
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM
CURRENT REPORT
Pursuant to Section 13 or 15(d) of the
Securities Exchange Act of 1934
Date of Report (Date of earliest event reported):
(Exact name of registrant as specified in its charter)
| (State or other jurisdiction of incorporation) |
(Commission File Number) | (IRS Employer Identification No.) |
(Address of Principal Executive Offices)
Registrant’s telephone
number: (
(Former name or former address, if changed since last report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions (see General Instruction A.2. below):
| Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) | |
| Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) | |
| Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) | |
| Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item 7.01 Regulation FD Disclosure.
On February 18, 2023, Actinium Pharmaceuticals, Inc. (the “Company”), announced that Iomab-B met the primary endpoint of the pivotal Phase 3 SIERRA trial producing higher rates of durable Complete Remission (“dCR”) 6-months following initial complete remission after bone marrow transplant (“BMT”) with high statistical significance (p<0.0001). Iomab-B also significantly improved event-free survival (“EFS”), a secondary endpoint of the SIERRA trial, for which Iomab-B produced a 78% lower probability of an event resulting an EFS Hazard Ratio=0.22 (p<0.0001). Patients receiving Iomab-B had a 100% increase in 1-year overall survival (“OS”) and median OS compared to patients on the control arm. Overall survival is a secondary endpoint of the SIERRA trial, but the crossover arm of the SIERRA trial confounds statistics. Patients receiving Iomab-B who reached 6-month dCR had long-term survival outcomes with 92% 1-year OS and 60 2-year OS. Iomab-B was well tolerated based on the targeted nature, resulting in four times lower rates of sepsis in patients receiving Iomab-B compared to the control arm and clinically meaningful lower rate of graft versus host disease (“GVHD”).
These results were presented at the 2023 Tandem Meetings: Transplantation and Cellular Therapy Meetings of the American Society for Transplantation and Cellular Therapy (ASTCT) and the Center for International Blood & Marrow Transplant Research (CIBMTR) in a late-breaker presentation on Saturday, February 18, 2023. Also on February 18, 2023, the Company hosted an investor conference call and presented the results of the SIERRA trial as well as the potential market and commercial opportunity for Iomab-B.
The Company issued a press release detailing these results, a copy of which is furnished as Exhibit 99.1 to this Current Report on Form 8-K and is incorporated by reference herein. The Company also made an investor presentation detailing the same results, a copy of which is furnished as Exhibit 99.2 to this Current Report on Form 8-K and is incorporated by reference herein. The Company undertakes no obligation to update, supplement or amend the materials attached hereto as Exhibit 99.1 and 99.2
In accordance with General Instruction B.2 of Form 8-K, the information in this Item 7.01 of this Current Report on Form 8-K, including Exhibit 99.1 and Exhibit 99.2, shall not be deemed “filed” for the purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that section, nor shall it be deemed incorporated by reference in any filing under the Exchange Act or the Securities Act of 1933, as amended, except as shall be expressly set forth by reference in such a filing. Furthermore, the furnishing of information under Item 7.01 of this Current Report on Form 8-K is not intended to constitute a determination by the Company that the information contained herein, including the exhibits hereto, is material or that the dissemination of such information is required by Regulation FD.
Item 9.01 Financial Statements and Exhibits
(d) Exhibits.
| Exhibit Number |
Description | |
| 99.1 | Press Release, dated February 18, 2023 (furnished herewith pursuant to Item 7.01) | |
| 99.2 | Investor Presentation, dated February 18, 2023 (furnished herewith pursuant to Item 7.01) | |
| 104 | Cover Page Interactive Data File (formatted as Inline XBRL) |
1
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| Actinium Pharmaceuticals, Inc. | ||
| Date: February 21, 2023 |
/s/ Sandesh Seth | |
| Name: | Sandesh Seth | |
| Title: | Chairman and Chief Executive Officer | |
2
Exhibit 99.1

