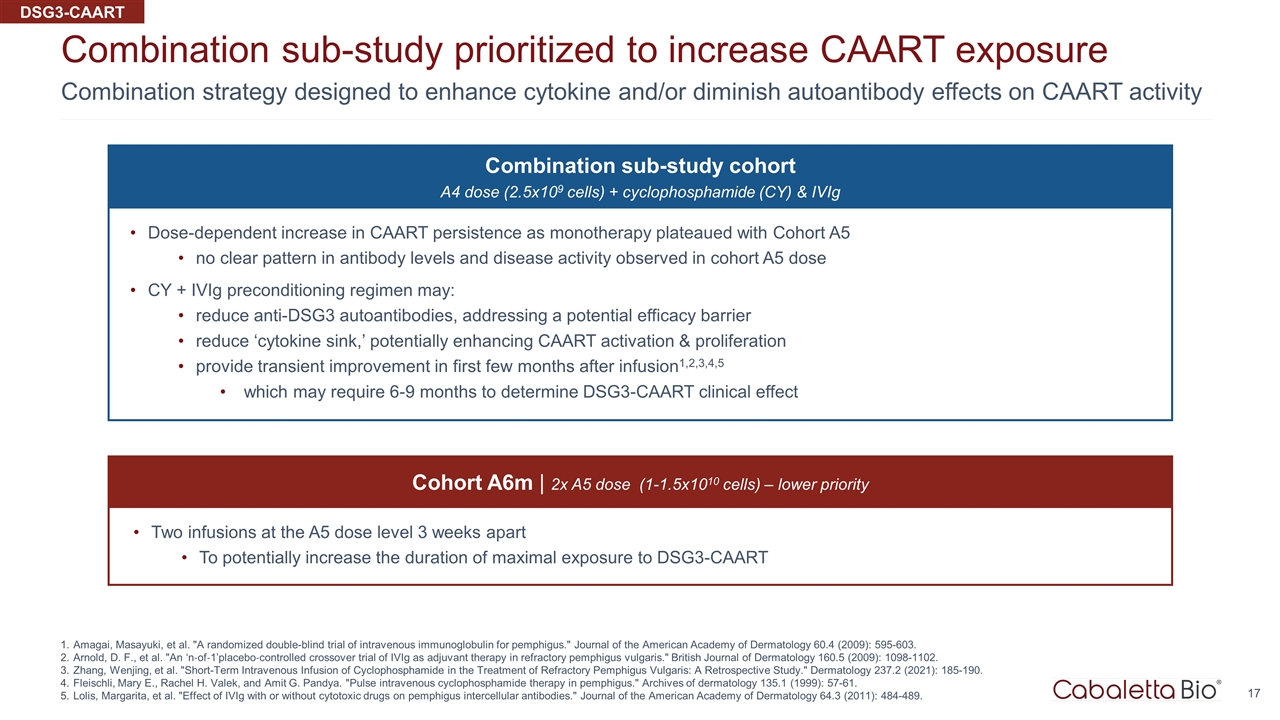
Combination strategy designed to enhance cytokine and/or diminish autoantibody effects on CAART activity Combination sub-study prioritized to increase CAART exposure Amagai, Masayuki, et al. "A randomized double-blind trial of intravenous immunoglobulin for pemphigus." Journal of the American Academy of Dermatology 60.4 (2009): 595-603. Arnold, D. F., et al. "An ‘n‐of‐1’placebo‐controlled crossover trial of IVIg as adjuvant therapy in refractory pemphigus vulgaris." British Journal of Dermatology 160.5 (2009): 1098-1102. Zhang, Wenjing, et al. "Short-Term Intravenous Infusion of Cyclophosphamide in the Treatment of Refractory Pemphigus Vulgaris: A Retrospective Study." Dermatology 237.2 (2021): 185-190. Fleischli, Mary E., Rachel H. Valek, and Amit G. Pandya. "Pulse intravenous cyclophosphamide therapy in pemphigus." Archives of dermatology 135.1 (1999): 57-61. Lolis, Margarita, et al. "Effect of IVIg with or without cytotoxic drugs on pemphigus intercellular antibodies." Journal of the American Academy of Dermatology 64.3 (2011): 484-489. Combination sub-study cohort A4 dose (2.5x109 cells) + cyclophosphamide (CY) & IVIg Dose-dependent increase in CAART persistence as monotherapy plateaued with Cohort A5 no clear pattern in antibody levels and disease activity observed in cohort A5 dose CY + IVIg preconditioning regimen may: reduce anti-DSG3 autoantibodies, addressing a potential efficacy barrier reduce ‘cytokine sink,’ potentially enhancing CAART activation & proliferation provide transient improvement in first few months after infusion1,2,3,4,5 which may require 6-9 months to determine DSG3-CAART clinical effect Cohort A6m | 2x A5 dose (1-1.5x1010 cells) – lower priority Two infusions at the A5 dose level 3 weeks apart To potentially increase the duration of maximal exposure to DSG3-CAART DSG3-CAART

