Anixa Biosciences, Inc.
3150 Almaden Expressway
Suite 250
San Jose, CA 95118
408.708.9808
NASDAQ: ANIX
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM
CURRENT REPORT
Pursuant to Section 13 or Section 15(d) of the Securities Exchange Act of 1934
Date
of Report (Date of earliest event reported):
(Exact name of registrant as specified in its charter)
| (State
or other jurisdiction of incorporation) |
(Commission File Number) |
(IRS Employer Identification No.) |
| (Address of principal executive offices) | (Zip Code) |
Registrant’s
telephone number, including area code:
(Former name or former address, if changed since last report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation to the registrant under any of the following provisions:
| Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) | |
| Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) | |
| Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) | |
| Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging
growth company
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
| Item 7.01. | Regulation FD Disclosure. |
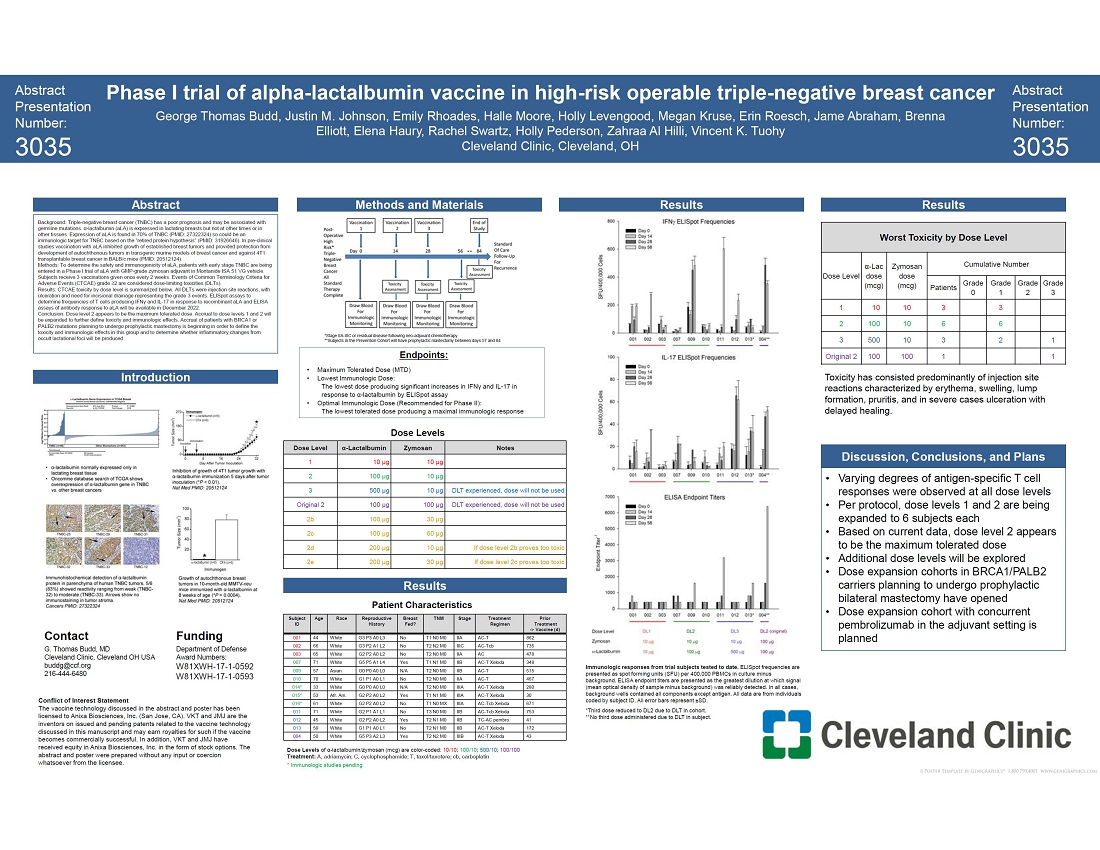
On April 17, 2023, Anixa Biosciences, Inc. (“we,” “us,” “our,” or the “Company”) issued a press release announcing that the Company and The Cleveland Clinic Foundation (“Cleveland Clinic”) presented positive data for the Phase 1 study of its breast cancer vaccine. The press release, which is furnished as Exhibit 99.1 hereto, was issued following a presentation made by G. Thomas Budd, M.D. of Cleveland Clinic’s Taussig Cancer Institute. Furnished hereto as Exhibit 99.2 is the poster utilized by Dr. Budd for the presentation.