Actinium Announces Positive Full Data Results From the Pivotal Phase 3 SIERRA Trial in Patients with Active, Relapsed or Refractory Acute Myeloid Leukemia
- Iomab-B met the primary endpoint of durable Complete Remission (dCR) of 6-months following initial complete remission after BMT with high statistical significance (p-value of <0.0001), 22% of patients achieved dCR in the Iomab-B arm compared to 0% in the control arm
- In patients achieving 6-month dCR with Iomab-b, 1-year survival of 92% and 2-year survival of 60% was achieved; median overall survival (OS) has not been reached in these patients
- Iomab-B demonstrated significant improvement in Event Free Survival (EFS) with a Hazard Ratio = 0.22, p<0.0001
- Iomab-B doubled 1-year survival and median overall survival compared to control arm patients who did not crossover
- Iomab-B was well tolerated with a favorable safety profile – 4 times lower rate of sepsis than control arm
- Company to host conference call and webcast on Saturday, February 18, 2023 at 6:00 PM EST to highlight full SIERRA results
NEW YORK, NY – February 18, 2023 – Actinium Pharmaceuticals, Inc. (NYSE AMERICAN: ATNM) (Actinium or the Company), a leader in the development of targeted radiotherapies, today announced positive results for the primary and secondary endpoints from its pivotal Phase 3 SIERRA trial of Iomab-B in patients age 55 and above with active relapsed or refractory acute myeloid leukemia (r/r AML). Iomab-B met the primary endpoint of durable Complete Remission (dCR) of 6-months following initial complete remission following BMT with a high degree of statistical significance (p<0.0001). Additionally, Iomab-B produced a significant and clinically meaningful improvement in the secondary endpoint of Event-Free Survival (EFS), with a 78% reduction in the probability of an event (Hazard Ratio=0.22, p<0.0001). Iomab-B doubled 1-year survival compared to the control arm excluding cross over patients (26.1% vs 13.1%) as well as median overall survival (6.4 months vs. 3.2 months). Iomab-B was well tolerated with four times lower rates of sepsis (6.1% vs 28.6%) and lower rates of febrile neutropenia, mucositis and acute graph versus host disease (aGVHD). Iomab-B enabled unprecedented access to BMT with 100% engraftment in patients receiving a therapeutic dose of Iomab-B compared to 18% of patients in the control arm and Iomab-B produced a 75% post-BMT Complete Remission (CR) rate compared to 6.3% post-BMT CR in the control arm. These high rates of access and post-BMT CR enabled the highly significant primary endpoint results. The full SIERRA results were presented in the late-breaker session at the 2023 Tandem Meetings: Transplantation & Cellular Therapy Meetings of the American Society for Transplantation and Cellular Therapy (ASTCT) and the Center for International Blood & Marrow Transplant Research (CIBMTR).
Investor Conference Call and Webcast Details:
| Time / Date: | 6:00 PM EST on Saturday, February 18, 2023 |
| Presenters: | Sandesh Seth, Chairman & CEO |
Madhuri Vusirikala M.D., VP, Clinical Development – BMT & Cellular Therapy
Avinash Desai, M.D., Chief Medical Officer
Caroline Yarbrough, Chief Commercial Officer
| Dial-in: | 1-877-407-0784 (toll-free domestic) or 1-201-689-8560 (international) or by clicking on this link and requesting a return call |
| Live webcast: | To access the live webcast of the call with slides please visit the Investors section of Actinium’s website https://ir.actiniumpharma.com/presentations-webinars or https://viavid.webcasts.com/starthere.jsp?ei=1590226&tp_key=580722640c |
An archived webcast will be available on the Actinium’s website (click here) after the event.
Dr. Sergio Giralt, Deputy Head, Division of Hematologic Malignancies, Attending Physician, Adult BMT Service at Memorial Sloan Kettering Cancer Center, stated, “The SIERRA trial results are an exciting advancement for older patients with active r/r AML and will be practice changing in how we treat these patients. I am thrilled to see a high percentage of Iomab-B patients who achieved durable remissions reaching the critical 2-year survival mark. Significant improvement in event-free survival and overall survival, with an excellent safety profile in the SIERRA trial, demonstrate the potential of Iomab-B becoming a new standard of care for active, r/r AML.”
SIERRA Trial Results
The pivotal Phase 3 SIERRA trial is a 153-patient, randomized, multi-center, controlled trial, where Iomab-B is compared to the control arm that allowed physician’s choice of over 20 available agents including chemotherapies and/or targeted therapies such as Venetoclax (Bcl-2), FLT3 inhibitors, IDH inhibitors and Mylotarg. The control arm reflects current best practices for the treatment of r/r AML patients. SIERRA was conducted at 24 of the leading BMT centers in the United States and Canada. SIERRA enrolled older, heavily pre-treated patients with active disease and high-risk characteristics who would not be offered BMT in standard practice outside of a clinical trial and therefore have dismal survival outcomes of two to three months.
Iomab-B Patient Characteristics:
| - | Patients with active, r/r disease |
| - | Median age: 64 (55-77) |
| - | Intermediate and adverse cytogenetics and molecular risk: >90% |
| - | Majority of patients had primary induction failure or first early relapse: 78% |
| - | Median blast count: 30% |
| - | Prior lines of treatment: 3 (1-8) |
2
BMT Access and Engraftment:
All patients receiving the therapeutic dose of Iomab-B were able to access BMT with 100% engraftment. Patients in the Iomab-B arm were able to access a BMT without having to first attain a CR, consequently they were able to access BMT in half the time compared to the control arm as those patients need to attain a CR prior to BMT, which is the norm per current practice.
| - | Iomab-B treatment provided unprecedented access to BMT and engraftment without delay (less than 20 days for platelets and neutrophils) in all patients who received the therapeutic dose of Iomab-B (66/66), (59/59 for per protocol analysis) |
| - | Iomab-B enabled more than a 6x increase in BMT access compared to the control arm where 17% of patients (11/64) were able to access a BMT per protocol analysis |
| - | Of the 82% of patients (62/76) in the control arm who failed to achieve a CR and access BMT, 67% of patients (40/62) were able to crossover. Crossover patients are counted as failures for the primary endpoint analysis. Of the 40 crossover patients, 100% (40/40) were able to receive Iomab-B and accessed BMT also achieving engraftment without delay |
| - | Iomab-B enabled access to BMT in approximately half the time (median of 29 days) compared to control arm patients (median 66.5 days) |
Post-BMT CR:
| - | 75% of patients (44/59) receiving Iomab-B achieved an initial remission after their BMT compared to 6.3% of patients (4/64) in the control arm which represents a 12x increase in post-BMT CR rates in favor of Iomab-B |
Primary Endpoint – dCR 6-months After Initial CR:
| - | Iomab-B met the primary endpoint of 6 months dCR with a high degree of statistical significance (p<0.0001) |
| - | 22% of patients (13/59) achieved dCR on the SIERRA arm compared to 0% of patients on the control arm |
| - | Patients who achieved 6-month dCR had 92% 1-year survival and 60% 2-year survival. Median OS has not been reached in these patients |
Secondary Endpoints – Event Free Survival and Overall Survival:
| - | Iomab-B demonstrated significant improvement in EFS with a Hazard Ratio = 0.22, p<0.0001, which means Iomab-B reduced the probability of an event by 78%. EFS is not confounded by the SIERRA crossover arm and allows for direct comparison of survival outcomes between Iomab-B and the control arm |
| ● | Event is defined as not achieving CR/CRp, crossover, not receiving BMT, relapse or death |
| - | Iomab-B doubled 1-year survival and median overall OS of Iomab-B compared to patients who did not crossover in the control arm was 26.1% vs 13.1% and Median OS was 6.4 months vs 3.2 months |
| - | In the crossover arm, 1-year overall survival was 35.8% in patients who received Iomab-Band median overall survival was 7.1 months |
3
Safety Information:
| - | Iomab-B was well-tolerated with a favorable safety profile |
| - | In transplanted patients, incidence of sepsis was four times lower in the Iomab-B arm then the control arm (6.1% vs 28.6%) |
| - | Rates of other treatment related adverse events were lower in favor of Iomab-B, including febrile neutropenia (43.9% vs. 50%), mucositis (15.2% vs 21.4%) and aGVHD (26.1% vs 35.7%) |
Dr. Avinash Desai, Chief Medical Officer of Actinium, said, “We are excited that Iomab-B met the primary endpoint and produced positive results across all SIERRA trial endpoints with improved safety compared to control arm in such a difficult patient population. In routine clinical BMT practice, patients enrolled on SIERRA would never be considered for transplant and often have dismal outcomes. Iomab-B provides unprecedented BMT access and improved outcomes with better tolerability – opening the promise of better transplant outcomes for the entire universe of relapsed and refractory AML patients. These results clearly demonstrate Iomab-B’s practice expanding opportunity as more patients will be able to access transplant and upon reaching the 100-day post-transplant mark they can return to their referring hematologist for long-term care. We look forward to launching an early access program, completing our BLA submission and initiating life cycle management activities to bring Iomab-B to as broad a patient population as possible.”
Sandesh Seth, Actinium’s Chairman and CEO, added, “These positive SIERRA results will help to establish Iomab-B as a new standard of care for r/r AML. Iomab-B is a very attractive option for patients due to its excellent safety and strong efficacy profile. It will enable physicians to provide a treatment intervention with potential long-term survival outcomes and will help bring more patients to curative BMTs. We truly believe that Iomab-B enables potentially better value to be unlocked by getting more patients safely to an effective BMT and by increasing the length and quality of life for patients who otherwise would have dismal outcomes using currently available options. The commercial opportunity for Iomab-B is attractive as the majority of relapsed/refractory patients cannot be treated with a BMT today and Iomab-B can enable them to access this potentially curative treatment. These patients comprise of over half of all AML patients. In addition, the lack of current or visible competition for Iomab-B and the concentration of BMT centers imply that successful commercialization of this high-value treatment can be achieved with a streamlined, efficient organization that is sparing to the balance sheet. We look forward to establishing this practice expanding treatment as the standard of care and to updating on our plans to file the BLA and progress toward this goal.”
About Iomab-B and the Pivotal Phase 3 SIERRA Trial
Iomab-B is a first-in-class targeted radiotherapy intended to improve patient access to potentially curative BMT by simultaneously and rapidly depleting blood cancer, immune and bone marrow stem cells that uniquely express CD45. Multiple studies have demonstrated increased survival in patients receiving BMT, however, an overwhelming majority of patients with blood cancers do not receive BMT as current approaches do not produce a remission, which is needed to advance to BMT, or are too toxic. Studied in over 400 patients, prior studies with Iomab-B have demonstrated nearly universal access to BMT, increased survival and tolerability in multiple clinical trials including the recently completed pivotal Phase 3 SIERRA trial in patients with active (leukemic blasts >5%), relapsed or refractory acute myeloid leukemia (r/r AML) age 55 and above.
Iomab-B met the primary endpoint of durable Complete Remission (dCR) of 6 months after initial remission post-BMT in the pivotal Phase 3 SIERRA trial with high statistical significance (p<0.0001). Iomab-B produced a 75% post-BMT CR rate (44/59 patients), which is 12-times greater than the post-BMT rate of 6.3% (4/64 patients) in the control arm. Patients receiving Iomab-B had a 78% lower probability of an event, defined as not achieving a CR/CRp, crossover, not receiving a BMT, relapse or death, with a Hazard Ratio of 0.22 (p<0.0001). Iomab-B doubled 1-year overall survival with 26.1% compared to 13.1% in the control arm for patients who did not crossover as well as median overall survival with 6.4 months vs 3.2 months. Overall survival statistics are confounded by the crossover arm. Crossover patients had a 35.8% 1-year overall survival rate. Due to its targeted nature, Iomab-B was well tolerated with four times lower rates of sepsis compared to the control arm (6.1% vs. 28.6%) and lower rates of BMT associated adverse events including febrile neutropenia, mucositis and graft versus host disease (GVHD). Actinium intends to submit a Biologics License Application (BLA) seeking approval for Iomab-B in 2023 to address patients age 55+ with r/r AML who cannot access BMT with currently available therapies. Iomab-B has been granted Orphan Drug Designation from the U.S. Food and Drug Administration (FDA) and has patent protection into 2037.
4
The pivotal Phase 3 SIERRA (Study of Iomab-B in Elderly relapsed or refractory AML) is a 153-patient, randomized, multi-center clinical trial, studying Iomab-B compared to the control arm of physician’s choice of salvage therapy. Control arm options included chemotherapies like cytarabine and daunorubicin and targeted agents such as a Bcl-2 inhibitor (Venetoclax), FLT3 inhibitors and IDH 1/2 inhibitors. The SIERRA control arm reflects real-world treatment of r/r AML patients with over 20 agents used alone or in combination as no standard of care exists for this patient population. The SIERRA trial enrolled patients at 24 leading transplant centers in the United States and Canada that perform over 30% of AML BMTs.
Developed at the Fred Hutchinson Cancer Research Center, a pioneer in the field of BMT, Iomab-B is supported by data in six disease indications including leukemias, lymphomas and multiple myeloma, which afflict over 100,000 patients annually. Actinium intends to pursue additional indications for Iomab-B beyond AML. Actinium also intends to pursue international regulatory approvals independently and through partnerships. In April 2022, Actinium licensed the European, Middle East and North African commercial rights for Iomab-B to Immedica AB, a fully-fledged independent pharmaceutical company headquartered in Sweden. In exchange, Actinium received an upfront payment of $35 million USD with the potential for an additional $417 million USD in regulatory and sales milestones and mid-twenty percent royalties. Europe represents a commercial opportunity double the size of the United States by number of patients with AML receiving BMT. Iomab-B has been granted Orphan Drug Designation by the European Medicines Agency (EMA) and has received positive Scientific Advice from the Committee for Medicinal Products for Human Use (CHMP) of the EMA indicating that the Phase 3 SIERRA trial design, primary endpoint and planned statistical analysis are acceptable as the basis for a Marketing Authorization Application.
About Actinium Pharmaceuticals, Inc.
Actinium Pharmaceuticals, Inc. is a clinical-stage biopharmaceutical company developing targeted radiotherapies to deliver cancer-killing radiation with cellular level precision to treat patients with high unmet needs. Actinium’s clinical pipeline is led by targeted radiotherapies that are being applied to targeted conditioning, which is intended to selectively deplete a patient’s disease or cancer cells and certain immune cells prior to a bone marrow transplant (BMT), gene therapy or adoptive cell therapy, such as CAR-T, to enable engraftment of these transplanted cells with minimal toxicities. Our lead product candidate, Iomab-B (I-131 apamistamab) has been studied in over four hundred patients, including the pivotal Phase 3 Study of Iomab-B in Elderly Relapsed or Refractory Acute Myeloid Leukemia (SIERRA) trial for BMT conditioning. The SIERRA trial was positive with Iomab-B meeting the primary endpoint of durable Complete Remission of 6-months with high statistical significance (p<0.0001). Iomab-B enabled 100% of patients to access a BMT and produced higher rates of post-BMT CR. Iomab-B produced positive results for the secondary endpoints of the SIERRA trial including reducing the probability of an event by 78% resulting in an Event-Free Survival (EFS) Hazard Ratio of 0.22 (p<0.0001), doubled 1-year overall survival and median overall survival. Iomab-ACT, low dose I-131 apamistamab, is being studied as a targeted conditioning agent in a Phase 1 study with a CD19 CAR T-cell Therapy with Memorial Sloan Kettering Cancer Center with NIH funding. Actimab-A, our second most advanced product candidate has been studied in approximately 150 patients with Acute Myeloid Leukemia or AML, including in combination trials with the chemotherapy regimen CLAG-M and with venetoclax, a targeted therapy. Actimab-A or lintuzumab-Ac225 is an Actinium-225 based antibody radiation conjugate targeting CD33, a validated target in AML. Actinium has entered into a Cooperative Research and Development Agreement (CRADA) with the National Cancer Institute (NCI) to develop Actimab-A as a single agent or combination with chemotherapy, targeted agents or immunotherapy in Phase 1, 2 or 3 trials. The NCI will fund clinical trial expenses under the CRADA while Actinium will supply Actimab-A. The NCI is currently accepting proposals for non-clinical and clinical studies with Actimab-A. Actinium is a pioneer and leader in the field of Actinium-225 alpha therapies with an industry leading technology platform comprising over 190 patents and patent applications including methods of producing the radioisotope AC-225. Our technology and expertise have enabled collaborative research partnerships with Astellas Pharma, Inc. for solid tumor theranostics, with AVEO Oncology Inc. to create an Actinium-225 HER3 targeting radiotherapy for solid tumors, and with EpicentRx, Inc. to create targeted radiotherapy combinations with their novel, clinical stage small molecule CD47-SIRPα inhibitor. More information is available on Actinium’s website: https://www.actiniumpharma.com/.
Investors:
Hans Vitzthum
LifeSci Advisors, LLC
Hans@LifeSciAdvisors.com
(617) 430-7578
5
Exhibit 99.2

ATNM: NYSE AMERICAN February 18, 2022 Iomab - B SIERRA Trial Phase 3 Results

Disclaimer and Safe Harbor The information presented herein contains express and implied forward - looking statements regarding the current intentions, expectations, estimates, opinions and beliefs of Actinium Pharmaceuticals, Inc . (“Actinium”) that are not historical facts . These forward - looking statements include statements regarding Actinium’s expectations for its product candidates (including their therapeutic and commercial potential, anticipated future development activities, anticipated timing of development activities, including initiation of clinical trials and presentations of clinical data and the indications Actinium and its collaborators plan to pursue), future results of operations and financial position, business strategy, strategic collaborations, any royalty or milestone payments and Actinium’s ability to obtain and maintain intellectual property protection for its product candidates . Such forward - looking statements may be identified by words such as “believes”, “may”, “will”, “expects”, “endeavors”, “anticipates”, “intends”, “plans”, “estimates”, “projects”, “should”, “objective” and variations of such words and similar words . These statements are based on management’s current expectations and are subject to risks and uncertainties that may cause actual results to differ materially from the anticipated or estimated future results, including the risks and uncertainties associated with preliminary study results varying from final results, estimates of potential markets for drugs under development, clinical trials, actions by the FDA and other governmental agencies, regulatory clearances, responses to regulatory matters, the market demand for and acceptance of Actinium’s products and services, performance of clinical research organizations and other risks detailed from time to time in Actinium's filings with the Securities and Exchange Commission (the “SEC”), including without limitation its most recent annual report on Form 10 - K, subsequent quarterly reports on Forms 10 - Q and Forms 8 - K, each as amended and supplemented from time to time . Any forward - looking statements that Actinium makes in this presentation speak only as of the date of this presentation . Except as required by law, Actinium assumes no obligation to update its forward - looking statements whether as a result of new information, future events or otherwise, after the date hereof . Nothing contained in this presentation is, or should be construed as, a recommendation, promise or representation by Actinium or any director, employee, agent, or adviser of Actinium . This presentation does not purport to be all - inclusive or to contain all of the information you may desire . The content of this presentation is subject to copyright, which will be asserted by Actinium, and no part of this presentation may be reproduced, stored in a retrieval system, or transmitted in any form or by any means without prior permission in writing from Actinium . 2