Strategy for first-in-human trial informed by learnings from DesCAARTes™ study High unmet need in MuSK myasthenia gravis MusCAARTes™ study of MuSK-CAART Ongoing open-label study to determine the maximum tolerated dose & to evaluate safety of MuSK-CAART Part Cohort Subjects A – Monotherapy Dose Escalation7 Dose increasing from 500M with 2 (+4) design A1-A3 2 (+4) per cohort A – Adaptive Combination Cohorts7 Combination cohorts8, starting at A2 dose A4+ 2 (+4) per cohort B – Expansion Expanded subject enrollment at final selected dose B ~12 MuSK-CAART IgG4-dominant disease Autoantibody titers drop after rituximab1,2 Pathogenic B cells incompletely eliminated by rituximab and persist during relapse3 Circulating anti-MuSK antibody titers may be ~90% lower than anti-DSG3 antibody in PV4,5,6 1 2 Study Endpoint & Objectives Primary Endpoint: Adverse Events, including DLT Secondary & Tertiary Objectives: CAART expansion/persistence, effect on serum anti-MuSK antibody titer, use of concomitant systemic medications, effect on clinical symptoms, manufacturing success rate Compelling biologic rationale, similar to PV Differentiated market opportunity Total U.S. MG prevalence 50,000 to 80,000 patients MuSK+ MG comprises 6-7.5% of total MG population Compared to AChR+ MG, MuSK+ disease is typically more severe with limited treatment options MuSK+ disease has early onset, 7:1 females * 500M refers to the number of MuSK-CAART cells infused for patients in dosing cohort A1 (M – millions). Hain, Berit, et al. "Successful treatment of MuSK antibody–positive MG with rituximab." Muscle & Nerve. 33.4 (2006): 575-580. Illa, Isabel, et al. "Sustained response to Rituximab in anti-AChR and anti-MuSK positive Myasthenia Gravis patients." Journal of neuroimmunology 201 (2008): 90-94. Jiang, Ruoyi, et al. "Single-cell repertoire tracing identifies rituximab-resistant B cells during myasthenia gravis relapses." JCI insight 5.14 (2020). Matthews, Ian, et al. "Muscle-specific receptor tyrosine kinase autoantibodies—a new immunoprecipitation assay." Clinica chimica acta 348.1-2 (2004): 95-99. McConville, John, et al. "Detection and characterization of MuSK antibodies in seronegative myasthenia gravis." Annals of neurology 55.4 (2004): 580-584. Marino, Mariapaola, et al. "Long-lasting rituximab-induced reduction of specific—but not total—IgG4 in MuSK-positive myasthenia gravis." Frontiers in immunology 11 (2020): 613. A total of 6 subjects will need to have received the final selected dose in Part A of the study. Cohorts A3 and A4 will be enrolled concurrently after cohort A2 is well-tolerated with a preference for enrollment into A4. Fast Track Designation Orphan Drug Designation