Statements that are not historical fact may be considered forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Forward-looking statements are not statements of historical facts, but rather reflect our current expectations concerning future events and results. We generally use the words “believes,” “expects,” “intends,” “plans,” “anticipates,” “likely,” “will” and similar expressions to identify forward-looking statements. Such forward-looking statements, including those concerning our clinical trials, involve risks, uncertainties and other factors, some of which are beyond our control, which may cause our actual results, performance or achievements, or industry results, to be materially different from any future results, performance, or achievements expressed or implied by such forward-looking statements. These risks, uncertainties and factors include, but are not limited to, those factors set forth in “Item 1A - Risk Factors” and other sections of our most recent Annual Report on Form 10-K as well as in our Quarterly Reports on Form 10-Q and Current Reports on Form 8-K. We undertake no obligation to publicly update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except as required by law. You are cautioned not to unduly rely on such forward-looking statements when evaluating the information presented in this Current Report.
| Item 9.01. | Financial Statements and Exhibits |
(d) Exhibits
The following exhibits are filed with this Current Report on Form 8-K:
| Exhibit No. | Description | |
| 99.1 | Press Release | |
| 99.2 | Presentation | |
| 104 | Cover Page Interactive Data File (embedded within the Inline XBRL document). |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
Dated: April 17, 2023
| ANIXA BIOSCIENCES, INC. | ||
| By: | /s/ Michael J. Catelani | |
| Name: | Michael J. Catelani | |
| Title: | President, Chief Operating Officer and Chief Financial Officer |
Exhibit 99.1
 |
Anixa Biosciences, Inc. 3150 Almaden Expressway Suite 250 San Jose, CA 95118 408.708.9808 NASDAQ: ANIX |
Anixa Biosciences and Cleveland Clinic Present Positive Data for Phase 1 Study of Breast Cancer Vaccine
Immune responses were observed at all dose levels
SAN JOSE, Calif., April 17, 2023 -- Anixa Biosciences, Inc. (NASDAQ: ANIX), a biotechnology company focused on the treatment and prevention of cancer today announced that Cleveland Clinic presented the most up-to-date data from the Phase 1 Trial of its breast cancer vaccine. The data presented showed that in the vaccinated women who have been tested to date, various levels of antigen-specific T cell responses were observed at all dose levels. The presentation was made by G. Thomas Budd, M.D., of Cleveland Clinic’s Taussig Cancer Institute and principal investigator of the study. This breast cancer vaccine technology was invented at Cleveland Clinic, where the trial is being conducted, and Anixa is the exclusive worldwide licensee. The trial is funded by a grant from the U.S. Department of Defense to Cleveland Clinic.
The Phase 1a study is designed to evaluate the safety of the vaccine, identify the Maximum Tolerated Dose (MTD), and monitor the immune response in vaccinated women. All participants in the Phase 1a study are women who have had triple negative breast cancer (TNBC) within the last three years and have been curatively treated having undergone standard of care. At the time of vaccination, these participants are tumor-free, as determined by standard diagnostic techniques, but are at high risk of recurrence.
“We are testing this vaccine to determine if a vaccinated patient’s immune system is trained to destroy cancer cells expressing α-lactalbumin, a protein found on TNBC cancer cells and not on normal cells. To evaluate the vaccination effect, immune mediated biomarkers of T cell activation and antibody production specific against α-lactalbumin are measured. We are heartened by the data, and look forward to additional studies,” stated Dr. Amit Kumar, Chairman and CEO of Anixa Biosciences.