Sandesh Seth Chairman & CEO Dr. Madhuri Vusirikala VP, Clinical Development, BMT & Cellular Therapy Dr. Avinash Desai Chief Medical Officer Caroline Yarbrough Chief Commercial Officer Today’s Speakers & Agenda 3 Introduction and Closing Remarks Iomab - B Phase 3 SIERRA Results Significance of SIERRA Results and Next Steps Iomab - B Market Positioning and Opportunity
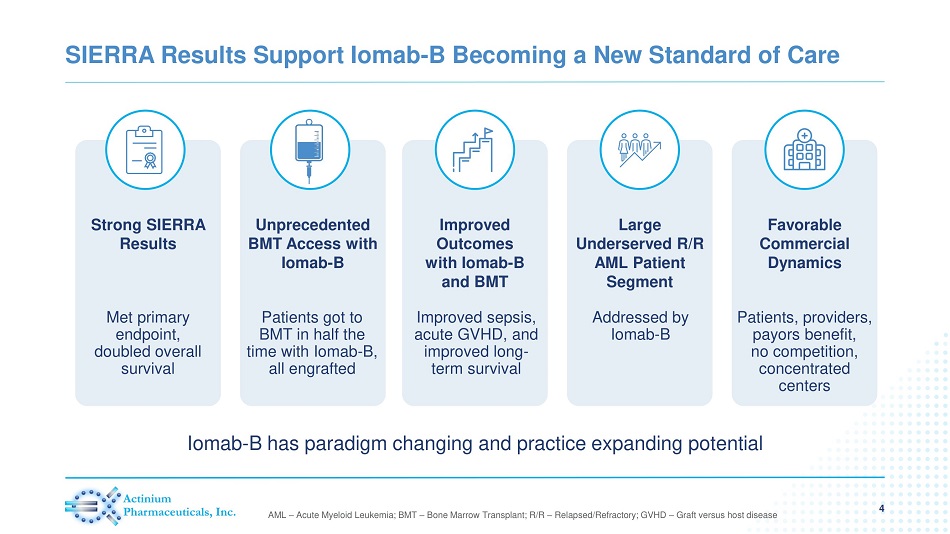
SIERRA Results Support Iomab - B Becoming a New Standard of Care 4 AML – Acute Myeloid Leukemia; BMT – Bone Marrow Transplant; R/R – Relapsed/Refractory; GVHD – Graft versus host disease Unprecedented BMT Access with Iomab - B Large Underserved R/R AML Patient Segment Improved Outcomes with Iomab - B and BMT Strong SIERRA Results Favorable Commercial Dynamics Iomab - B has paradigm changing and practice expanding potential Met primary endpoint, doubled overall survival Patients got to BMT in half the time with Iomab - B, all engrafted Improved sepsis, acute GVHD, and improved long - term survival Addressed by Iomab - B Patients, providers, payors benefit, no competition, concentrated centers

Dr. Madhuri Vusirikala, VP, Clinical Development BMT & Cell Therapy 5 • Joined Actinium in October 2022 as Vice President, Clinical Development, Transplant & Cellular Therapy • Over 20 years of clinical experience specializing in adult allogeneic bone marrow transplant • Most recently, Director of the Allogeneic Stem Cell Transplant Program at University of Texas – Southwestern (UTSW) and Professor of Medicine in the Division of Hematology and Oncology • Serves on several national committees including the National Comprehensive Cancer Network (NCCN) panels for Hematopoietic Stem Cell Transplantation and Acute Lymphoblastic Leukemia, BMT Infonet, and the MDS/Aplastic Anemia Foundation • Fellowship in Bone Marrow Transplant at Vanderbilt University • Fellowship in Hematology - Oncology at the University of Pittsburgh • Residency at SUNY Syracuse • Medical Training at Lady Hardinge College (India)

SIERRA: Positive Efficacy, Safety, and Long - Term Outcomes in R/R AML 6 Trial Endpoints and Metrics Results 6 - month durable Complete Remission (dCR) p <0.0001 Event - Free Survival (EFS) EFS Hazard Ratio of 0.22, p<0.0001 Overall Survival (OS) 100% Increase over control arm 2 - year survival in patients achieving dCR 60% in Iomab - B patients vs. 0% in control arm patients Sepsis 4x lower with Iomab - B GVHD Clinically meaningful lower rate of GVHD with Iomab - B Primary Secondary Long - term Outcomes Key Safety & Tolerability Metrics SIERRA results support the potential for Iomab - B to become the new standard of care for BMT conditioning in R/R AML
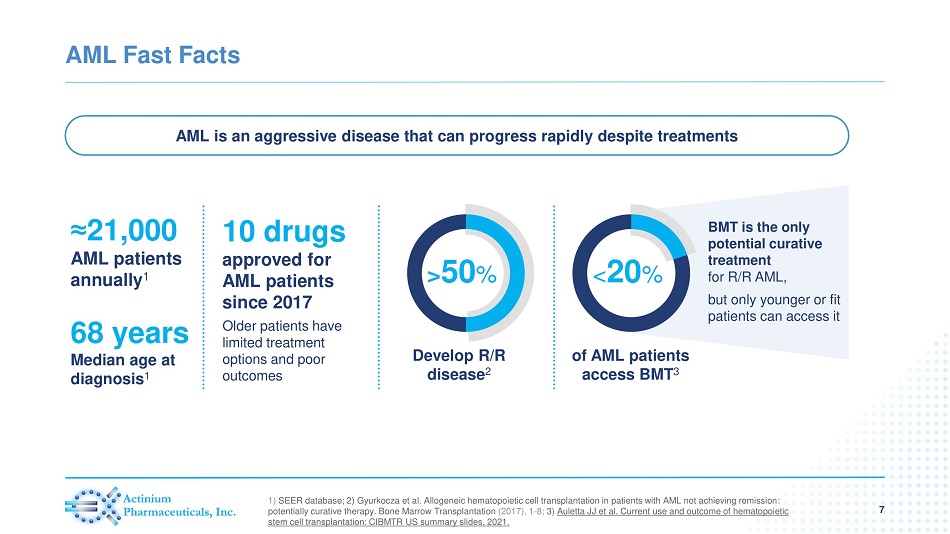
AML Fast Facts 7 ≈ 21,000 AML patients annually 1 AML is an aggressive disease that can progress rapidly despite treatments 68 years Median age at diagnosis 1 Older patients have limited treatment options and poor outcomes Develop R/R disease 2 > 50 % of AML patients access BMT 3 < 20 % BMT is the only potential curative treatment for R/R AML, but only younger or fit patients can access it 1) SEER database; 2) Gyurkocza et al. Allogeneic hematopoietic cell transplantation in patients with AML not achieving remission : potentially curative therapy. Bone Marrow Transplantation (2017), 1 - 8; 3) Auletta JJ et al. Current use and outcome of hematopoietic stem cell transplantation: CIBMTR US summary slides, 2021. 10 drugs approved for AML patients since 2017

Challenges to Achieving Cures in AML Patients must be able to overcome several challenges related to curative BMT Curative Potential Access Need to attain a complete remission (CR) Tolerate and survive effective BMT conditioning • Achieve BMT engraftment • Achieve post - BMT CR • Surmount BMT related complications – Graft failure – Side effects: sepsis, GVHD Outcomes Challenge #1 Challenge #2 Challenge #3 Challenge #4 Overcoming these challenges can result in long - term survival and curative outcomes 8
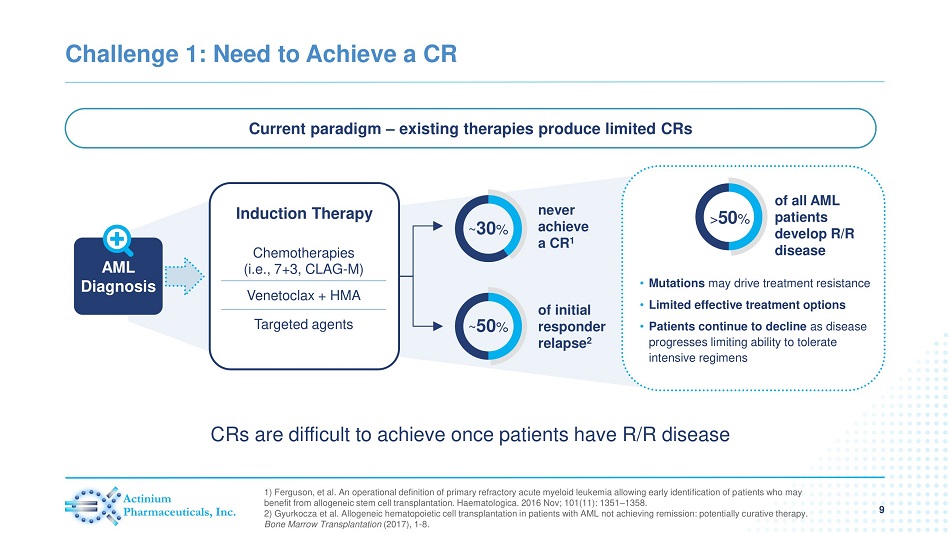
Challenge 1: Need to Achieve a CR 9 1) Ferguson, et al. An operational definition of primary refractory acute myeloid leukemia allowing early identification of p ati ents who may benefit from allogeneic stem cell transplantation. Haematologica. 2016 Nov; 101(11): 1351 – 1358. 2) Gyurkocza et al. Allogeneic hematopoietic cell transplantation in patients with AML not achieving remission: potentially c ura tive therapy. Bone Marrow Transplantation (2017), 1 - 8. Current paradigm – existing therapies produce limited CRs AML Diagnosis of initial responder relapse 2 ~ 50 % never achieve a CR 1 ~ 30 % • Mutations may drive treatment resistance • Limited effective treatment options • Patients continue to decline as disease progresses limiting ability to tolerate intensive regimens of all AML patients develop R/R disease > 50 % Induction Therapy Chemotherapies (i.e., 7+3, CLAG - M) Venetoclax + HMA Targeted agents CRs are difficult to achieve once patients have R/R disease
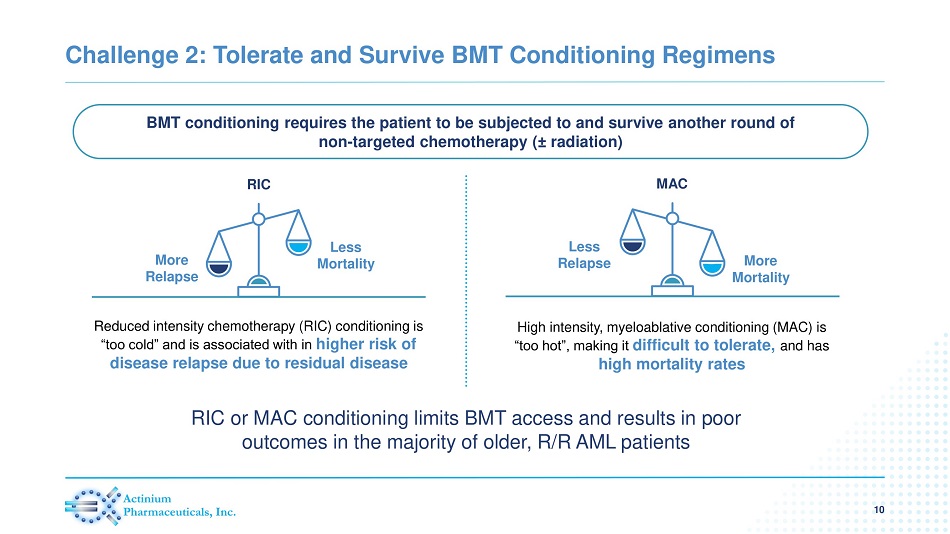
Challenge 2: Tolerate and Survive BMT Conditioning Regimens 10 Reduced intensity chemotherapy (RIC) conditioning is “too cold” and is associated with in higher risk of disease relapse due to residual disease High intensity, myeloablative conditioning (MAC) is “too hot”, making it difficult to tolerate, and has high mortality rates BMT conditioning requires the patient to be subjected to and survive another round of non - targeted chemotherapy ( ± radiation) RIC Less Mortality More Relapse MAC More Mortality Less Relapse RIC or MAC conditioning limits BMT access and results in poor outcomes in the majority of older, R/R AML patients
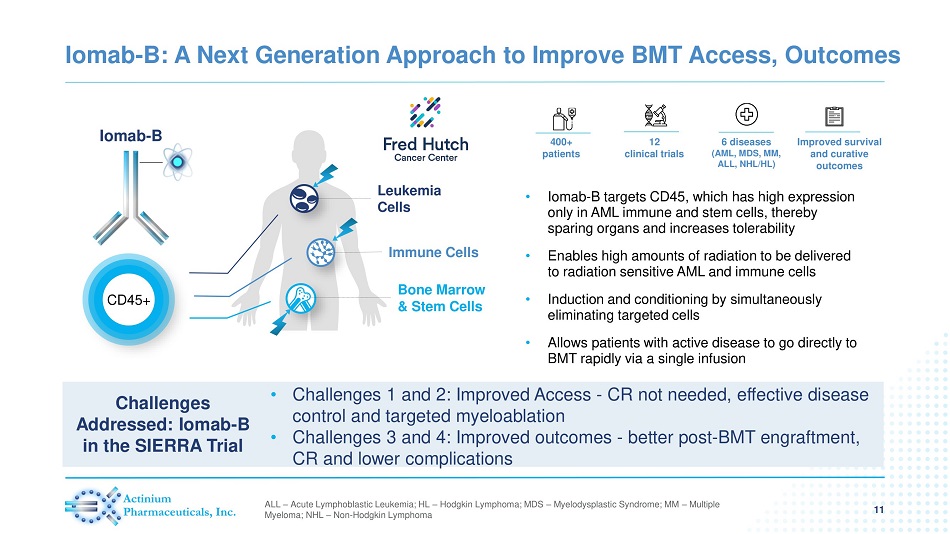
Iomab - B: A Next Generation Approach to Improve BMT Access, Outcomes 11 • Iomab - B targets CD45, which has high expression only in AML immune and stem cells, thereby sparing organs and increases tolerability • Enables high amounts of radiation to be delivered to radiation sensitive AML and immune cells • Induction and conditioning by simultaneously eliminating targeted cells • Allows patients with active disease to go directly to BMT rapidly via a single infusion Iomab - B CD45+ Leukemia Cells Immune Cells Bone Marrow & Stem Cells 400+ patients 12 clinical trials 6 diseases (AML, MDS, MM, ALL, NHL/HL) Improved survival and curative outcomes ALL – Acute Lymphoblastic Leukemia; HL – Hodgkin Lymphoma; MDS – Myelodysplastic Syndrome; MM – Multiple Myeloma; NHL – Non - Hodgkin Lymphoma • Challenges 1 and 2: Improved Access - CR not needed, effective disease control and targeted myeloablation • Challenges 3 and 4: Improved outcomes - better post - BMT engraftment, CR and lower complications Challenges Addressed: Iomab - B in the SIERRA Trial
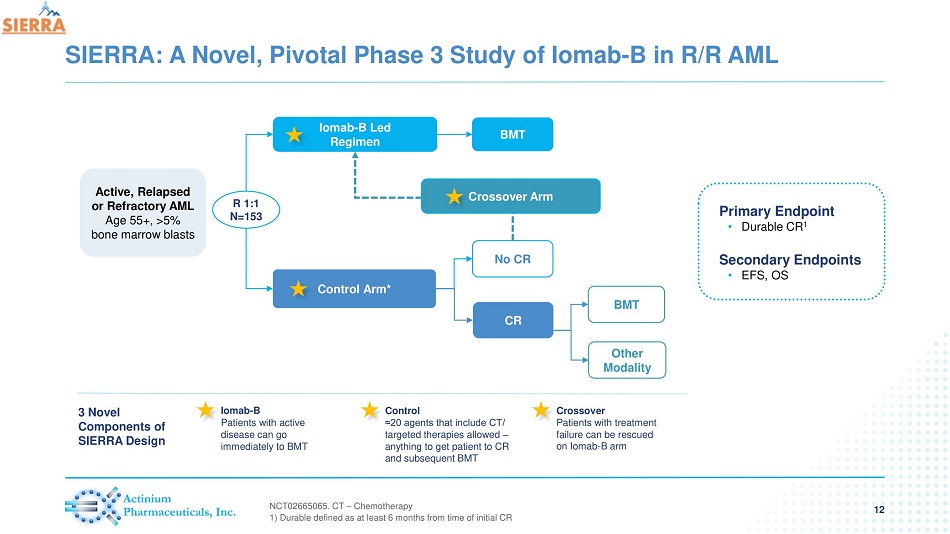
SIERRA: A Novel, Pivotal Phase 3 Study of Iomab - B in R/R AML 12 Primary Endpoint • Durable CR 1 Secondary Endpoints • EFS, OS Iomab - B Led Regimen Control Arm* BMT CR Other Modality No CR BMT Crossover Arm R 1:1 N=153 NCT 02665065 . CT – Chemotherapy 1) Durable defined as at least 6 months from time of initial CR Iomab - B Patients with active disease can go immediately to BMT Control ≈20 agents that include CT/ targeted therapies allowed – anything to get patient to CR and subsequent BMT Crossover Patients with treatment failure can be rescued on Iomab - B arm 3 Novel Components of SIERRA Design Active, Relapsed or Refractory AML Age 55+, >5% bone marrow blasts