Corporate Summary
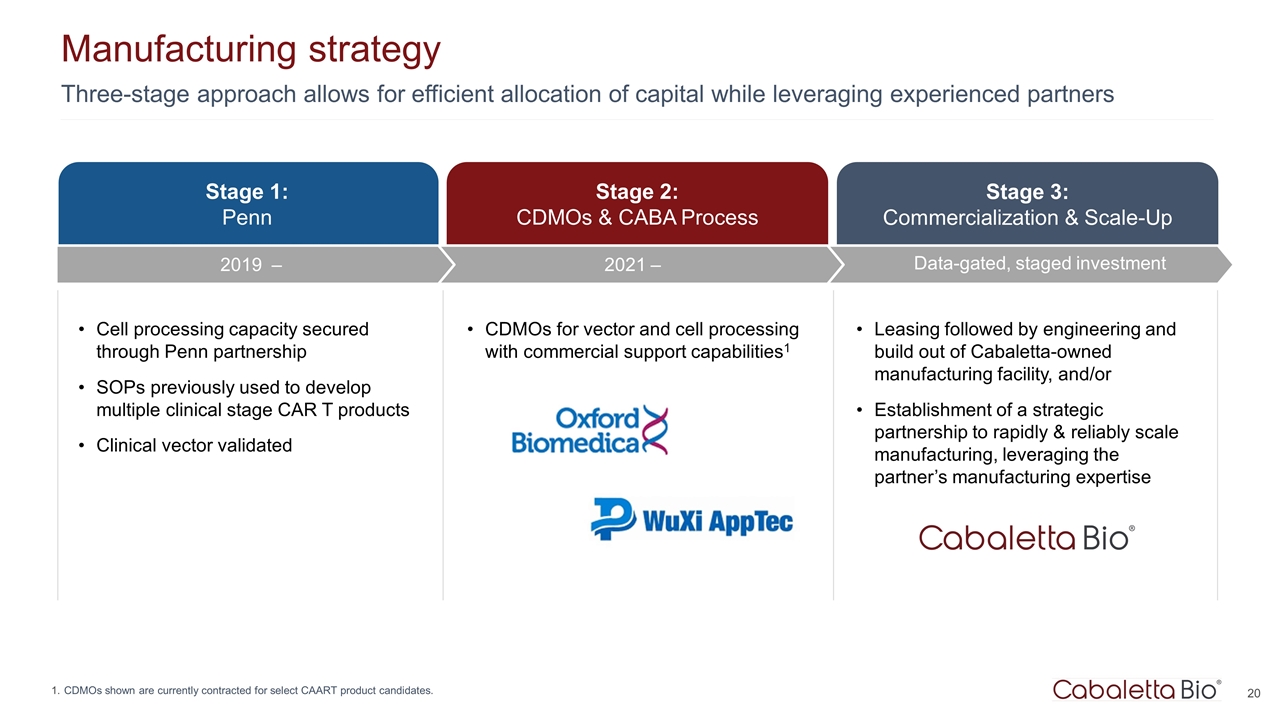
Three-stage approach allows for efficient allocation of capital while leveraging experienced partners Manufacturing strategy CDMOs shown are currently contracted for select CAART product candidates. Stage 3: Commercialization & Scale-Up Data-gated, staged investment Stage 1: Penn Stage 2: CDMOs & CABA Process Cell processing capacity secured through Penn partnership SOPs previously used to develop multiple clinical stage CAR T products Clinical vector validated CDMOs for vector and cell processing with commercial support capabilities1 Leasing followed by engineering and build out of Cabaletta-owned manufacturing facility, and/or Establishment of a strategic partnership to rapidly & reliably scale manufacturing, leveraging the partner’s manufacturing expertise 2021 – 2019 –

BOARD OF DIRECTORS Cabaletta Bio leadership LEADERSHIP TEAM Anup Marda Chief Financial Officer Arun Das, M.D. Chief Business Officer David J. Chang, M.D., M.P.H., FACR Chief Medical Officer Martha O’Connor Chief HR Officer Michael Gerard General Counsel Steven Nichtberger, M.D. President, CEO & Chairman Heather Harte-Hall Chief Compliance Officer Samik Basu, M.D. Chief Scientific Officer Gwendolyn Binder, Ph.D. President, Science & Technology SCIENTIFIC ADVISORY BOARD Track record of operational success employing novel cell therapies in autoimmunity across preclinical, clinical, manufacturing & regulatory domains Aimee Payne, M.D., Ph.D. Co-Founder and Co-Chair Michael C. Milone, M.D., Ph.D. Co-Founder and Co-Chair Carl June, M.D. Jay Siegel, M.D. Brian Daniels, M.D. Steven Nichtberger, M.D. Richard Henriques, M.B.A. Scott Brun, M.D. Mark Simon, M.B.A. Catherine Bollard, M.D. Drew Weissman, M.D., Ph.D. Iain McInnes, Ph.D., FRCP, FRSE, FMedSci Georg Schett, M.D.