“We are pleased that varying degrees of antigen-specific T cell responses were observed at all dose levels tested to date, however, the Phase 1 trial is not designed to determine whether the responses are sufficient to prevent recurrence or primary tumorogenesis, said Dr. Budd. “We expect successive studies to determine how effective the immune responses are in preventing cancer.”
About Triple-Negative Breast Cancer
One in eight women in the U.S. will be diagnosed with an invasive breast cancer at some point in their lives. Approximately 10-15% of those diagnoses are TNBC, however TNBC accounts for a disproportionately higher percentage of breast cancer deaths and has a higher rate of recurrence. This form of breast cancer is twice as likely to occur in African-American women, and approximately 70% to 80% of the breast tumors that occur in women with mutations in the BRCA1 genes are triple-negative breast cancer.

About Anixa Bioscience’s Breast Cancer Vaccine
Anixa’s breast cancer vaccine takes advantage of endogenously produced proteins that have a function at certain times in life, but then become “retired” and disappear from the body. One such protein is a breast-specific lactation protein, α-lactalbumin, which is no longer found post-lactation in normal, aging tissues, but is present in the majority of triple-negative breast cancers. Activating the immune system against this “retired” protein provides preemptive immune protection against emerging breast tumors that express α-lactalbumin. The vaccine also contains an adjuvant that activates an innate immune response, which allows the immune system to mount a response against emerging tumors to prevent them from growing. This vaccine technology was invented by the late Dr. Vincent Tuohy, who was the Mort and Iris November Distinguished Chair in Innovative Breast Cancer Research in the Department of Inflammation and Immunity at Cleveland Clinic’s Lerner Research Institute. Dr. Tuohy was inventor of the technology, which Cleveland Clinic exclusively licensed to Anixa Biosciences. He was entitled to a portion of the commercialization revenues received by Cleveland Clinic and also held equity in Anixa.
About Anixa Biosciences, Inc.
Anixa is a clinical-stage biotechnology company focused on the treatment and prevention of cancer. Anixa’s therapeutic portfolio consists of an ovarian cancer immunotherapy program being developed in collaboration with Moffitt Cancer Center, which uses a novel type of CAR-T, known as chimeric endocrine receptor T-cell (CER-T) technology. The company’s vaccine portfolio includes a novel vaccine being developed in collaboration with Cleveland Clinic to prevent breast cancer – specifically triple negative breast cancer (TNBC), the most lethal form of the disease – as well as a vaccine to prevent ovarian cancer. These vaccine technologies focus on immunizing against “retired” proteins that have been found to be expressed in certain forms of cancer. Anixa’s unique business model of partnering with world-renowned research institutions on clinical development allows the company to continually examine emerging technologies in complementary fields for further development and commercialization. To learn more, visit www.anixa.com or follow Anixa on Twitter, LinkedIn, Facebook and YouTube.
Forward-Looking Statements: Statements that are not historical fact may be considered forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Forward-looking statements are not statements of historical facts, but rather reflect Anixa’s current expectations concerning future events and results. We generally use the words “believes,” “expects,” “intends,” “plans,” “anticipates,” “likely,” “will” and similar expressions to identify forward-looking statements. Such forward-looking statements, including those concerning our expectations, involve risks, uncertainties and other factors, some of which are beyond our control, which may cause our actual results, performance or achievements, or industry results, to be materially different from any future results, performance, or achievements expressed or implied by such forward-looking statements. These risks, uncertainties and factors include, but are not limited to, those factors set forth in “Item 1A - Risk Factors” and other sections of our most recent Annual Report on Form 10-K as well as in our Quarterly Reports on Form 10-Q and Current Reports on Form 8-K. We undertake no obligation to publicly update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except as required by law. You are cautioned not to unduly rely on such forward-looking statements when evaluating the information presented in this press release.
Contact:
Mike Catelani
mcatelani@anixa.com
408-708-9808
Exhibit 99.2