First Randomized Trial with Goal to Transplant R/R AML Patients 13 1) Maiti et al. Outcomes of relapsed or refractory acute myeloid leukemia after front - line hypomethylating agent and venetoclax regimens . Haematologica . 2021 Mar 1; 106(3): 894 – 898. Late Breaking Abstract, TCT 2023, Efficacy and Safety Results of the Sierra Trial: A Multicenter, Pivotal Phase 3 Study of Iomab - B Prior to Allogeneic HCT Versus Conventional Care in Older Patients with Active, R/ R AML Patients enrolled in SIERRA have poor expected survival SIERRA Trial: Iomab - B Arm Patient Characteristics • Active R/R disease • Median age: 64 (55 - 77) • Intermediate and adverse cytogenetics and molecular risk: >90% • Majority of patients had primary induction failure or first early relapse: 78% • Median blast count: 30% • Prior lines of treatment: 3 (1 - 8) SIERRA Designed to Evaluate if Iomab - B + BMT Can Improve the Outcomes of Active R/R AML Patients R/R after Frontline Therapy 1 Median OS (months) Venetoclax + HMA 2.3 Intensive Chemo 3.6 34% 44% 2% 10% 5% 6% Chemotherapy (34%) Venetoclax (44%) CD33 (2%) FLT3 (10%) IDH (5%) Other (6%) SIERRA Patients Had Significant and Diverse Prior Therapy Representative of Current AML Treatment
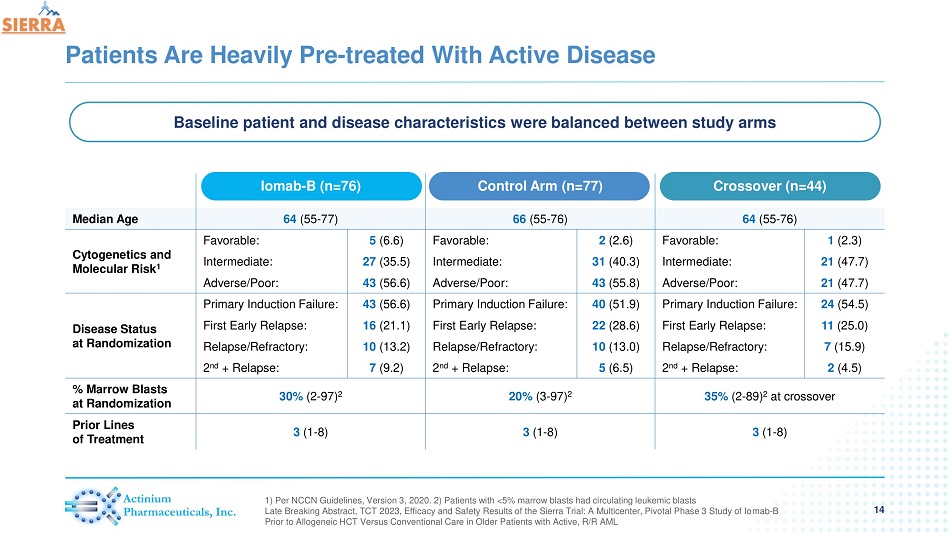
Patients Are Heavily Pre - treated With Active Disease 14 1) Per NCCN Guidelines, Version 3, 2020. 2) Patients with <5% marrow blasts had circulating leukemic blasts Late Breaking Abstract, TCT 2023, Efficacy and Safety Results of the Sierra Trial: A Multicenter, Pivotal Phase 3 Study of Io mab - B Prior to Allogeneic HCT Versus Conventional Care in Older Patients with Active, R/R AML Iomab - B (n=76) Control Arm (n=77) Crossover (n=44) Median Age 64 (55 - 77) 66 (55 - 76) 64 (55 - 76) Cytogenetics and Molecular Risk 1 Favorable: 5 (6.6) Favorable: 2 (2.6) Favorable: 1 (2.3) Intermediate: 27 (35.5) Intermediate: 31 (40.3) Intermediate: 21 (47.7) Adverse/Poor: 43 (56.6) Adverse/Poor: 43 (55.8) Adverse/Poor: 21 (47.7) Disease Status at Randomization Primary Induction Failure: 43 (56.6) Primary Induction Failure: 40 (51.9) Primary Induction Failure: 24 (54.5) First Early Relapse: 16 (21.1) First Early Relapse: 22 (28.6) First Early Relapse: 11 (25.0) Relapse/Refractory: 10 (13.2) Relapse/Refractory: 10 (13.0) Relapse/Refractory: 7 (15.9) 2 nd + Relapse: 7 (9.2) 2 nd + Relapse: 5 (6.5) 2 nd + Relapse: 2 (4.5) % Marrow Blasts at Randomization 30% (2 - 97) 2 20% (3 - 97) 2 35% (2 - 89 ) 2 at crossover Prior Lines of Treatment 3 (1 - 8) 3 (1 - 8) 3 (1 - 8) Baseline patient and disease characteristics were balanced between study arms
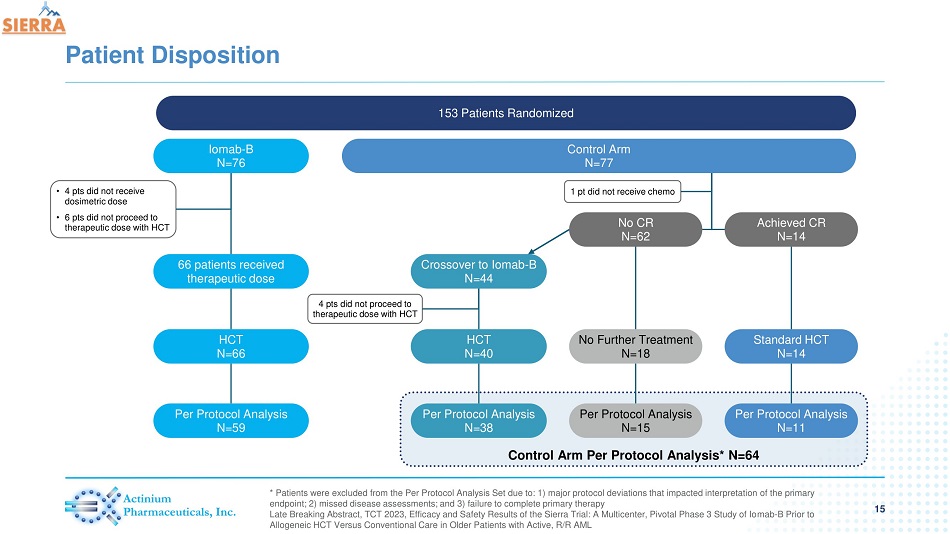
Control Arm Per Protocol Analysis* N=64 Patient Disposition * Patients were excluded from the Per Protocol Analysis Set due to: 1) major protocol deviations that impacted interpretation of the primary endpoint; 2) missed disease assessments; and 3) failure to complete primary therapy Late Breaking Abstract, TCT 2023, Efficacy and Safety Results of the Sierra Trial: A Multicenter, Pivotal Phase 3 Study of Io mab - B Prior to Allogeneic HCT Versus Conventional Care in Older Patients with Active, R/R AML 153 Patients Randomized Control Arm N=77 Iomab - B N=76 66 patients received therapeutic dose HCT N=66 Per Protocol Analysis N=59 • 4 pts did not receive dosimetric dose • 6 pts did not proceed to therapeutic dose with HCT Crossover to Iomab - B N=44 HCT N=40 Per Protocol Analysis N=38 No CR N=62 Achieved CR N=14 1 pt did not receive chemo 4 pts did not proceed to therapeutic dose with HCT No Further Treatment N=18 Per Protocol Analysis N=15 Standard HCT N=14 Per Protocol Analysis N=11 15
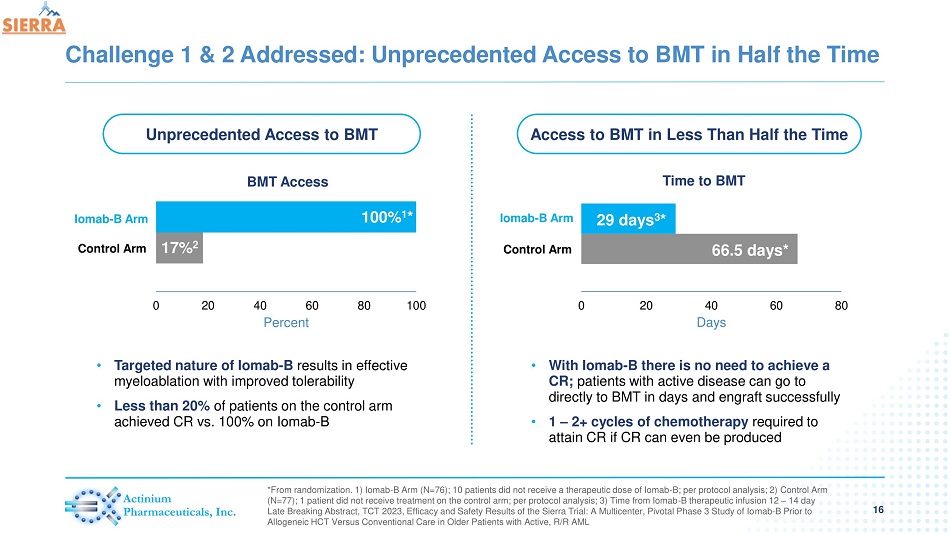
Challenge 1 & 2 Addressed: Unprecedented Access to BMT in Half the Time 16 *From randomization. 1) Iomab - B Arm (N=76); 10 patients did not receive a therapeutic dose of Iomab - B; per protocol analysis; 2) Control Arm (N=77); 1 patient did not receive treatment on the control arm; per protocol analysis; 3) Time from Iomab - B therapeutic infusion 12 – 14 day Late Breaking Abstract, TCT 2023, Efficacy and Safety Results of the Sierra Trial: A Multicenter, Pivotal Phase 3 Study of Io mab - B Prior to Allogeneic HCT Versus Conventional Care in Older Patients with Active, R/R AML Iomab - B Arm 66.5 days* 29 days 3 * 0 20 40 60 80 Days Time to BMT 17% 2 100 % 1 * 0 20 40 60 80 100 Percent BMT Access Control Arm Iomab - B Arm Control Arm Unprecedented Access to BMT Access to BMT in Less Than Half the Time • Targeted nature of Iomab - B results in effective myeloablation with improved tolerability • Less than 20% of patients on the control arm achieved CR vs. 100% on Iomab - B • With Iomab - B there is no need to achieve a CR; patients with active disease can go to directly to BMT in days and engraft successfully • 1 – 2+ cycles of chemotherapy required to attain CR if CR can even be produced
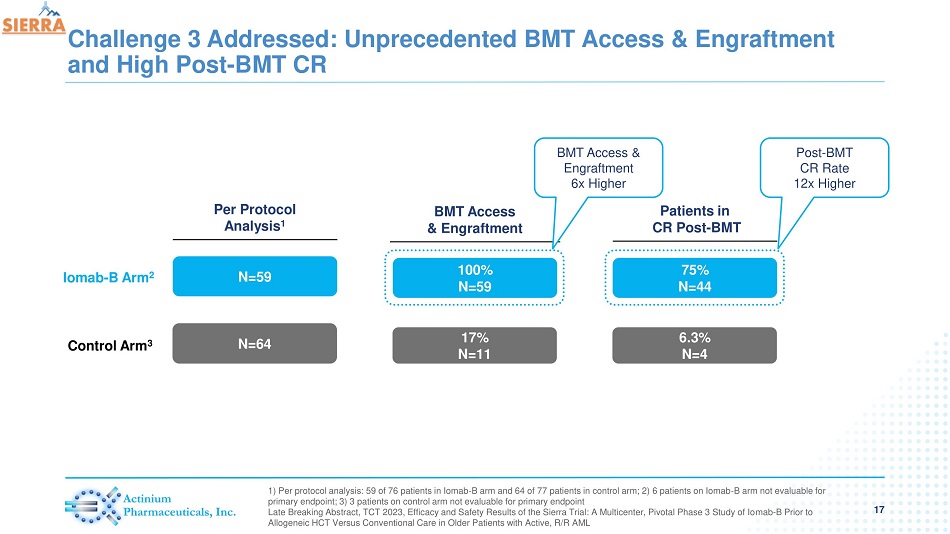
Challenge 3 Addressed: Unprecedented BMT Access & Engraftment and High Post - BMT CR 17 1) Per protocol analysis: 59 of 76 patients in Iomab - B arm and 64 of 77 patients in control arm; 2) 6 patients on Iomab - B arm no t evaluable for primary endpoint; 3) 3 patients on control arm not evaluable for primary endpoint Late Breaking Abstract, TCT 2023, Efficacy and Safety Results of the Sierra Trial: A Multicenter, Pivotal Phase 3 Study of Io mab - B Prior to Allogeneic HCT Versus Conventional Care in Older Patients with Active, R/R AML Patients in CR Post - BMT N=59 N=64 Per Protocol Analysis 1 75% N=44 6.3% N=4 BMT Access & Engraftment 100% N=59 17% N=11 Iomab - B Arm 2 Control Arm 3 BMT Access & Engraftment 6 x Higher Post - BMT CR Rate 12x Higher
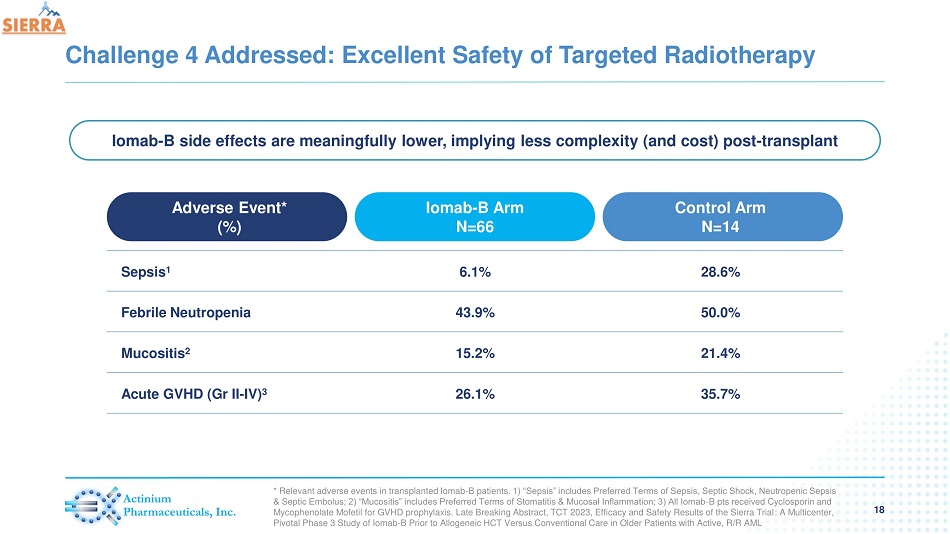
Challenge 4 Addressed: Excellent Safety of Targeted Radiotherapy 18 Adverse Event* (%) Iomab - B Arm N=66 Control Arm N=14 Sepsis 1 6.1% 28.6% Febrile Neutropenia 43.9% 50.0% Mucositis 2 15.2% 21.4% Acute GVHD (Gr II - IV) 3 26.1% 35.7% * Relevant adverse events in transplanted Iomab - B patients. 1) “Sepsis” includes Preferred Terms of Sepsis, Septic Shock, Neutropenic Sepsis & Septic Embolus; 2) “Mucositis” includes Preferred Terms of Stomatitis & Mucosal Inflammation; 3) All Iomab - B pts received Cycl osporin and Mycophenolate Mofetil for GVHD prophylaxis. Late Breaking Abstract, TCT 2023, Efficacy and Safety Results of the Sierra Trial : A Multicenter, Pivotal Phase 3 Study of Iomab - B Prior to Allogeneic HCT Versus Conventional Care in Older Patients with Active, R/R AML Iomab - B side effects are meaningfully lower, implying less complexity (and cost) post - transplant
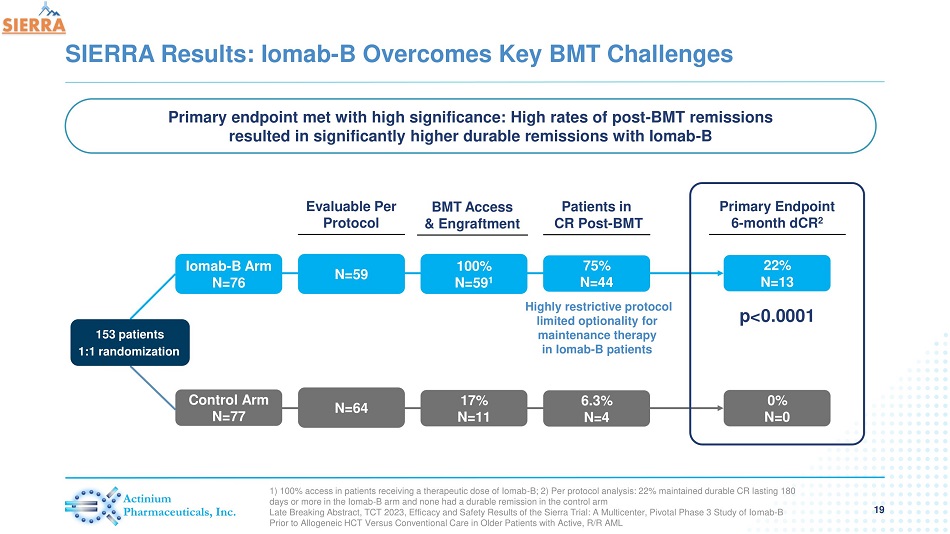
SIERRA Results: Iomab - B Overcomes Key BMT Challenges 19 1) 100% access in patients receiving a therapeutic dose of Iomab - B; 2) Per protocol analysis: 22% maintained durable CR lasting 180 days or more in the Iomab - B arm and none had a durable remission in the control arm Late Breaking Abstract, TCT 2023, Efficacy and Safety Results of the Sierra Trial: A Multicenter, Pivotal Phase 3 Study of Io mab - B Prior to Allogeneic HCT Versus Conventional Care in Older Patients with Active, R/R AML Iomab - B Arm N=76 Control Arm N=77 153 patients 1:1 randomization Patients in CR Post - BMT Primary Endpoint 6 - month dCR 2 N=59 N=64 Evaluable Per Protocol 75% N=44 6.3% N=4 22% N=13 0% N=0 BMT Access & Engraftment 100% N=59 1 17% N=11 p<0.0001 Highly restrictive protocol limited optionality for maintenance therapy in Iomab - B patients Primary endpoint met with high significance: High rates of post - BMT remissions resulted in significantly higher durable remissions with Iomab - B
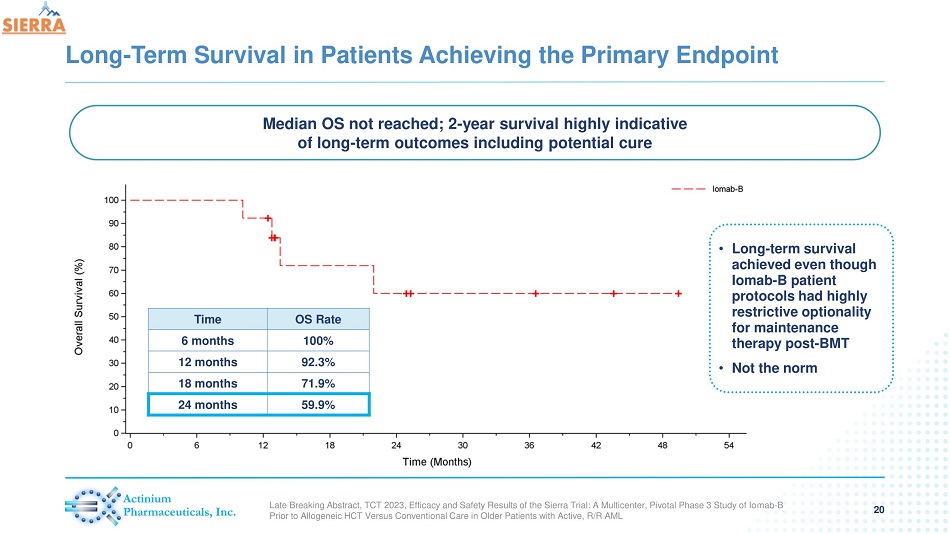
Long - Term Survival in Patients Achieving the Primary Endpoint 20 Time OS Rate 6 months 100% 12 months 92.3% 18 months 71.9% 24 months 59.9% Median OS not reached; 2 - year survival highly indicative of long - term outcomes including potential cure • Long - term survival achieved even though Iomab - B patient protocols had highly restrictive optionality for maintenance therapy post - BMT • Not the norm Late Breaking Abstract, TCT 2023, Efficacy and Safety Results of the Sierra Trial: A Multicenter, Pivotal Phase 3 Study of Io mab - B Prior to Allogeneic HCT Versus Conventional Care in Older Patients with Active, R/R AML
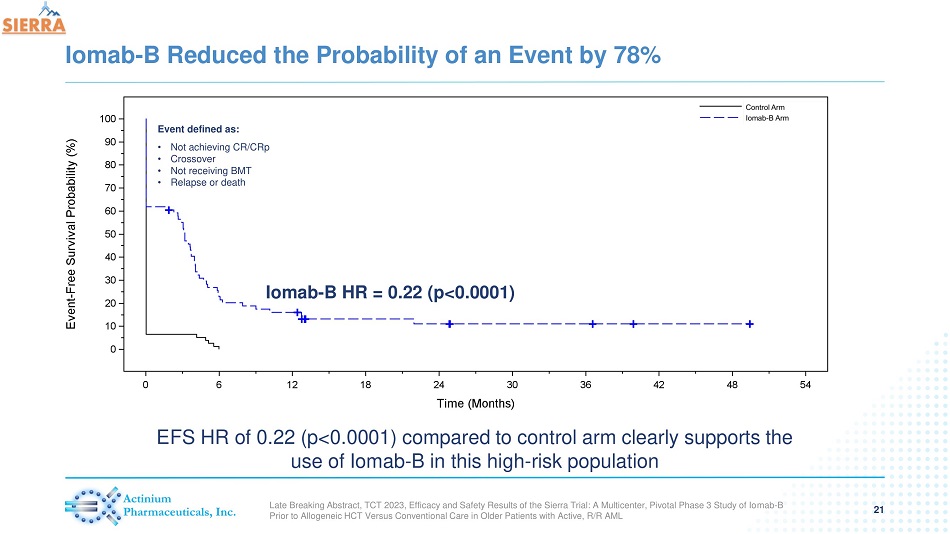
Iomab - B Reduced the Probability of an Event by 78% 21 Iomab - B HR = 0.22 (p<0.0001) Event defined as: • Not achieving CR/CRp • Crossover • Not receiving BMT • Relapse or death EFS HR of 0.22 (p< 0.0001) compared to control arm clearly supports the use of Iomab - B in this high - risk population Late Breaking Abstract, TCT 2023, Efficacy and Safety Results of the Sierra Trial: A Multicenter, Pivotal Phase 3 Study of Io mab - B Prior to Allogeneic HCT Versus Conventional Care in Older Patients with Active, R/R AML
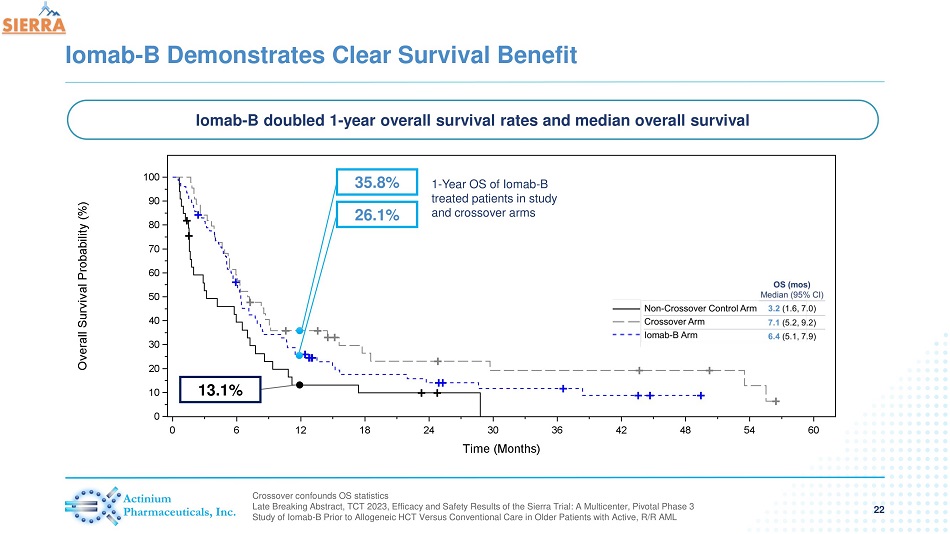
Iomab - B Demonstrates Clear Survival Benefit 22 Crossover confounds OS statistics Late Breaking Abstract, TCT 2023, Efficacy and Safety Results of the Sierra Trial: A Multicenter, Pivotal Phase 3 Study of Iomab - B Prior to Allogeneic HCT Versus Conventional Care in Older Patients with Active, R/R AML 13.1% 26.1% 35.8% Iomab - B doubled 1 - year overall survival rates and median overall survival 1 - Year OS of Iomab - B treated patients in study and crossover arms
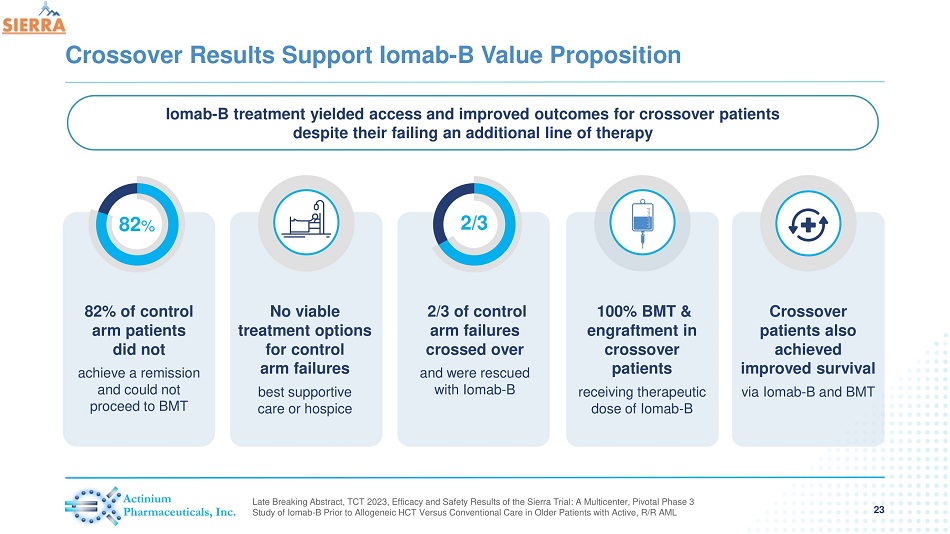
Crossover Results Support Iomab - B Value Proposition 23 82% of control arm patients did not achieve a remission and could not proceed to BMT 82 % No viable treatment options for control arm failures best supportive care or hospice 2/3 of control arm failures crossed over and were rescued with Iomab - B 2/3 100% BMT & engraftment in crossover patients receiving therapeutic dose of Iomab - B Crossover patients also achieved improved survival via Iomab - B and BMT Iomab - B treatment yielded access and improved outcomes for crossover patients despite their failing an additional line of therapy Late Breaking Abstract, TCT 2023, Efficacy and Safety Results of the Sierra Trial: A Multicenter, Pivotal Phase 3 Study of Iomab - B Prior to Allogeneic HCT Versus Conventional Care in Older Patients with Active, R/R AML
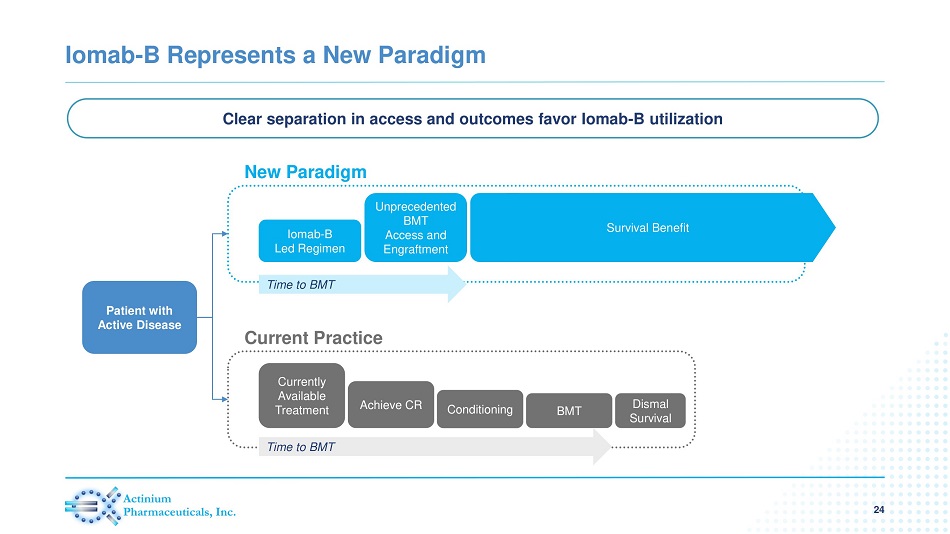
Iomab - B Represents a New Paradigm 24 Clear separation in access and outcomes favor Iomab - B utilization Patient with Active Disease New Paradigm Current Practice Iomab - B Led Regimen Unprecedented BMT Access and Engraftment Time to BMT Currently Available Treatment Achieve CR Conditioning BMT Dismal Survival Time to BMT Survival Benefit