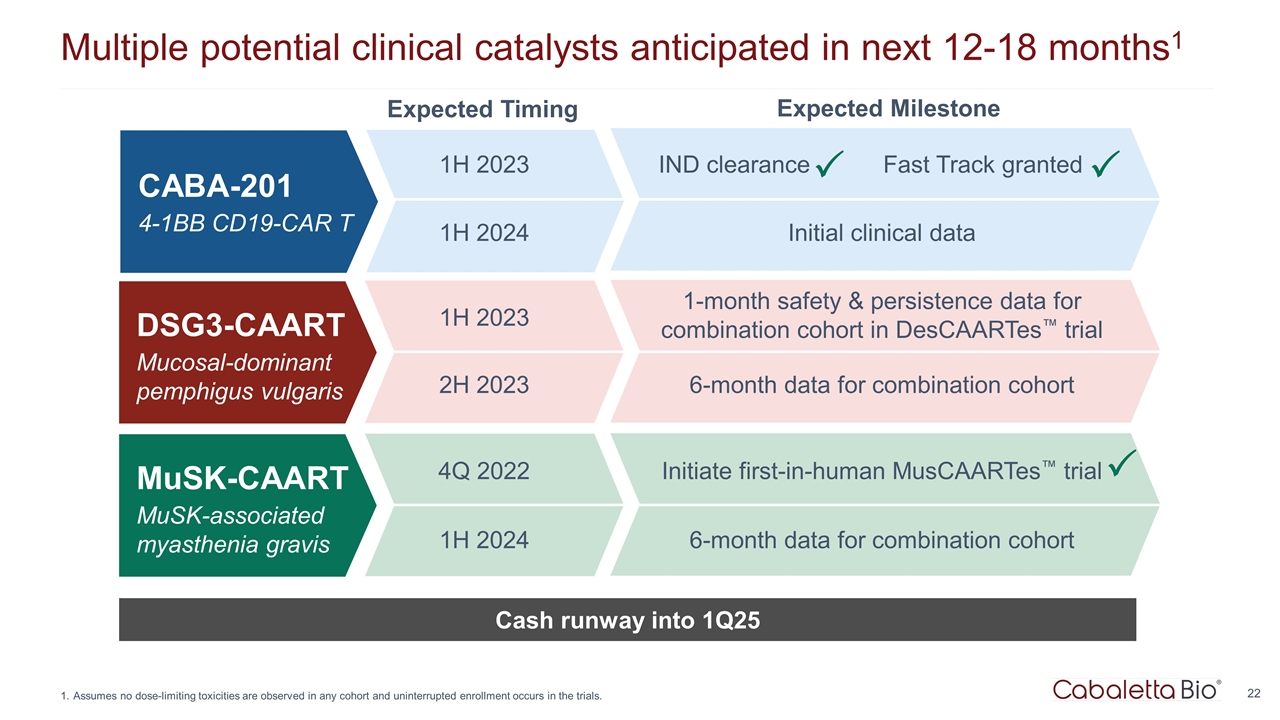
Multiple potential clinical catalysts anticipated in next 12-18 months1 Assumes no dose-limiting toxicities are observed in any cohort and uninterrupted enrollment occurs in the trials. Cash runway into 1Q25 CABA-201 4-1BB CD19-CAR T Expected Timing Expected Milestone 1H 2023 1H 2024 Initial clinical data DSG3-CAART Mucosal-dominant pemphigus vulgaris 1H 2023 1-month safety & persistence data for combination cohort in DesCAARTes™ trial 2H 2023 6-month data for combination cohort MuSK-CAART MuSK-associated myasthenia gravis 4Q 2022 Initiate first-in-human MusCAARTes™ trial 1H 2024 6-month data for combination cohort P IND clearance P Fast Track granted P

Corporate Presentation MAY 2023
Exhibit 99.2

Cabaletta Bio Receives FDA Fast Track Designation for CABA-201
PHILADELPHIA, May 1, 2023 — Cabaletta Bio, Inc. (Nasdaq: CABA), a clinical-stage biotechnology company focused on developing and launching the first curative targeted cell therapies for patients with autoimmune diseases, today announced that the U.S. Food and Drug Administration (FDA) has granted Fast Track Designation for CABA-201, a 4-1BB-containing fully human CD19-CAR T cell investigational therapy, designed to deplete CD19-positive B cells and improve disease activity in patients with systemic lupus erythematosus (SLE) and lupus nephritis (LN). Cabaletta has been cleared to initiate a Phase 1/2 clinical trial of CABA-201 for the treatment of 6 SLE patients with active LN, and in a separate parallel cohort, 6 patients with active SLE without renal involvement, with an initial dose that is equivalent to the dose used in the September 2022 Nature Medicine publication of a 4-1BB containing CD19-CAR T construct evaluated in patients with SLE.