Dr. Avinash Desai, Chief Medical Officer 25 • Joined Actinium in November 2020 and promoted to Chief Medical Officer in 2021 • 27 years of biopharmaceutical experience in clinical development and medical affairs • Joined Actinium from GSK where he was Vice President, Head of U.S. Medical Affairs - Oncology, launching 3 oncology products in 15 months • Previous experience at Janssen Pharmaceuticals (Johnson & Johnson), Eli Lilly & Co. Takeda, Inc. and Sanofi • Contributed to the development and supported multiple blockbuster products including Darzalex and Velcade at Janssen • Participated in multiple successful NDA submissions, launch readiness strategies and execution and life cycle management plans
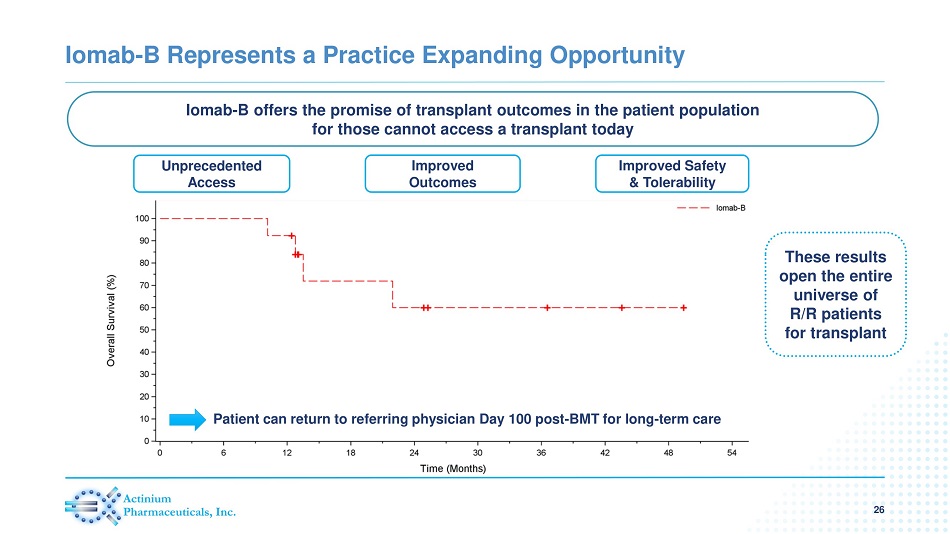
Iomab - B Represents a Practice Expanding Opportunity 26 Improved Safety & Tolerability Improved Outcomes Patient can return to referring physician Day 100 post - BMT for long - term care Unprecedented Access Iomab - B offers the promise of transplant outcomes in the patient population for those cannot access a transplant today These results open the entire universe of R/R patients for transplant
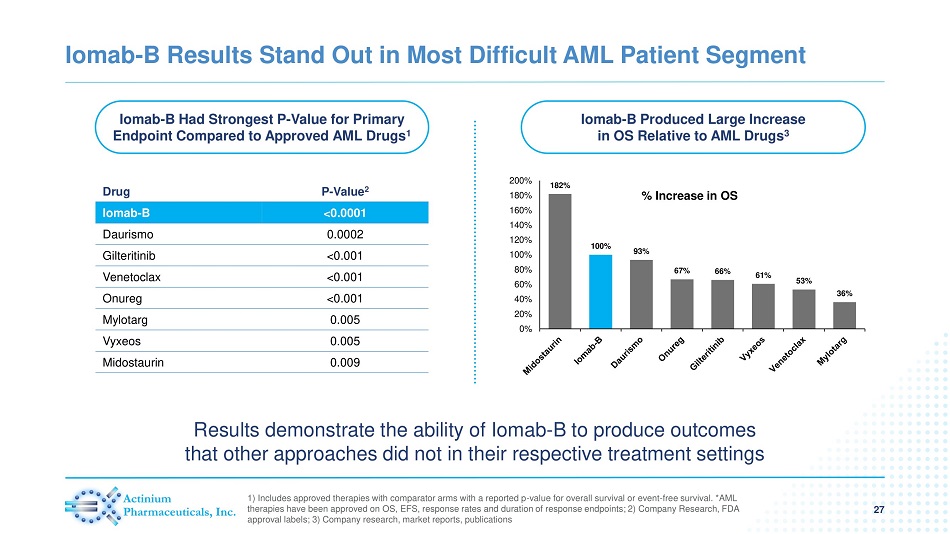
Iomab - B Had Strongest P - Value for Primary Endpoint Compared to Approved AML Drugs 1 Iomab - B Produced Large Increase in OS Relative to AML Drugs 3 Iomab - B Results Stand Out in Most Difficult AML Patient Segment 27 1) Includes approved therapies with comparator arms with a reported p - value for overall survival or event - free survival. *AML therapies have been approved on OS, EFS, response rates and duration of response endpoints; 2) Company Research, FDA approval labels; 3) Company research, market reports, publications % Increase in OS Drug P - Value 2 Iomab - B <0.0001 Daurismo 0.0002 Gilteritinib <0.001 Venetoclax <0.001 Onureg <0.001 Mylotarg 0.005 Vyxeos 0.005 Midostaurin 0.009 182% 100% 93% 67% 66% 61% 53% 36% 0% 20% 40% 60% 80% 100% 120% 140% 160% 180% 200% Results demonstrate the ability of Iomab - B to produce outcomes that other approaches did not in their respective treatment settings
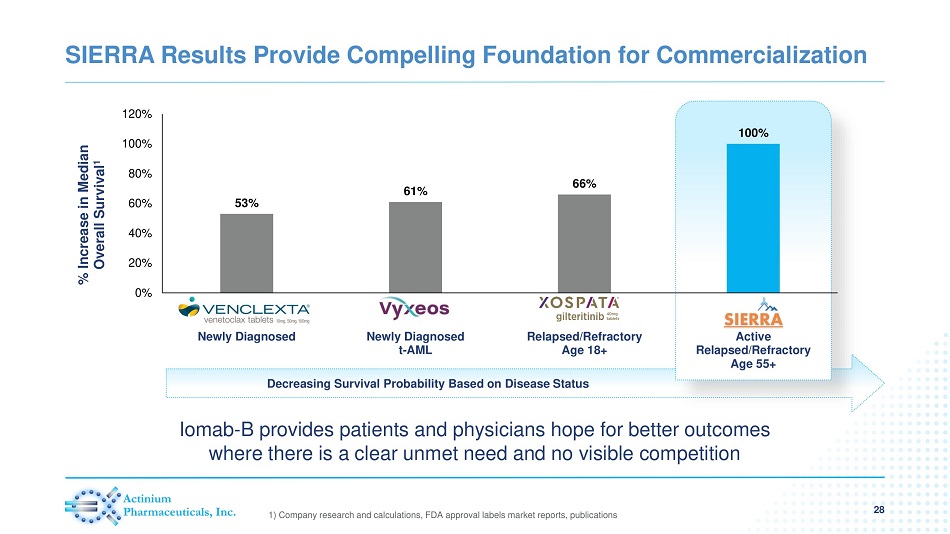
Decreasing Survival Probability Based on Disease Status SIERRA Results Provide Compelling Foundation for Commercialization 28 1) Company research and calculations, FDA approval labels market reports, publications 53% 61% 66% 100% 0% 20% 40% 60% 80% 100% 120% Newly Diagnosed Newly Diagnosed t-AML Relapsed/Refractory Age 18+ Active Relapsed/Refractory Age 55+ % Increase in Median Overall Survival 1 Iomab - B provides patients and physicians hope for better outcomes where there is a clear unmet need and no visible competition
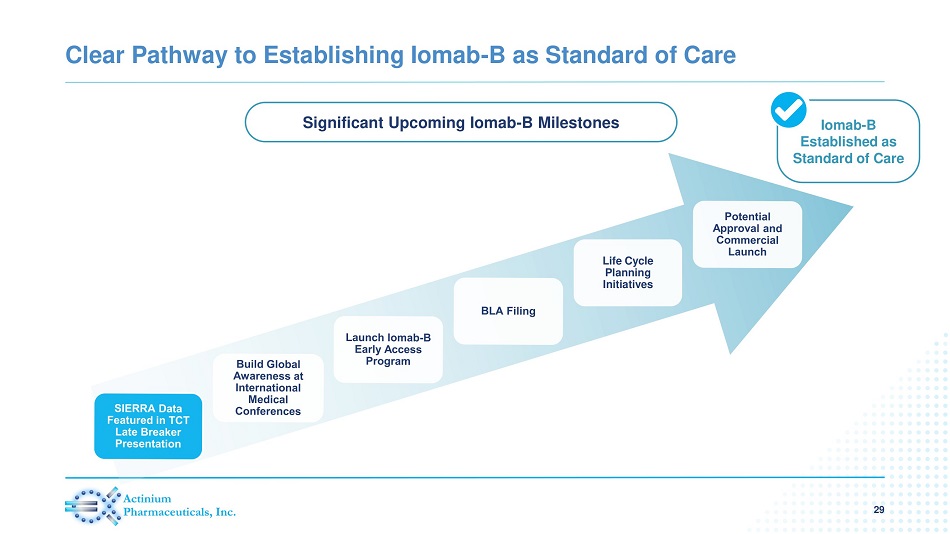
Clear Pathway to Establishing Iomab - B as Standard of Care 29 Iomab - B Established as Standard of Care Life Cycle Planning Initiatives Significant Upcoming Iomab - B Milestones

SIERRA Sets Foundation to Leverage Robust Iomab - B Data • Extensive data and consistent results from more than 400 patients including Fred Hutchinson Cancer Center studies demonstrate high BMT access and engraftment and improved outcomes in patients with MDS, younger AML, ALL, HL/NHL and MM • These indications represent tens of thousands of patients with R/R disease having similar unmet needs to the AML patients in SIERRA • These data, together with the strong results from the pivotal Phase 3 trial will be leveraged to execute our comprehensive life cycle management strategies to further expand Iomab - B’s role in these variety of malignant and non - malignant hematological disorders • Patients are treated by BMT physicians in the same concentrated, high - volume centers, most of which are SIERRA sites Life cycle management and indication expansion opportunity supported by significant body of data with Iomab - B 400+ patients 12 clinical trials 6 diseases (AML, MDS, MM, ALL, NHL/HL) Improved survival and curative outcomes 30
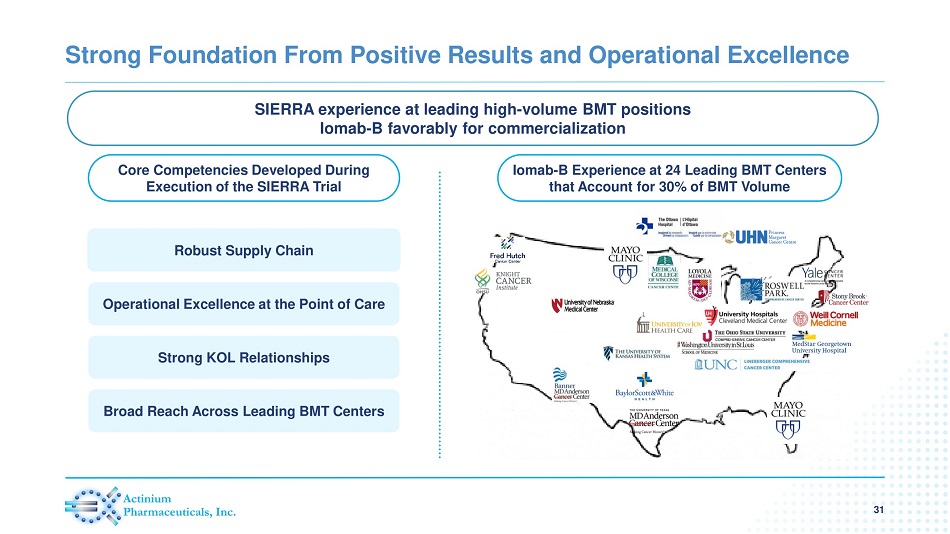
Core Competencies Developed During Execution of the SIERRA Trial Iomab - B Experience at 24 Leading BMT Centers that Account for 30% of BMT Volume Strong Foundation From Positive Results and Operational Excellence 31 Robust Supply Chain Operational Excellence at the Point of Care Strong KOL Relationships Broad Reach Across Leading BMT Centers % Increase in OS SIERRA experience at leading high - volume BMT positions Iomab - B favorably for commercialization