“Despite advances over the last few decades, treatment options for SLE remain inadequate. There are currently no curative options available that achieve durable disease remission. Existing therapies typically result in general immunosuppression, require chronic administration, and are often administered in conjunction with steroids and other immunosuppressive medications to reduce disease burden, which can leave patients with continued disease activity, treatment-associated side effects, and impaired quality of life,” said David J. Chang, M.D., Chief Medical Officer of Cabaletta. “We believe the FDA’s decision to grant Fast Track Designation for CABA-201 underscores the unmet need for a treatment that has the potential to provide deep and durable responses for people living with lupus and potentially other autoimmune diseases where B cells contribute to disease. We look forward to initiating the Phase 1/2 trial for CABA-201 and further evaluating its therapeutic potential for patients in need.”
CABA-201 is designed to be given as a one-time infusion, to evaluate its potential to transiently, but fully, eliminate B cells, enabling an “immune system reset” with durable remission in patients with SLE. The Phase 1/2 clinical trial is an open-label study designed to evaluate CABA-201 in SLE subjects with active LN or active SLE without renal involvement. CABA-201 will be administered at a dose of 1.0 x 106 cells/kg and the study will enroll 6 subjects in an active LN cohort and 6 subjects in an active SLE without renal involvement cohort, in parallel. Subjects will be treated with a standard preconditioning regimen consisting of fludarabine and cyclophosphamide prior to CABA-201 infusion. This represents the first trial assessing Cabaletta’s CARTA (Chimeric Antigen Receptor T cells for Autoimmunity) strategy.
About Fast Track Designation
The FDA’s Fast Track process is intended to facilitate the expedited development and review of therapeutics intended to treat serious or life-threatening conditions and to address unmet medical needs. Companies that receive Fast Track Designation are eligible for several potential benefits, including the opportunity for more frequent meetings and interactions with the FDA during clinical development as well as eligibility for accelerated approval and/or priority review, if relevant criteria are met. Companies may also be allowed to submit sections of their Biologics License Application on a rolling basis.
About Systemic Lupus Erythematosus
SLE is a chronic, potentially severe, autoimmune disease in which the immune system attacks healthy tissue throughout the body, most commonly impacting young women between the ages of 15 and 40 with higher frequency and greater severity in people of color. It is characterized by abnormal B cell function and autoantibody production resulting in a range of clinical manifestations including end organ damage and an increased risk of death. SLE affects an estimated 160,000-320,000 patients in the U.S, with LN as the most common end-organ manifestation, affecting approximately 40% of SLE patients. Among these patients, the risk of end-stage renal disease is approximately 17% and the risk of death is approximately 12%, each within 10 years of diagnosis.