Caroline Yarbrough, Chief Commercial Officer 32 • Joined Actinium in October 2022 with 25 years of BioPharma commercial experience • Most recently, Portfolio General Manager, US Oncology at Novartis, a $1+ Billion product portfolio • Strong hematology experience from leading Novartis Chronic Myelogenous Leukemia (CML) portfolio including Scemblix and Tasigna • Led strategic account management to support the launch of the first approved CAR - T therapy Kymriah • Significant prior experience at GSK, BMS, Viropharma and Merck • Deep understanding of the hematology market, CAR - T/BMT center dynamics, and experience with strategic planning and growing businesses in multiple oncology disease areas
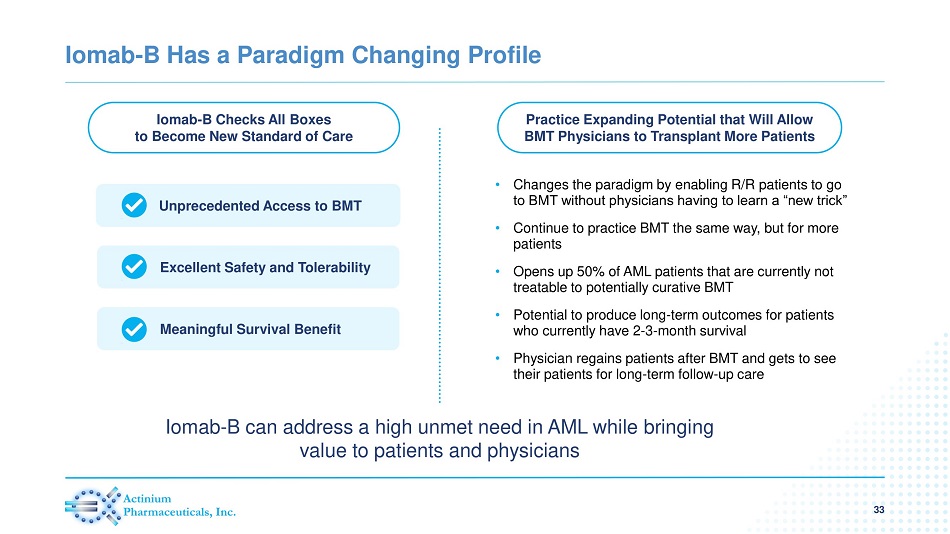
Iomab - B Has a Paradigm Changing Profile Iomab - B Checks All Boxes to Become New Standard of Care Practice Expanding Potential that Will Allow BMT Physicians to Transplant More Patients Unprecedented Access to BMT Excellent Safety and Tolerability Meaningful Survival Benefit • Changes the paradigm by enabling R/R patients to go to BMT without physicians having to learn a “new trick” • Continue to practice BMT the same way, but for more patients • Opens up 50% of AML patients that are currently not treatable to potentially curative BMT • Potential to produce long - term outcomes for patients who currently have 2 - 3 - month survival • Physician regains patients after BMT and gets to see their patients for long - term follow - up care Iomab - B can address a high unmet need in AML while bringing value to patients and physicians 33

Highly Favorable Dynamics Support Iomab - B Commercial Prospects Patients • Unprecedented access to potentially curative BMT in AML with favorable safety and tolerability Physicians • Increased opportunity to treat patients that previously had no other treatment options without disruption to current practice • Referring physicians get their patient back post - BMT for follow - up and long - term care Payors • Strong pharmacoeconomic value proposition driven by Iomab - B product profile Competition • No direct or indirect visible competition for the next 5 - 10 years Concentrated Market • Top 50 centers perform 75% of BMTs; concentrated in metropolitan areas • 12 - 15 account managers to cover BMT center footprint , 35 - 50 - person commercial organization 3P’s, 2C’s: Key variables impacting commercial success align favorably toward Iomab - B 34
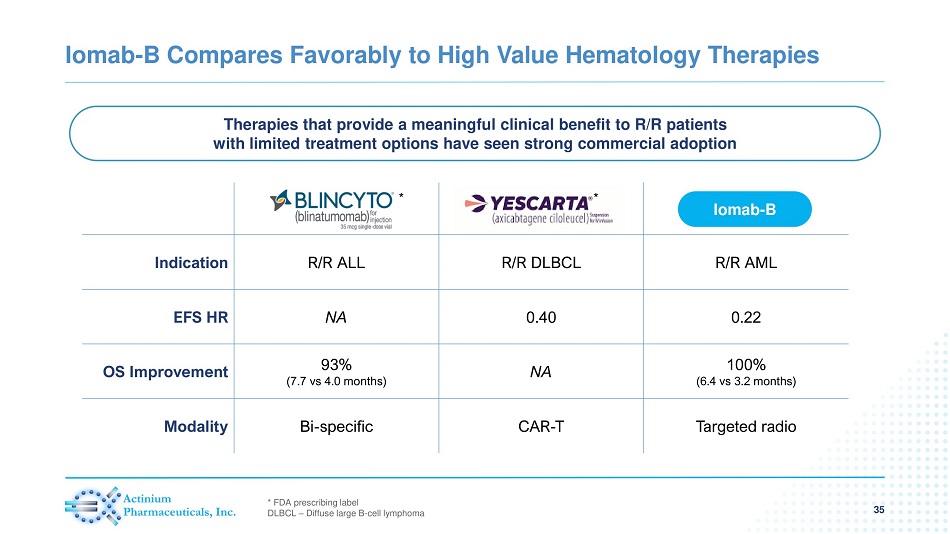
Indication R/R ALL R/R DLBCL R/R AML EFS HR NA 0.40 0.22 OS Improvement 93% (7.7 vs 4.0 months) NA 100% (6.4 vs 3.2 months) Modality Bi - specific CAR - T Targeted radio Iomab - B Compares Favorably to High Value Hematology Therapies 35 Iomab - B Therapies that provide a meaningful clinical benefit to R/R patients with limited treatment options have seen strong commercial adoption * FDA prescribing label DLBCL – Diffuse large B - cell lymphoma * *

Closing Remarks Actinium Pharmaceuticals, Inc.
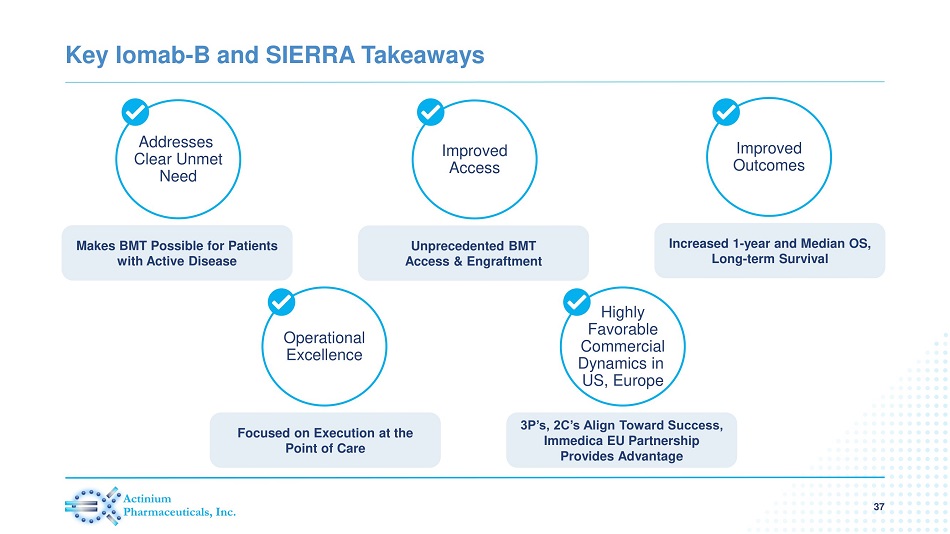
Key Iomab - B and SIERRA Takeaways 37 Addresses Clear Unmet Need Makes BMT Possible for Patients with Active Disease Improved Access Unprecedented BMT Access & Engraftment Improved Outcomes Increased 1 - year and Median OS, Long - term Survival Highly Favorable Commercial Dynamics in US, Europe 3P’s, 2C’s Align Toward Success, Immedica EU Partnership Provides Advantage Operational Excellence Focused on Execution at the Point of Care
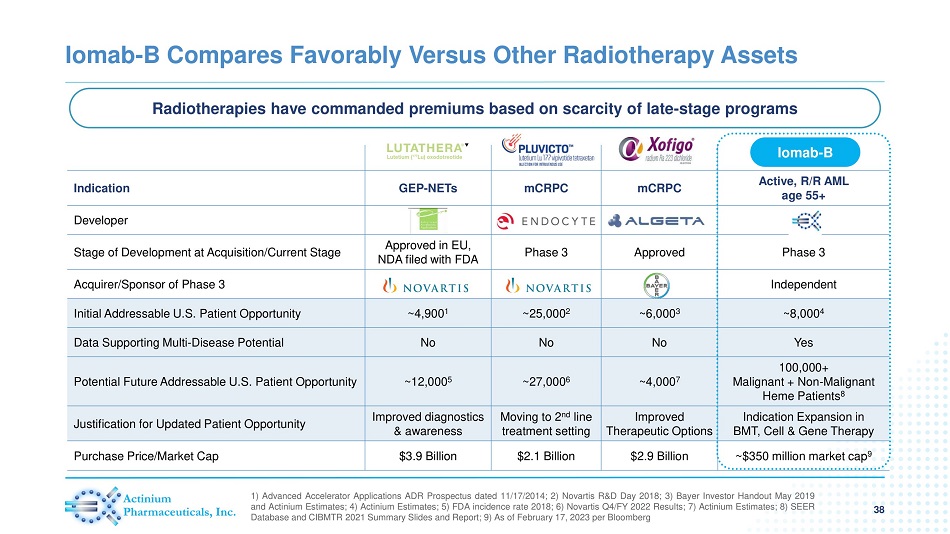
Iomab - B Compares Favorably Versus Other Radiotherapy Assets 38 1 ) Advanced Accelerator Applications ADR Prospectus dated 11 / 17 / 2014 ; 2 ) Novartis R&D Day 2018 ; 3 ) Bayer Investor Handout May 2019 and Actinium Estimates ; 4 ) Actinium Estimates ; 5 ) FDA incidence rate 2018 ; 6 ) Novartis Q 4 /FY 2022 Results ; 7 ) Actinium Estimates ; 8 ) SEER Database and CIBMTR 2021 Summary Slides and Report ; 9 ) As of February 17 , 2023 per Bloomberg Indication GEP - NETs mCRPC mCRPC Active, R/R AML age 55+ Developer Stage of Development at Acquisition/Current Stage Approved in EU, NDA filed with FDA Phase 3 Approved Phase 3 Acquirer/Sponsor of Phase 3 Independent Initial Addressable U.S. Patient Opportunity ~4,900 1 ~25,000 2 ~6,000 3 ~8,000 4 Data Supporting Multi - Disease Potential No No No Yes Potential Future Addressable U.S. Patient Opportunity ~12,000 5 ~27,000 6 ~4,000 7 100,000+ Malignant + Non - Malignant Heme Patients 8 Justification for Updated Patient Opportunity Improved diagnostics & awareness Moving to 2 nd line treatment setting Improved Therapeutic Options Indication Expansion in BMT, Cell & Gene Therapy Purchase Price/Market Cap $3.9 Billion $2.1 Billion $2.9 Billion ~$350 million market cap 9 Iomab - B Radiotherapies have commanded premiums based on scarcity of late - stage programs

Actimab - A Development Bolstered By Recent NCI CRADA 39 NCI sponsored Actimab - A development to include Phase 1, 2 and 3 trials featuring with the potential to develop Actimab - A as a backbone of AML combination therapy Balance sheet sparing with full rights to data – Actinium supplies Actimab - A while NCI covers all clinical trial execution and development expenses • Survival benefit in CLAG - M combination demonstrates potential in high - risk, heavily treated R/R patients • 150 patients treated with Actimab - A as monotherapy or combination, m ost clinical experience with Ac - 225 isotope • Broad applicability – agnostic to mutations and cytogenetics enables broad utilization • CRADA provides access to 2,000 clinical trial sites in Experimental Clinical Trial Network – ECOG, SWOG and Alliance and NCI’s MyeloMATCH program • Actinium to review and approve trials and protocols with NCI and has rights to all data • Enables broad and aggressive development as single agent and combination - backbone
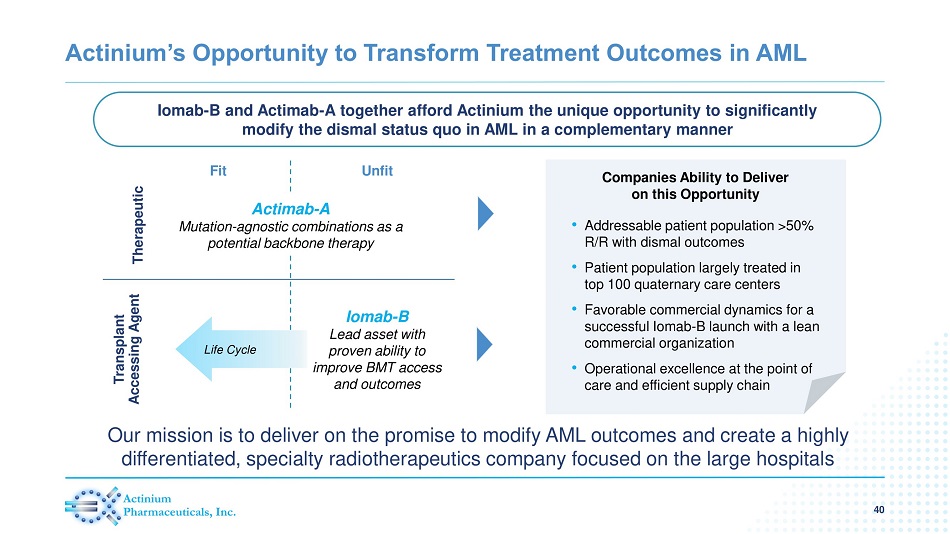
Actinium’s Opportunity to Transform Treatment Outcomes in AML • Addressable patient population >50% R/R with dismal outcomes • Patient population largely treated in top 100 quaternary care centers • Favorable commercial dynamics for a successful Iomab - B launch with a lean commercial organization • Operational excellence at the point of care and efficient supply chain Iomab - B and Actimab - A together afford Actinium the unique opportunity to significantly modify the dismal status quo in AML in a complementary manner 40 Companies Ability to Deliver on this Opportunity Therapeutic Transplant Accessing Agent Fit Unfit Actimab - A Mutation - agnostic combinations as a potential backbone therapy Life Cycle Iomab - B Lead asset with proven ability to improve BMT access and outcomes Our mission is to deliver on the promise to modify AML outcomes and create a highly differentiated, specialty radiotherapeutics company focused on the large hospitals

Q&A Actinium Pharmaceuticals, Inc.

ATNM: NYSE AMERICAN February 18, 2022 Thank you Actinium Pharmaceuticals, Inc.