About Cabaletta Bio
Cabaletta Bio (Nasdaq: CABA) is a clinical-stage biotechnology company focused on the discovery and development of engineered T cell therapies that have the potential to provide a deep and durable, perhaps curative, treatment for patients with autoimmune diseases. The CABA™ platform encompasses two strategies: the CARTA (Chimeric Antigen Receptor T cells for Autoimmunity) strategy, with CABA-201, a 4-1BB-containing fully human CD19-CAR T, as the lead product candidate being evaluated in lupus nephritis and systemic lupus erythematosus without renal involvement, and the CAART (Chimeric AutoAntibody Receptor T cells) strategy, with multiple clinical-stage candidates, including DSG3-CAART for mucosal pemphigus vulgaris and MuSK-CAART for MuSK myasthenia gravis. The expanding CABA™ platform is designed to develop potentially curative therapies for patients with a broad range of autoimmune diseases. Cabaletta Bio’s headquarters and labs are located in Philadelphia, PA.
Forward-Looking Statements
This press release contains “forward-looking statements” of Cabaletta Bio within the meaning of the Private Securities Litigation Reform Act of 1995, as amended, including without limitation, express or implied statements regarding: Cabaletta’s ability to retain and recognize the intended incentives conferred Fast Track Designation for CABA-201 to deplete CD19-positive B cells and improve disease activity in patients with SLE and LN; Cabaletta’s ability to grow its autoimmune-focused pipeline; the Company’s business plans and objectives; Cabaletta Bio’s expectations around the potential success and therapeutic benefits of CABA-201, including its belief that CABA-201 may enable an “immune system reset” and provide deep and durable responses for patients with SLE and potentially for patients diagnosed with other autoimmune disease; the Company’s plans to initiate a Phase 1/2 clinical trial of CABA-201 in patients with SLE, including its anticipated progress, clinical trial design, ability to leverage its experience in autoimmune cell therapy and lupus product development; Cabaletta’s ability to enroll the requisite number of patients, dose each dosing cohort in the intended manner and advance the trial as planned in its Phase 1/2 clinical trial of CABA-201; and the ability to accelerate Cabaletta’s pipeline and develop meaningful therapies for patients, including in collaboration with academic and industry partners and the ability to optimize such collaborations on its development programs.
Any forward-looking statements in this press release are based on management’s current expectations and beliefs of future events, and are subject to a number of risks and uncertainties that could cause actual results to differ materially and adversely from those set forth in or implied by such forward-looking statements. These risks and uncertainties include, but are not limited to: the risk that signs of biologic activity or persistence may not inform long-term results; Cabaletta’s ability to demonstrate sufficient evidence of safety, efficacy and tolerability in its preclinical studies and clinical trials of DSG3-CAART, MuSK-CAART and CABA-201; the risk that the results observed with the similarly-designed construct employed in the recent Nature
Medicine publication, including due to the dosing regimen, are not indicative of the results we seek to achieve with CABA-201; risks related to clinical trial site activation or enrollment rates that are lower than expected; risks related to unexpected safety or efficacy data observed during clinical studies; risks related to volatile market and economic conditions; risks related to the impact of public health epidemics affecting countries or regions in which Cabaletta has operations or does business, such as COVID-19; Cabaletta’s ability to retain and recognize the intended incentives conferred by Orphan Drug Designation and Fast Track Designation for its product candidates, as applicable; risks related to Cabaletta’s ability to protect and maintain its intellectual property position; risks related to fostering and maintaining successful relationships with Cabaletta’s collaboration and manufacturing partners; uncertainties related to the initiation and conduct of studies and other development requirements for its product candidates; the risk that any one or more of Cabaletta’s product candidates will not be successfully developed and/or commercialized; and the risk that the initial or interim results of preclinical studies or clinical studies will not be predictive of future results in connection with future studies. For a discussion of these and other risks and uncertainties, and other important factors, any of which could cause Cabaletta’s actual results to differ from those contained in the forward-looking statements, see the section entitled “Risk Factors” in Cabaletta’s most recent annual report on Form 10-K as well as discussions of potential risks, uncertainties, and other important factors in Cabaletta’s other and subsequent filings with the Securities and Exchange Commission. All information in this press release is as of the date of the release, and Cabaletta undertakes no duty to update this information unless required by law.
Contacts:
Anup Marda
Chief Financial Officer
investors@cabalettabio.com
Sarah McCabe
Stern Investor Relations, Inc.
sarah.mccabe@sternir.com